Purpose
To define the role of increased fundus autofluorescence (FAF), a surrogate for lipofuscin content, as a risk factor for progression of geographic atrophy (GA).
Design
Prospective natural history cohort study, the GAIN (Characterization of geographic atrophy progression in patients with age-related macular degeneration).
Methods
setting : Single-center study conducted in Barcelona, Spain. patients : After screening of 211 patients, 109 eyes of 82 patients with GA secondary to age-related macular degeneration and a minimum follow-up of 6 months were included. observation procedures : Lipofuscin content was classified independently by 2 masked observers according to FAF patterns described previously. Bivariate, stratified, and multivariable analyses were used to explore the associations between GA growth and independent variables. Mediation analysis was used to evaluate the contribution of FAF patterns to GA progression. main outcome : Progression of GA in mm 2 /year as measured with FAF.
Results
Median follow-up was 18 months (range, 6–42). Median GA growth was 1.61 mm 2 /year. FAF, baseline area of atrophy, and time of follow-up were independently associated with GA progression ( P < .004). FAF patterns and baseline area of atrophy were strongly associated ( P < .0001), suggesting potential confounding. Mediation analysis suggested that most of the effect of FAF patterns on GA growth was actually caused by baseline area of atrophy.
Conclusions
FAF patterns, baseline area of atrophy, and time of follow-up were associated with GA progression. However, FAF patterns seem to be a consequence (not a cause) of enlarging atrophy and their effect on GA progression seems mostly driven by baseline area of atrophy.
Geographic atrophy (GA), the advanced form of dry age-related macular degeneration (AMD), is characterized by enlarging areas of retinal pigment epithelium (RPE) atrophy. Regions affected by GA are devoid of RPE, photoreceptors, and choriocapillaris, and thus they cause absolute scotoma. It has been reported that antiangiogenic therapy in patients with neovascular AMD may foster GA growth and that most of these patients develop macular atrophy over the long term, which is one of the main reasons for lack of visual improvement in this population. These issues emphasize the need to understand disease pathogenesis to develop rational therapies.
Lipofuscin accumulation within the RPE is a well-known feature of aging and, possibly, of GA. It is the result of incomplete phagocytosis of photoreceptor outer segments by the RPE. As such, lipofuscin is formed by a mixture of metabolites from the visual cycle, such as N-retinyledene-N-retinylethanolamine (A2E). Some compounds of lipofuscin are autofluorescent, which contributes to identify its topographic distribution in vivo using fundus autofluorescence (FAF).
In 2001, Holz and associates reported that GA progression took place solely in areas that showed increased FAF. Some years later, the “Fundus autofluorescence in age-related macular degeneration” (FAM), a prospective, multicenter, natural history study of GA, used FAF to classify patients with GA according to the distribution of increased autofluorescence around atrophy. The FAM reported 10 distinct categories (patterns or phenotypes), which showed a marked association with GA growth on bivariate analysis. These patterns became an important risk factor in GA progression and pointed toward a causal relationship between lipofuscin accumulation and GA growth, supporting the development of visual cycle modulators, a therapeutic strategy aimed at slowing down the build-up of lipofuscin bisretinoids within the RPE.
Nonetheless, the role of lipofuscin in GA pathogenesis remains controversial. Most studies suggest that it has deleterious effects on RPE that may lead to cell death and GA progression, while other clinical and basic research findings do not support this hypothesis. Indeed, a visual cycle modulator did not meet the primary efficacy endpoint in a phase II clinical trial.
We conducted a study to evaluate factors associated with progression of prevalent GA. In particular, we tested whether increased FAF (as a surrogate for increased lipofuscin) was the primary predictor of GA progression. The results may contribute to clarify the role of this compound on GA pathogenesis.
Subjects and Methods
Study Design
The “Characterization of geographic atrophy progression in patients with age-related macular degeneration” (GAIN) study (NCT01694095) was a prospective, natural history study that aimed to identify risk factors associated with GA progression. It was conducted at the Institut de la Màcula i de la Retina (Centro Médico Teknon) in Barcelona, Spain. Recruitment began on December 21, 2009 and finished on December 31, 2012. Data collection was completed on August 9, 2013. The study followed the tenets of the Declaration of Helsinki and was prospectively approved by the Ethics Committee of the Centro Médico Teknon, and all patients signed an informed consent after explanation of the nature and possible consequences of the study.
