The Glaucoma Suspect: When to Treat?
Distinguishing healthy persons in the general population from those at considerably increased risk for chronic open-angle glaucoma (COAG) is important because patients in the latter group—commonly referred to as “glaucoma suspects”—need to be followed up more carefully to decide whether and how to begin prophylactic therapy. This chapter outlines the definition and prevalence of glaucoma suspect and reviews key diagnostic elements that need to be considered. The chapter also highlights challenges in management, summarizing the results of the Ocular Hypertension Treatment Study (OHTS), and addresses when it may be appropriate to initiate therapy. Practical guidelines for follow-up are also offered.
TERMINOLOGY
The term ocular hypertension was advocated in the 1970s to distinguish persons with “normal” intraocular pressure (IOP) (i.e., ≤21 mm Hg) from those with an IOP greater than 21 mm Hg, who were considered to be at increased risk for COAG (1,2). Chandler and Grant (3) suggested referring to this condition as early open-angle glaucoma without damage.
However, in addition to those with consistently elevated IOP, there are individuals who exhibit optic nerve features suggestive of early glaucoma or who have suspicious visual field defects. To include these categories and identify a subpopulation of individuals or eyes at increased risk for COAG glaucoma (Table 10.1) (4), the term “glaucoma suspect” was advocated by Shaffer (5). There are also patients at higher risk for angle-closure glaucoma—for example, those with an occludable angle as determined by gonioscopy. Given recent advances in molecular genetics, there are also patients who can be identified as having an elevated risk for glaucomatous optic nerve damage by virtue of harboring one or more disease-causing genetic mutations (see Chapter 8). In this chapter, we will use the term glaucoma suspect in the context of a patient at greater-than-average risk (compared with the general population) for COAG. (Individuals at increased risk for angle-closure glaucoma are discussed in Chapter 12, and those at increased risk by virtue of a genetic susceptibility are discussed in Chapter 8.)

PREVALENCE AND DEVELOPMENT OF CHRONIC OPEN-ANGLE GLAUCOMA
Studies that have used a definition of IOP greater than or equal to 21 mm Hg in one or both eyes (with normal visual fields and optic nerves) have reported a prevalence rate of 4% to 10% in persons older than 40 years (6–11).
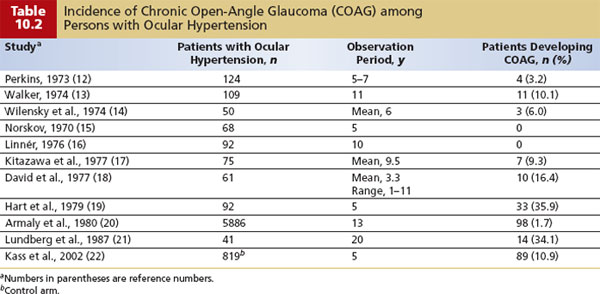
Patients considered to be glaucoma suspects on the basis of elevated IOP have a rate of progression to COAG of approximately 1% per year over 5 to 15 years (Table 10.2) (12–25). In the OHTS (22), a randomized trial of topical ocular hypotensive treatment versus close observation in participants with “ocular hypertension,” the cumulative probability of developing COAG over 5 years was about 1% per year in the medication group and about 2% per year in the observation group. In patients who are at higher risk for developing glaucomatous optic nerve damage (see risk factors in Table 10.3), the rate is approximately 3% to 5% per year (26–28).

Although elevated IOP is a major risk factor for COAG, normotensive individuals can develop glaucoma. Some of these patients may have normal-tension glaucoma (see Chapter 11), whereas others may demonstrate elevated IOP at subsequent examinations (12,23).
SCREENING AND EARLY DETECTION
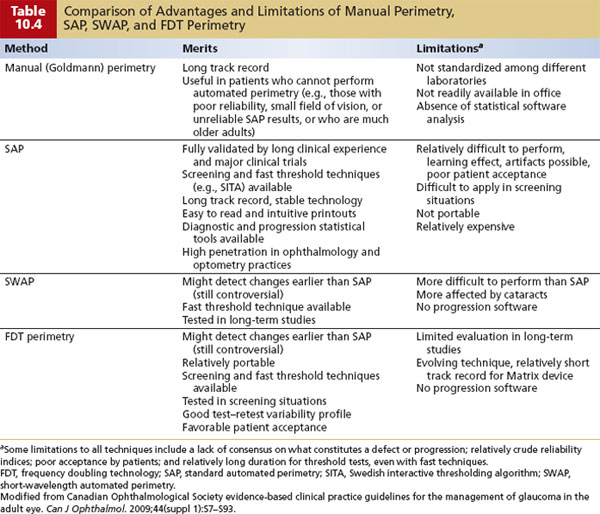
Screening is discussed in Chapter 9. In brief, if an IOP value of more than 21 mm Hg is used for screening, then there is a high rate of false-positive and false-negative results for COAG. Skilled optic nerve examination is good but not always practical. Standard automated perimetry (SAP) can detect glaucomatous defects, but by the time a defect is detected, a substantial loss of axons has often occurred (29,30). A number of studies have demonstrated that defects on short-wavelength automated perimetry (SWAP) and frequency doubling technology (FDT) perimetry can precede development of SAP-detected defects in patients with elevated IOP. Table 10.4 provides a comparative summary of these types of perimeters (31). Imaging devices may also be useful in the early detection of glaucoma. (These are covered in greater detail in Chapter 4 and below.)
