Purpose
To investigate whether the prophylactic use of bevacizumab reduces the rate of rubeosis after proton therapy for uveal melanoma and improves the possibility to treat ischemic, reapplicated retina with laser photocoagulation.
Design
Comparative retrospective case series.
Methods
Uveal melanoma patients with ischemic retinal detachment and treated with proton therapy were included in this institutional study. Twenty-four eyes received prophylactic intravitreal bevacizumab injections and were compared with a control group of 44 eyes without bevacizumab treatment. Bevacizumab injections were performed at the time of tantalum clip insertion and were repeated every 2 months during 6 months, and every 3 months thereafter. Ultra-widefield angiography allowed determination of the extent of retinal ischemia, which was treated with laser photocoagulation after retinal reapplication. Main outcome measures were the time to rubeosis, the time to retinal reattachment, and the time to laser photocoagulation of ischemic retina.
Results
Baseline characteristics were balanced between the groups, except for thicker tumors and larger retinal detachments in the bevacizumab group, potentially to the disadvantage of the study group. Nevertheless, bevacizumab prophylaxis significantly reduced the rate of iris rubeosis from 36% to 4% (log-rank test P = .02) and tended to shorten the time to retinal reapplication until laser photocoagulation of the nonperfusion areas could be performed.
Conclusions
Prophylactic intravitreal bevacizumab in patients treated with proton therapy for uveal melanoma with ischemic retinal detachment prevented anterior segment neovascularization, until laser photocoagulation to the reapplied retina could be performed.
Proton therapy for uveal melanoma has shown a very high success rate in terms of local tumor control (94%–99% after 5 years) and eye retention probability (89%–91% after 5 years). However, some irradiated eyes still undergo secondary enucleation owing to radiation-induced complications. Relative risk analyses have indicated that tumor height, tumor diameter, ciliary body involvement, and proximity to the optic disc or fovea are significant risk factors for enucleation. Neovascular glaucoma has been cited as a primary cause of enucleation. Rates of neovascular glaucoma following proton therapy for uveal melanoma vary widely in the literature (7%–47%). Neovascular glaucoma has been associated with tumor size, proximity to the optic nerve or macula, uveal involvement anterior to the equator, and the presence of a serous retinal detachment. Our group has previously demonstrated that Baerveldt shunt implantation is a successful treatment option for neovascular glaucoma in cases of anterior uveal melanoma treated with proton therapy, which negated the need for enucleation owing to neovascular glaucoma. However, the ideal situation would be to prevent iris neovascularization and the development of neovascular glaucoma and avoid this secondary surgical intervention with its associated risks.
The advent of intravitreal anti–vascular endothelial growth factor (VEGF) injections has provided an efficacious treatment for anterior segment neovascularization. However, it does not always prevent secondary enucleation when applied in florid neovascularization. The use of prophylactic intravitreal anti-VEGF to inhibit iris neovascularization in uveal melanoma eyes treated with proton therapy has not been previously reported. The aim of this study was to examine whether the prophylactic use of bevacizumab in eyes with a high risk profile for neovascular glaucoma following proton therapy may reduce the rate of anterior segment neovascularization. In addition, we investigated whether the use of anti-VEGF increased the rate of retinal reapplication and reduced the time until photocoagulation of ischemic retina became possible.
Methods
This study is an interventional case series that was retrospectively compared with a historic control group. This study was approved by the local ethics committee. Patients gave their informed consent for the study intervention (bevacizumab injections).
Since October 2007, patients referred to the Ocular Oncology Unit of the Jules-Gonin Eye Hospital (Lausanne, Switzerland) for posterior uveal melanoma were examined with fluorescein and indocyanine green ultra-widefield angiography, using the Staurenghi 230 SLO Retina Lens (Ocular Instruments, Inc, Bellevue, Washington, USA) and the HRA 2 (Heidelberg Engineering GmbH, Heidelberg, Germany). These 150-degree images facilitated the global assessment of tumor characteristics and precisely documented the presence or absence of retinal/choroidal ischemia ( Figure 1 ).