Inclusion and Exclusion Criteria
The GAIN study included patients of either sex, 50 years or older, with GA secondary to AMD followed for at least 6 months. GA was defined as unifocal or multifocal areas of RPE atrophy on a 35-degree fundus photograph (TRC 50DX IA, IMAGEnet; Topcon Corporation, Tokyo, Japan); at least 1 of the atrophic lesions had to be larger than 0.5 disc area (1.27 mm 2 ). Both eyes of each patient were eligible for the study. Eyes were excluded in the following situations: RPE atrophy secondary to other causes (eg, macular dystrophy, high myopia); a history of wet AMD or any other macular disease thought to interfere with interpretation of FAF images in the study eye; contact between GA and peripapillary atrophy precluding the measurement of the lesion; lesion borders extending beyond the 30-degree field used for FAF imaging; a history of laser in the macula, intravitreal injection or intraocular surgery (aside from phacoemulsification) in the study eye; or poor image quality precluding the assignment of patient’s eyes to a particular FAF pattern.
Procedures
All patients underwent a complete ophthalmic examination that included best-corrected visual acuity (BCVA), intraocular pressure, fundus biomicroscopy, and imaging (fundus photography, infrared, FAF, and spectral-domain optical coherence tomography [SD OCT]) after pupil dilation with 1.0% tropicamide and 10% phenylephrine. FAF imaging (λ = 480 nm, approximate emission 500–700 nm) was acquired with Spectralis HRA+OCT (Heidelberg Engineering, Heidelberg, Germany). High-resolution (1536 × 1536 pixels), 30 × 30-degree-field-of-view images centered on the fovea with a minimum averaging of 10 frames were captured. Fluorescein angiography was performed when required according to medical criteria. Refraction and imaging were performed by certified optometrists and technicians.
All areas of atrophy in each image were measured with FAF by a single observer (M.B.) using the Region Finder software, version 2.4.3.0 (Heidelberg Engineering). The observer was masked to previous measurements. To evaluate intraobserver agreement, a random sample of 20% of all images was drawn and the area was reevaluated at least 1 month apart from the first measure.
FAF patterns were independently determined by 2 experienced observers (F.T. and M.B.) using the 10-item classification (see below). In case of disagreement, a senior observer (J.M.) arbitrated. A consensus was reached in all cases.
Main Outcome Variable
The main outcome was growth of GA (mm 2 /year). For bivariate analysis, it was measured between the last and the first visit, divided by the time between them. For multivariable models, GA growth was expressed as the change in the area of atrophy from one visit to the next, including the time between visits as an independent variable.
Main Predictor and Other Independent Variables
The predictor of primary interest in this study was FAF pattern. As originally described in the FAM study, there are 10 different patterns: none (no increased FAF in the junctional zone of atrophy), focal (single or individual small spots of FAF at the junction), banded (an almost continuous ring of atrophy), patchy (laminar, homogeneous FAF around GA), reticular (linear structures with predominantly radial orientation), branching (ramified FAF), fine granular (FG; grainy, heterogeneous), fine granular with peripheral punctate spots (FGPPS; grainy at the junction and spot-like, well-defined FAF elsewhere), trickling (grayish atrophy, with high FAF at the margins that seeps toward the periphery), and undetermined (FAF features different from those previously described). This detailed classification was the one used by the observers to classify each eye. For analytic purposes, more simple classifications, described below, were used.
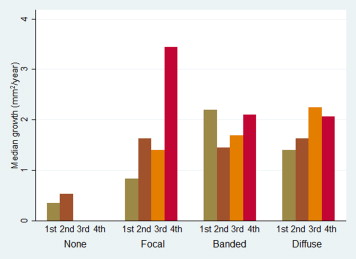
For bivariate analysis, we used a classification with 5 categories: none, focal, banded, diffuse, and undetermined. The diffuse category gathers patterns characterized by the presence of FAF beyond the borders of atrophy (the reticular, branching, FG, FGPPS, and trickling patterns). No patient was classified into the patchy pattern ( Figure 1 ).