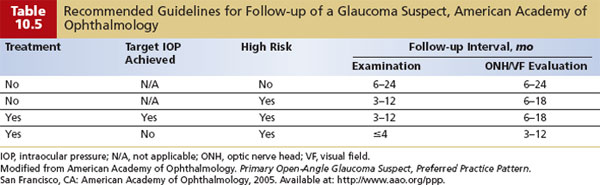
According to the American Academy of Ophthalmology, the best method to detect early glaucoma is a comprehensive eye evaluation, which includes assessment of the IOP, optic nerve, and visual field (see Chapter 9). Guidelines for frequency of screening for glaucoma are listed in Table 10.5 (4).
Intraocular Pressure and Pachymetry
To detect any change in IOP, optic nerve, or visual field status (i.e., early progression with structural or functional damage evident), it is essential to obtain good baseline documentation. In the case of IOP, it is worthwhile to measure central corneal thickness (CCT) with a pachymeter (Fig. 10.1). Patients classified as glaucoma suspects have been reported to have a higher CCT than individuals with COAG or healthy individuals (32–34), with 42% of glaucoma suspects having a CCT of greater than 585 µm (34). This is significant because the Goldmann applanation tonometer was calibrated for a CCT of approximately 530 µm (35,36). Any significant deviation from this induces an artifact of measurement. It has been estimated that 30% to 57% of elevated IOPs in glaucoma suspects are actually artifacts of measurement (33,37,38).
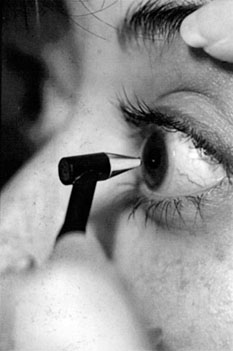
Figure 10.1 Proper technique for measuring CCT, with probe placed perpendicular to central cornea. A structurally thick cornea can artifactually raise measured applanation IOP.
There is no universally accepted formula, however, that can be applied to “correct” the IOP measurement for any given CCT. Based on a review of various correction-factor approaches, the range probably falls between 2.5 and 3.5 mm Hg per 50 µm of difference from normal (39). Hence, if a patient’s CCT measured 650 µm (in the absence of any visible corneal pathology), then the “true” IOP would likely be several millimeters of mercury less than measured. To avoid confusion, however, when sharing patient information with other practitioners, it is recommended that IOP should always be communicated as the measured IOP rather than a “corrected” IOP.
Slitlamp Biomicroscopy and Gonioscopy
Baseline documentation requires precise slitlamp examination and gonioscopy to exclude secondary causes of glaucoma. This includes angle closure and other secondary causes, such as angle recession, pigment dispersion, and inflammatory forms of glaucoma. After dilation, the anterior lens capsule should be examined for the presence of exfoliation.
Fundus Examination
In the posterior segment, it is important to document the appearance of the optic nerve head with careful drawings or stereo optic nerve head photos. Optic nerve head imaging devices (e.g., confocal laser scanning tomography) may also be useful. It is also worth studying the disc rim carefully for small hemorrhages, because these can precede visual field loss and future optic nerve damage. Similarly, the appearance of the nerve fiber layer (NFL) can be noted using red-free (green) light. It is important to document the presence or absence of NFL defects. Additional tools to document the NFL include laser polarimetry with the nerve fiber analyzer, scanning laser ophthalmoscopy, and optical coherence tomography (OCT) (see Chapter 4).
Visual Fields
An attempt should be made to obtain two or three baseline visual fields. Our preferred options include one or more of the following: (a) 24-2 Swedish interactive threshold algorithm (SITA) standard on Humphrey field analyzer II perimeter, (b) 24-2 full threshold white-on-white Humphrey perimetry or equivalent program on a different automated perimeter, (c) FDT (matrix preferred) or SWAP.
If an abnormality is found, it needs to be confirmed on repeated visual field examination. This was dramatically illustrated in OHTS (40). Over a 5-year period, 21,603 visual fields were obtained from 1637 OHTS participants. When follow-up visual field results were outside the normal limits on the Glaucoma Hemifield Test, the Corrected Pattern Standard Deviation, or both, follow-up visual fields were obtained to confirm the abnormality. Results of 748 visual fields were abnormal; of these, 703 (94%) were abnormal and reliable, and 45 (6%) were abnormal and unreliable. On retesting, abnormalities were not confirmed for 604 (85.9%) of the originally abnormal and reliable visual fields. Hence, most visual field abnormalities in OHTS participants were not verified on repeated testing and were probably due to the learning curve or long-term variability in the visual field.
Imaging of the Optic Nerve and Nerve Fiber Layer
Photographic assessment of the optic nerve head remains a mainstay in the diagnosis and management of glaucoma suspects. However, there are imaging tools capable of documenting the topographic features of the optic nerve head and measuring the thickness of the retinal NFL that can be useful adjuncts in the management of glaucoma suspects. These tools are reviewed in Chapter 4 and include the confocal scanning laser ophthalmoscope (manufactured as Heidelberg retinal tomography [HRT]), OCT, and scanning laser polarimetry (e.g., the GDx nerve fiber analyzer with variable corneal compensator [GDx-VCC]). Each of the technologies has good reproducibility and provides objective and quantitative analysis of ocular structure. An evidence-based medicine review of these technologies by the American Academy of Ophthalmology (41) came to the following conclusion:
The [optic nerve head] and [retinal] NFL imaging devices provide quantitative information for the clinician. Based on studies that have compared the various available technologies directly, there is no single imaging device that outperforms the others in distinguishing patients with glaucoma from controls …. The information obtained from imaging devices is useful in clinical practice when analyzed in conjunction with other relevant parameters that define glaucoma diagnosis and progression.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