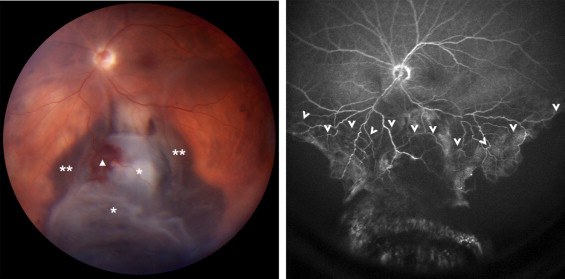
From September 2009 onwards, all uveal melanoma patients with secondary ischemic retinal detachment who were to undergo proton therapy were offered a prophylactic, adjuvant anti-VEGF treatment. Consenting patients received a first intravitreal bevacizumab injection (1.25 mg in 0.05 mL) following the insertion of tantalum clips and before proton therapy (dose of 60 cobalt gray relative biological effectiveness equivalents in 4 fractions). Following proton therapy, bevacizumab injections were repeated every 2 months for 6 months and every 3 months thereafter until the retina became reattached. When complete reapplication of the retina was achieved, ultra-widefield angiography was repeated in order to define the extension of retinal ischemia. Laser photocoagulation was subsequently applied to the nonperfusion areas, and the injections were stopped ( Figure 2 ).
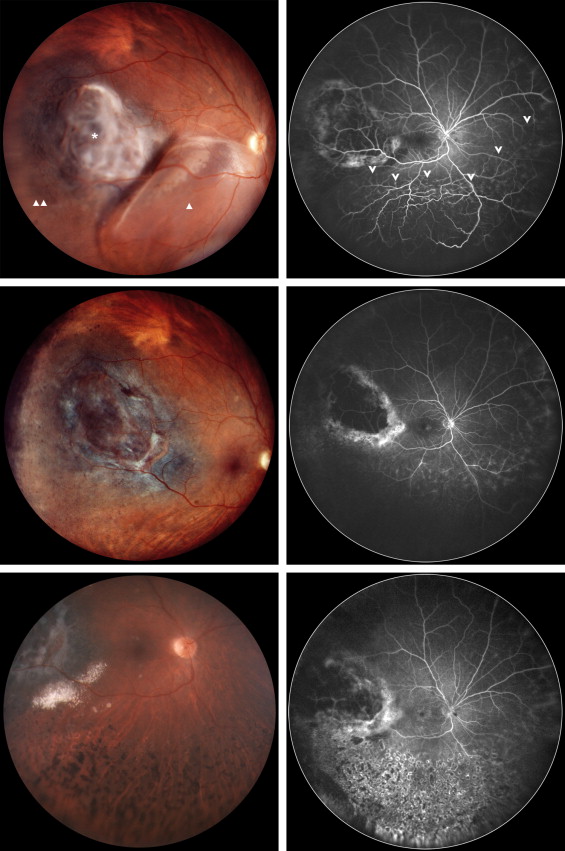
A retrospective review of all 150-degree angiographic images was performed to identify all eyes with uveal melanoma and ischemic retinal detachment at presentation. Eyes that had undergone the prophylactic bevacizumab protocol as described above were labeled as the“B”evacizumab group (Group B). The remaining eyes, which had not received prophylactic anti-VEGF, composed the “C”ontrol group (Group C).
Data were collected from the clinical charts, including a baseline systemic history and complete ophthalmic examination, fundus color photographs with transscleral illumination (Panoret 1000M camera; Medibel/CMT Technology, Haifa, Israel), ultra-widefield angiographies, 10 and 20 MHz B-scan ultrasonography (Quantel Medical CineScan, Version S 5.06; Quantel Medical, Clermont-Fernand, France), and the Paul Scherrer Institute proton therapy plans.
The collected baseline data included: age, sex, laterality, logMAR best-corrected visual acuity (BCVA), intraocular pressure (IOP), estimated extent of serous retinal detachment (in number of quadrants), extent of retinal ischemia (in number of quadrants), tumor characteristics including the7th TNM (tumor, nodes, metastasis) Classification of Malignant Tumors, the presence of an extrascleral extension, tumor size (largest tumor diameter, thickness, and estimated volume), location of the anterior tumor margin relative to the equator, distance from the macula and disc, and proton therapy treatment details (proportion of the dose of collateral irradiation to the ciliary body, disc, and macula) ( Tables 1 and 2 ). Follow-up data that were collected from the medical reports of the 6 months’ interval control visits after proton therapy included BCVA, IOP, extent of retinal detachment, and tumor thickness ( Table 3 ).