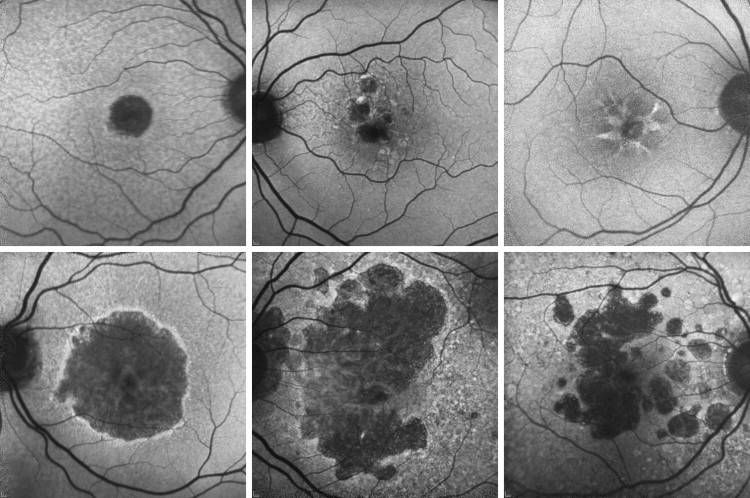
For multivariable analysis, the classification was further simplified into just 2 categories: low and high FAF. The “low FAF” category includes patterns characterized by a small area of retina with hyperautofluorescence (patterns none and focal), while the “high FAF” assembles those with larger retinal areas with elevated FAF (patterns banded and diffuse). The category “undetermined” was excluded, given the small number of patients involved.
Other independent variables were age, sex, race, baseline area of atrophy, time of follow-up, bilateral GA, eye, BCVA, lens status, central retinal thickness (CRT), high blood pressure, smoking status, body mass index (BMI), familiar history of AMD, antioxidant use, concomitant ocular diseases, and atrophy description (their definition is available as Supplemental Table 1 ; available at AJO.com ).
Statistical Analysis
Some data were not captured at some visits. Those values were assumed to be missing at random, and multiple imputation using iterative chained equations was used to infer them.
Univariate and bivariate analyses were used to describe the characteristics of participants and the association between variables, respectively. Bivariate analyses were conducted using Mann-Whitney or Kruskal-Wallis tests for comparison of continuous variables between groups, Spearman correlation coefficient (rho) when both variables were continuous, and Fisher exact test for categorical variables. A stratified analysis of baseline area of atrophy on FAF patterns in relation to GA growth was conducted to inspect interactions.
A multivariable analysis using a hierarchal mixed-effects regression model was fitted to evaluate the independent contribution of each variable on GA progression. The model accounted for the increased correlation between the 2 eyes of a given patient. We also determined the relative contribution of FAF patterns and baseline area of atrophy on GA growth under the current hypotheses using mediation analysis, with hierarchical mixed-effects regression models. Finally, Bland-Altman plots were used to evaluate intraobserver agreement of GA size measures.
Results were analyzed using Stata IC/13.1 (Stata Corp, College Station, Texas, USA). A 2-tailed P value ≤.05 was considered statistically significant. Since all analyses were prespecified and type I errors were preferred over type II errors, no correction for multiple comparisons was made.
Results
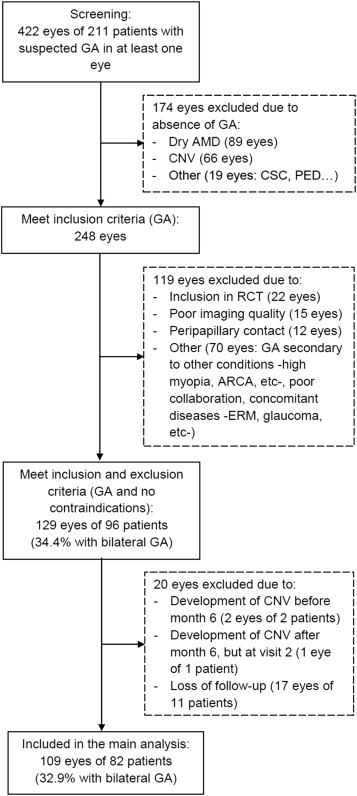
Figure 2 shows the flow diagram of all screened patients. Longitudinal data were available from 109 eyes of 82 patients, after exclusion of 11.5% of patients who were lost to follow-up. A positive familial history of AMD was the only statistically significant difference between patients remaining in the study and those lost to follow-up (30.5% vs 0%, respectively, P = .03). The main characteristics of patients included in the study are shown in Table 1 (a more detailed description is provided in Supplemental Table 2 , available at AJO.com ).
