Purpose
To evaluate the outcomes of femtosecond-assisted arcuate keratotomy combined with cataract surgery in eyes with low to moderate corneal astigmatism.
Design
Retrospective, interventional case series.
Methods
This study included patients who underwent combined femtosecond-assisted phacoemulsification and arcuate keratotomy between March 2013 and August 2013. Keratometric astigmatism was evaluated before and 2 months after the surgery. Vector analysis of the astigmatic changes was performed using the Alpins method.
Results
Overall, 54 eyes of 54 patients (18 male and 36 female; mean age, 68.8 ± 11.4 years) were included. The mean preoperative (target-induced astigmatism) and postoperative astigmatism was 1.33 ± 0.57 diopters (D) and 0.87 ± 0.56 D, respectively ( P < .001). The magnitude of error (difference between surgically induced and target-induced astigmatism) (−0.13 ± 0.68 D), as well as the correction index (ratio of surgically induced and target-induced astigmatism) (0.86 ± 0.52), demonstrated slight undercorrection. The angle of error was very close to 0, indicating no significant systematic error of misaligned treatment. However, the absolute angle of error showed a less favorable range (17.5 ± 19.2 degrees), suggesting variable factors such as healing or alignment at an individual level. There were no intraoperative or postoperative complications.
Conclusions
Combined phacoemulsification with arcuate keratotomy using femtosecond laser appears to be a relatively easy and safe means for management of low to moderate corneal astigmatism in cataract surgery candidates. Misalignment at an individual level can reduce its effectiveness. This issue remains to be elucidated in future studies.
Arcuate keratotomy is a commonly used technique to treat astigmatism. It has been used to treat postkeratoplasty astigmatism and astigmatism at the time of cataract surgery. However, manual arcuate keratotomy may be associated with poor predictability because of the lack of reproducibility in incision length and axis alignment. With the advent of femtosecond laser technology, promising results have been reported for reducing astigmatism with arcuate keratotomy. It has been shown to successfully reduce high astigmatism associated with keratoplasty, as well as low to moderate residual astigmatism after cataract or refractive surgeries.
The aim of our study was to evaluate the use of femtosecond-assisted arcuate keratotomy combined with cataract surgery in eyes with low to moderate corneal astigmatism. A single penetrating arcuate keratotomy was paired with an opposite phacoemulsification incision along the steep axis. Vector analysis using the Alpins method was performed.
Methods
This was a retrospective study performed at the Hong Kong Laser Surgery Center in Hong Kong. The Joint Chinese University of Hong Kong–New Territories East Cluster Clinical Research Ethics Committee approved the study protocol. The study adhered to the tenets of the Declaration of Helsinki. The case records of consecutive patients who underwent combined femtosecond-assisted phacoemulsification and arcuate keratotomy between March 2013 and August 2013 were analyzed.
Preoperative and postoperative examinations included slit-lamp biomicroscopy, dilated funduscopy, and keratometry using partial coherence interferometry (IOLMaster; Carl Zeiss Meditec Inc, Dublin, California, USA) and autorefraction using Nidek OPD-Scan III (Nidek Technologies, Gamagori, Japan). Patients were given a combination of topical phenylephrine and tropicamide before the procedure. A single reference point at the temporal limbus was marked with the patient sitting upright. Three additional points at the 4, 8, and 12 o’clock position at the limbus were also marked to help identify the cornea center. Corneal topography (Nidek OPD-Scan III; Nidek Technologies) was performed immediately before the surgery. The angle between the steep axis and the reference point measured at the cornea center was recorded to determine the location of the steep axis for arcuate keratotomy.
A combined phacoemulsification and arcuate keratotomy was performed using the VICTUS (Bausch & Lomb Inc, Dornach, Germany) femtosecond laser platform, which has real-time intraoperative spectral-domain optical coherence tomography guidance. A single arcuate keratotomy was paired with the main corneal incision for phacoemulsification at 180 degrees, which was located in close proximity to the limbus, preferably at the superior or temporal cornea. The depth of the keratotomy was set at 450 μm, and the side-cut angle was set at 90 degrees with an arc diameter of 8.0 mm. The angular arc length of the incision ranged from 30 to 80 degrees according to the magnitude of preoperative keratometric astigmatism. The surgeon’s nomogram was modified from the Wallace Limbal Relaxing Incision Nomogram ( Table 1 ). The incisions were created with a programmed energy setting of 1.7 μJ and spot/line separation of 6 × 4 μm. The main corneal incision had a tri-planar configuration with a width of 3.0 mm and length of approximately 1.8–.0 mm. Two 1.5-mm-wide peripheral incisions with a tri-planar configuration were fashioned 60 degrees apart from the main corneal incision. The paracentesis sites were selected to augment the main incision for a larger astigmatic effect. An example of the programmed corneal incisions is shown in Figure 1 .
| Intended Astigmatic Correction (Diopters) | Arc Length at 8 mm Optical Zone (Degrees) |
|---|---|
| 1.00 | 30 |
| 1.25 | 40 |
| 1.50 | 50 |
| 2.00 | 70 |
| 2.50 | 80 |
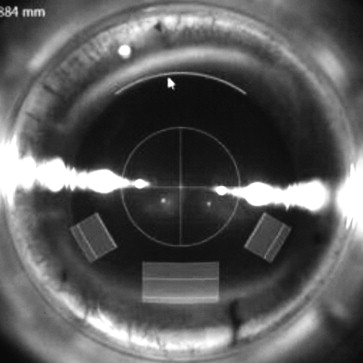
The same experienced surgeon (G.C.) performed surgery under topical anesthesia. A proprietary Victus suction ring was applied and the vacuum switched on. The suction ring was then filled with a few drops of balanced salt solution and a curved docking cone was docked into the suction ring. The femtosecond laser was used to create anterior capsulotomy, lens fragmentation, corneal incision, and astigmatic keratotomy in this sequence. After these steps were completed, the docking cone and suction ring were removed. Phacoemulsification was then performed in the sterile field. The arcuate keratotomy incisions were not opened after the procedure. Postoperatively, all patients were treated with a combination of topical dexamethasone and tobramycin for 1 month. Follow-up data at 2 months were analyzed.
