Purpose
To examine the association between best corrected visual acuity (BCVA) and subfoveal choroidal thickness.
Design
Population-based study.
Methods
The Beijing Eye Study 2011 included 3468 subjects with ages of 50+ years. The participants underwent an ophthalmologic examination including spectral-domain optical coherence tomography with enhanced depth imaging for measurement of choroidal thickness. BCVA was measured as logarithm of the minimal angle of resolution.
Results
Of the 3468 participants, choroidal measurements were available for 3233 (93.2%) subjects. In multivariate analysis, better BCVA was significantly associated with thicker subfoveal choroid ( P < 0.001) in general and a subfoveal choroid thicker than 30 μm ( P < 0.001) in particular, after adjusting for younger age ( P < 0.001), higher level of education ( P < 0.001), taller body stature ( P < 0.001), higher body mass index ( P = 0.005), absence of glaucoma ( P = 0.001), absence of diabetic retinopathy ( P < 0.001), absence of late-stage age-related macular degeneration ( P < 0.001), and axial length shorter than 26.0 mm ( P < 0.001) (correlation coefficient r:0.56). If eyes with glaucoma, diabetic retinopathy, late-stage age-related macular degeneration or myopic retinopathy were excluded, better BCVA was still significantly associated with thicker subfoveal choroid ( P < 0.001) and subfoveal choroid thicker than 30 μm ( P < 0.001) in multivariate analysis. In a reverse manner, thicker subfoveal choroid was associated with better BCVA ( P < 0.001) after adjusting for younger age ( P < 0.001), male gender ( P < 0.001), longer axial length ( P < 0.001), and higher corneal curvature radius ( P < 0.001).
Conclusions
Better visual acuity is strongly associated with thicker subfoveal choroid independent of additional factors, such as age, axial length, education level, and major ocular diseases.
Best corrected visual acuity (BCVA), as 1 of the main parameters of visual function, depends on a multitude of factors, including age, transparency of the optic media and presence of optic nerve or retinal diseases including myopic retinopathy. Recent studies revealed that subfoveal choroidal thickness may be added to the list of factors potentially associated with visual acuity. These investigations by Nishida and colleagues, Flores-Moreno and coworkers, and Ho and associates included, however, relatively few subjects, had a study design with a hospital-based recruitment of the study participants, and examined the potential association between BCVA and subfoveal choroidal thickness in univariate analyses, although both BCVA and subfoveal choroidal thickness depend on a multitude of factors, such as age and axial length, which influence both BCVA and subfoveal choroidal thickness. We conducted the present investigation to examine the potential influence of subfoveal choroidal thickness on visual acuity in a multivariate analysis of several factors that have an influence on both visual acuity and subfoveal choroidal thickness. We chose a population-based study design to avoid the risk of a referral bias, which is inherent in any hospital-based study.
Methods
The Beijing Eye Study 2011 is a population-based cross-sectional study based in northern China. It has been described in detail previously. The study design was approved by the Medical Ethics Committee of the Beijing Tongren Hospital, and all participants prospectively gave informed consent. Of a total eligible population of 4403 individuals 50 years of age or older, 3468 (78.8%) individuals participated. The mean age was 64.6 ± 9.8 years (median, 64 years; range, 50–93 years). All study participants underwent an interview with standardized questions, including questions about their socioeconomic backgrounds.