| Characteristic | Bevacizumab Group B (n = 24) | Control Group C (n = 44) | P Value a |
|---|---|---|---|
| Sex (F/M) | 14/10 | 16/28 | .14 |
| Laterality (R/L) | 9/15 | 24/20 | .28 |
| TNM classification, n (T 2 –T 3 –T 4 ) | 2–7–15 | 4–16–24 | .78 |
| Tumor margin anterior to the equator, n (%) | 20 (83.3%) | 30 (68.2%) | .18 |
| Extrascleral extension, n (%) | 4 (16.6%) | 3 (6.8%) | .44 |
| Mean (SD), Median [Range] | Mean (SD), Median [Range] | P Value b | |
| Age, y | 50.1 (12.2), 53 [23–69] | 51.4 (16.4), 53 [23–69] | .48 |
| Largest tumor diameter, mm | 19.3 (3.4), 19.25 [11–26] | 18.4 (3.2), 18.4 [11–26] | .41 |
| Tumor thickness, mm | 8.7 (2.5), 8.6 [4–12] | 7.1 (2.3), 7 [2–13] | .05 |
| Tumor volume, mm 3 | 1413 (646), 1461 [209–2560] | 1109 (596), 1160 [182–2969] | .06 |
| Distance to disc, mm | 3.5 (3.1), 3 [0–10] | 3.5 (3.5), 3 [0–13] | .98 |
| Distance to macula, mm | 3.2 (2.3), 3 [0–7] | 3.0 (2.9), 2 [0–10] | .82 |
| Retinal detachment, n of quadrants | 1.8 (0.6), 2 [0–3] | 1.3 (0.8), 1 [0–4] | .01 |
| Retinal ischemia, n of quadrants | 1.0 (0.4), 1 [0–2] | 0.8 (0.5), 1 [0–2] | .08 |
| Group B | Group C | P Value a | |
|---|---|---|---|
| Irradiation dose | 60 Gy | 60 Gy | 1.0 |
| Average proportion of the area that received ≥50% of the irradiation dose | |||
| Ciliary body | 0.63 | 0.50 | .29 |
| Disc | 0.66 | 0.60 | .58 |
| Macula | 0.30 | 0.29 | .65 |
| Average proportion of the area that received ≥90% of the irradiation dose | |||
| Ciliary body | 0.21 | 0.16 | .16 |
| Disc | 0.61 | 0.41 | .09 |
| Macula | 0.57 | 0.54 | .79 |
| Group B | Group C | P Value a | |||
|---|---|---|---|---|---|
| Median/Mean b | Range | Median/Mean b | Range | ||
| Preoperatively | |||||
| BCVA, logMAR | 0.40 | 0–3.0 | 0.26 | −0.1–2.3 | .44 |
| IOP, mm Hg | 14.0 | 9–17 | 14.0 | 7–24 | .54 |
| RD quadrants, n | 1.8 | 0.5–3 | 1.3 | 0.1–4.0 | .01 |
| Tumor thickness, mm | 8.7 | 3.8–12.2 | 7.1 | 2.4–13.5 | .05 |
| Postoperatively, month 6 | |||||
| BCVA, logMAR | 1.65 | 0.2–3.0 | 1 | −0.1–3.0 | .13 |
| IOP, mm Hg | 14 | 7–19 | 14 | 9–45 | .71 |
| RD quadrants, n | 1.8 | 0–4 | 1.4 | 0–4 | .27 |
| Tumor thickness, mm | 6.1 | 2.3–12.1 | 5.5 | 1.9–10.1 | .36 |
| Postoperatively, month 12 | |||||
| BCVA, logMAR | 1.5 | 0.15–3.0 | 1.0 | 0–3.0 | .12 |
| IOP, mm Hg | 13.5 | 3–44 | 14 | 10–42 | .48 |
| RD quadrants, n | 0.9 | 0–4 | 0.7 | 0–4 | .77 |
| Tumor thickness, mm | 5.1 | 1.6–11.4 | 4.3 | 0.9–7.5 | .21 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