The keratometric astigmatism was analyzed using vector analysis to evaluate the effective change in astigmatism, with consideration of the change in the astigmatic axis, measuring 3 vectors and relationships among them: the target-induced astigmatism vector (TIA), defined as the astigmatic change that the surgery was intended to induce; the surgically induced astigmatism vector (SIA), defined as the astigmatic change that the surgery actually induced; and the difference vector (DV), defined as the induced astigmatic change that would enable the initial surgery to achieve its intended target or the postoperative astigmatism. The Student t test was used to compare continuous variables (PASW software version 18.0; SPSS/IBM Inc, Chicago, Illinois, USA). P < .05 was considered statistically significant.
Results
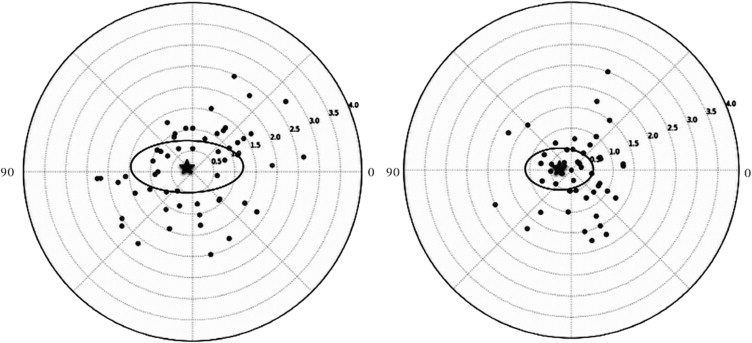
Overall, 54 eyes of 54 patients (18 male and 36 female) were included in this study. The mean age of the patients was 68.8 ± 11.4 years at the time of surgery. The mean preoperative astigmatism or TIA was 1.33 ± 0.57 diopters (D), which was reduced significantly to 0.87 ± 0.56 D postoperatively ( P < .001). The postoperative spherical equivalent was −1.16 ± 1.12 D. Refractive astigmatism dropped from −1.23 ± 0.83 D to −0.81 ± 0.85 D ( P < .002). No significant difference was found between the change in keratometric (0.46 ± 0.59 D) and refractive (0.38 ± 0.86 D) astigmatism postoperatively ( P = .573). Figure 2 demonstrates the double-angle plot of preoperative and postoperative keratometric astigmatism. The centroid is the mean astigmatism vector, and is close to zero in both plots. The ellipse signifies 1 standard deviation around the centroid. The postoperative ellipse is smaller, demonstrating a reduction in astigmatism after surgery. The keratometric vector analysis is shown in Table 2 . The arithmetic mean SIA magnitude (1.20 ± 0.68 D) was less than the arithmetic mean TIA. The magnitude of error (arithmetic difference between SIA and TIA) was −0.13 ± 0.68 D, demonstrating slight undercorrection. This was also represented as the correction index (CI) (0.86 ± 0.52), calculated as the ratio of SIA to TIA. The CI value indicates an overcorrection if it is more than 1 or an undercorrection if it is less than 1. A scatterplot of SIA vs TIA is shown in Figure 3 . There was undercorrection when TIA is more than 1 D and overcorrection when TIA is less than 1 D.
| TIA | |
| Arithmetic mean ± SD (diopters) | 1.33 ± 0.57 |
| Range a (diopters) | 1.31–1.35 |
| SIA | |
| Arithmetic mean ± SD (diopters) | 1.20 ± 0.68 |
| Range a (diopters) | 1.17–1.22 |
| DV | |
| Arithmetic mean ± SD (diopters) | 0.87 ± 0.56 |
| Range a (diopters) | 0.85–0.89 |
| ME | |
| Arithmetic mean ± SD (diopters) | −0.13 ± 0.68 |
| Range a (diopters) | −0.16 to −0.11 |
| AE | |
| Arithmetic mean ± SD (degrees) | 0.10 ± 26.10 |
| Range a (degrees) | −0.90 to 1.00 |
| Absolute AE | |
| Arithmetic mean ± SD (degrees) | 17.50 ± 19.20 |
| Range a (degrees) | 16.80–18.20 |
| CI | |
| Geometric mean ± SD | 0.86 ± 0.52 |
| Range a | 0.84 to 0.88 |
| CA | |
| Geometric mean ± SD | 1.16 ± 1.11 |
| Range a | 1.12 to 1.20 |
| FI | |
| Geometric mean ± SD | 0.53 ± 0.76 |
| Range a | 0.50 to 0.56 |
| IOS | |
| Geometric mean ± SD | 0.62 ± 0.45 |
| Range a | 0.60 to 0.64 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