BCVA was assessed by automatic refractometry (Auto Refractometer AR-610; Nidek, Tokyo, Japan). If uncorrected visual acuity was lower than 1.0, we additionally performed subjective refractometry to measure BCVA. The ophthalmic examination additionally included tonometry, slit-lamp examination of the anterior and posterior segment of the eye, and digital photography the cornea, lens, macula, and optic disc. Spectral domain optical coherence tomography (OCT) (Spectralis, Wavelength 870 nm; Heidelberg Engineering, Heidelberg, Germany) with enhanced depth imaging modality was performed after pupil dilation. We obtained 31 sections, which covered a 30 degree × 30 degree rectangle centered on the fovea. Using the enhanced-depth imaging mode, 7 additional sections (each comprising 100 averaged scans) were obtained in a 5 degree × 30 degree rectangle centered on the fovea. The horizontal section running through the center of the fovea was selected for measurement of subfoveal choroidal thickness. Subfoveal choroidal thickness was defined as the vertical distance between the hyper-reflective line of the Bruch membrane and the hyper-reflective line of the inner surface of the sclera. Only the right eye of each study participant was assessed. The images were taken by 1 technician (CXC), and the images were assessed by 2 ophthalmologists (LS, KFD). Using the OCT, we additionally measured the retinal nerve fiber thickness as described recently. Biometry for measurement of the anterior corneal curvature, central corneal thickness, anterior chamber depth, lens thickness, and axial length (optical low-coherence reflectometry; Lensstar 900 Optical Biometer; Haag-Streit, Koeniz, Switzerland) was carried out in the right eyes of the study participants. Using fundus photographs, we also assessed the presence of retinal vein occlusions, diabetic retinopathy, glaucomatous optic neuropathy, and age-related macular degeneration, as defined and described in detail previously. The late stage of age-related macular degeneration was defined as either neovascular age-related macular degeneration or geographic atrophy. Neovascular age-related macular degeneration included serous or hemorrhagic detachment of the retinal pigment epithelium or sensory retina; intraretinal or subretinal hemorrhages or hemorrhages beneath the retinal pigment epithelium, or a combination thereof; or subretinal fibrous scars.
Additionally, we examined fasting blood samples for the concentration of blood lipids, glucose, glycosylated hemoglobin A1C, creatinine, and C-reactive protein, and we measured body height and weight and blood pressure. All OCT sections of all highly myopic eyes defined as a myopic refractive error of more than -6 diopters or an axial length of more than 26.5 mm were examined for epiretinal membranes, vitreofoveal adhesion, incomplete posterior vitreous detachment, retinoschisis, macular holes, disruption of the photoreceptor inner segment or outer segment interface, changes in the Bruch membrane such as disruption, and clumping of the retinal pigment epithelium.
Statistical analysis was performed using a commercially available statistical software package (SPSS for Windows, v 21.0, IBM-SPSS, Chicago, Illinois, USA). For the analysis of BCVA, we converted the measurements into the logarithm of the minimal angle of resolution (logMAR). In the first step, we examined the mean values (presented as mean ± standard deviation) of the parameters. In the second step, we performed a univariate analysis, with BCVA as the dependent parameter and ocular and general parameters as independent parameters. In the third step, we performed a multivariate regression analysis, with BCVA as a dependent parameter and all those parameters as independent parameters that were significantly associated with subfoveal choroidal thickness in univariate analysis. We then dropped in a step-wise manner all those parameters from the list of independent variables that were no longer significantly associated with BCVA in the multivariate analysis. In a similar manner, we performed a multivariate analysis with subfoveal choroidal thickness as a dependent parameter; 95% confidence intervals (CIs) were presented. All P values were 2-sided and were considered statistically significant when the values were less than 0.05.
Results
Of the 3468 participants, measurements of subfoveal choroidal thickness were available for 3233 (93.2%) subjects, (1818 (56.2%) women). The mean age was 64.3 ± 9.6 years (median, 63 years; range, 50–93 years); the mean refractive error (spherical equivalent) was −0.18 ± 1.98 diopters (median, 0.25 diopters; range, −20.0–+7.00 diopters); and the mean axial length was 23.2 ± 1.11 mm (median, 23.13 mm; range, 18.96–30.88 mm). As also previously described, the group of subjects without subfoveal choroidal thickness measurements as compared with the group of subjects with subfoveal choroidal thickness measurements was significantly older (69.6 ± 10.9 years) ( P < 0.001) and was more myopic (−1.44 ± 4.75 diopters vs P = 0.007) and did not vary significantly in gender ( P = 0.12).
Mean BCVA was 0.04 ± 0.15 logMAR, and mean subfoveal choroidal thickness was 254 ± 107.4 μm (median, 251 μm; range, 8 μm–54 μm).
In univariate analysis, higher BCVA was significantly associated with the systemic parameters of younger age ( P < 0.001), male gender ( P < 0.001), higher level of education ( P < 0.001), lower systolic blood pressure ( P < 0.001), higher diastolic blood pressure ( P = 0.002), higher body height ( P < 0.001), greater weight ( P < 0.001) and body mass index ( P < 0.001); and with the ocular parameters of thicker subfoveal choroidal thickness ( P < 0.001), thicker retinal nerve fiber layer ( P < 0.001), higher intraocular pressure ( P < 0.001), shorter axial length ( P < 0.001), thinner lens ( P < 0.001), flatter corneal curvature ( P = 0.001), absence of glaucoma ( P < 0.001), diabetic retinopathy ( P < 0.001), retinal vein occlusion ( P < 0.001), and late age-related macular degeneration ( P < 0.001), lower degree of nuclear cataract ( P < 0.001), cortical cataract ( P < 0.001) and subcapsular cataract ( P < 0.001), and absence of myopic retinopathy-related clumping of the retinal pigment epithelium in the macular region ( P = 0.04), epiretinal membranes ( P < 0.001), disruption of the inner segment/outer segment interface ( P < 0.001), maculoschisis ( P < 0.001), macular defects in the Bruch membrane, and staphylomata ( P < 0.001). BCVA was not significantly associated with central corneal thickness ( P = 0.93), optic disc size ( P = 0.20), neuroretinal rim area ( P = 0.53), or vertical cup/disc diameter ratio ( P = 0.20).
The association between subfoveal choroidal thickness and BCVA, expressed as logMAR or in Snellen units, was not linear but showed a curvilinear form ( Figure 1 , Figure 2 , Figure 3 ). On the charts showing the relationship between subfoveal choroidal thickness and BCVA, the curve for BCVA sharply increased at a subfoveal choroidal thickness of approximately 50 μm ( Figure 1 , Figure 2 ). In the subsequent multivariate analysis, we therefore introduced an additional parameter indicating whether the subfoveal choroidal thickness was thinner than a defined cut-off value.
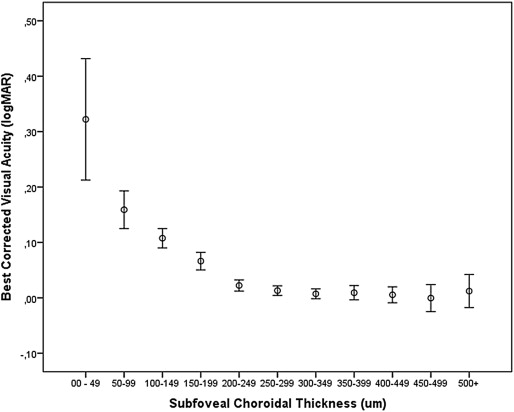
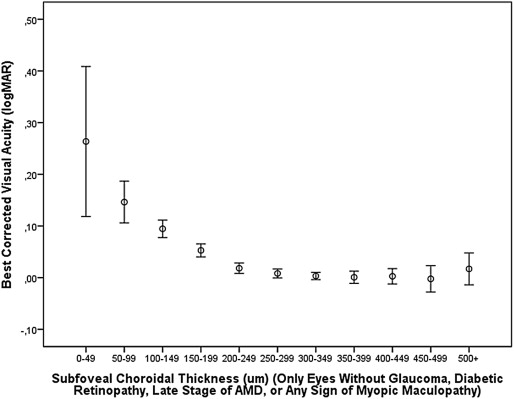
In the multivariate analysis, all parameters were included as independent variables that were significantly associated with BCVA in the univariate analysis. We then excluded body weight due to a variance inflation factor higher than 3. In the next step, we dropped gender ( P = 0.43), diastolic blood pressure ( P = 0.71), systolic blood pressure ( P = 0.78), lens thickness ( P = 0.36), myopic retinopathy with epiretinal membranes ( P = 0.74), corneal curvature ( P = 0.15), macular Bruch membrane defects ( P = 0.51), macular staphyloma ( P = 0.25), intraocular pressure ( P = 0.11), presence of retinal vein occlusions ( P = 0.14), disruption of the macular photoreceptor inner segment/outer segment interface ( P = 0.08), and retinal nerve fiber layer thickness ( P = 0.10). In the subsequent model, BCVA (logMAR) remained significantly associated with younger age, higher level of education, taller body stature, higher body mass index, thicker subfoveal choroidal thickness, absence of glaucoma, absence of diabetic retinopathy, absence of the late stage of age-related macular degeneration, and axial length shorter than 26.0 mm. We then added the parameter of subfoveal choroidal thickness thinner than 30, 40, 50, 60, 70, 80, 90, 100, 110, or 120 μm, respectively, and assessed at which cut-off value the correlation coefficient was highest. It revealed that with a cut-off value of a subfoveal choroidal thickness equal to or thinner than 30 μm, the highest overall correlation coefficient (r = 0.556) and standardized correlation coefficient beta for this parameter (beta, 0.12) were achieved ( Table 1 ). If the logarithm of the parameter of subfoveal choroidal thickness was taken for the statistical analysis, a similar result was obtained. In the diagram of the logarithm of subfoveal choroidal thickness vs BCVA, the logMAR value of BCVA sharply increased at a logarithm value of <1.50 (equivalent to a non-logarithmic value of 32 μm of choroidal thickness). If all eyes with glaucoma, diabetic retinopathy, late-stage age-related macular degeneration, or any sign of myopic retinopathy as examined by OCT (ie, vitreofoveal traction, epiretinal membrane, retinoschisis, chorioretinal neovascularization, destruction of the inner segment or outer segment interface, clumping of the retinal pigment epithelium, macular holes, disruption of the macular Bruch membrane, or macular staphyloma) were excluded, better BCVA (logMAR) was still significantly associated with thicker subfoveal choroidal thickness ( P < 0.001) and subfoveal choroidal thickness greater than 30 μm ( P < 0.001), after adjusting for younger age ( P < 0.001), higher level of education ( P < 0.001), higher body height ( P < 0.001), higher body mass index ( P = 0.001), and axial length ≤26 mm ( P < 0.001) ( Table 2 ).
| Parameter | P Value | Standardized Coefficient beta | Regression Coefficient | 95% Confidence Intervals | Variance Inflation Factor |
|---|---|---|---|---|---|
| Age (years) | <0.001 | 0.37 | 0.006 | 0.005, 0.006 | 1.31 |
| Level of education | <0.001 | −0.19 | −0.03 | −0.03, −0.02 | 1.15 |
| Body height (cm) | <0.001 | −0.08 | −0.001 | −0.002, −0.001 | 1.13 |
| Body mass index (kg/m 2 ) | 0.005 | −0.05 | −0.002 | −0.003, −0.001 | 1.08 |
| Presence of glaucomatous optic neuropathy | 0.001 | 0.05 | 0.03 | 0.01, 0.05 | 1.03 |
| Presence of diabetic retinopathy | <0.001 | 0.13 | 0.12 | 0.09, 0.14 | 1.01 |
| Late age-related macular degeneration | <0.001 | 0.07 | 0.11 | 0.06, 0.16 | 1.01 |
| Axial length longer than 26 mm | <0.001 | 0.09 | 0.07 | 0.05, 0.10 | 1.12 |
| Subfoveal choroidal thickness (μm) | <0.001 | −0.08 | −0.00007 | 0.000, 0.000 | 1.31 |
| Subfoveal choroidal thickness thinner than 30 μm | <0.001 | 0.12 | 0.31 | 0.23, 0.38 | 1.08 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