| Characteristic | Value |
|---|---|
| Age (y) | 80 (74.5–84.5) |
| Female, % | 66.7 |
| Follow-up (mo) | 18 (10–31) |
| Baseline area of atrophy (mm 2 ) | 6.85 (3.14–11.88) |
| Fundus autofluorescence pattern, % | |
| None | 4.7 |
| Focal | 27.1 |
| Banded | 25.6 |
| Fine granular | 17.8 |
| Branching | 14.0 |
| Trickling | 3.1 |
| Reticular | 3.1 |
| Fine granular with peripheral punctate spots | 1.6 |
| Undetermined | 3.1 |
| Best-corrected visual acuity (letters) | 71 (59–79) |
| Pseudophakia, % | 48.8 |
| Antioxidant use, % | 72.9 |
| Current smokers, % | 9.4 |
| Configuration of atrophy, % | |
| Unifocal foveal | 22.5 |
| Multifocal foveal | 22.5 |
| Unifocal extrafoveal | 3.9 |
| Multifocal extrafoveal | 20.2 |
| C-shaped | 4.7 |
| C-shaped multifocal | 17.1 |
| Annular | 9.3 |
Median follow-up time was 18 months (range, 6–42 months). The median (mean) growth was 1.62 (1.76) mm 2 /year (interquartile range, 1.01–2.44; range, 0.11–5.55). The only variable with missing values was central retinal thickness (CRT; 21/483, 4.3%). These values were imputed as previously described.
The median (mean) BCVA change from baseline to the last follow-up visit was a loss of 7 (9) letters, from 20/40+ to 20/50−; 17.4% of eyes (19/109) lost ≥3 lines and 18.4% (20/109) had a BCVA ≤20/200 at the last visit. Ten eyes developed neovascular AMD (10/129, 7.8%—including those lost to follow-up) and were excluded from further analyses.
Bivariate Analysis
The relationship between GA growth and FAF patterns (using the classification with 5 categories) was statistically significant ( P < .0013) and mirrored the distribution found in the FAM study ( Figure 3 ).
In other bivariate analyses, GA growth was also correlated with baseline area of atrophy (rho = 0.38, P < .0001), time of follow-up (rho = −0.19, P = .05) and age (rho = 0.23, P = .02). Extrafoveal location of atrophy ( P = .002) and pseudophakia ( P = .03) were also associated with faster growth. No statistically significant relationship was found with other variables ( P ≥ .08).
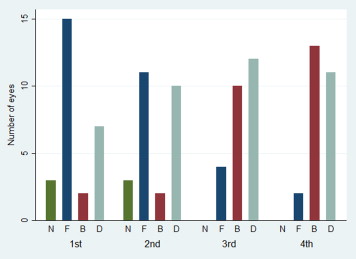
The association of FAF patterns with other independent variables was explored to assess the possibility that confounding by those variables may explain the relationship between FAF patterns and GA growth. FAF patterns were associated with baseline area of atrophy ( P = .0001). Patterns characterized by larger areas with hyperautofluorescence (banded and diffuse) had larger baseline area of atrophy than other patterns ( P = .0001; Table 2 ). In fact, the number of eyes with low FAF (patterns none or focal) decreased progressively with increasing baseline area of atrophy, while the opposite was true for patterns with high FAF (banded or diffuse; Figure 4 ).
| Fundus Autofluorescence Patterns (n) | Baseline Area of Atrophy in mm 2 , Median (Mean) | Interquartile Range | P Value |
|---|---|---|---|
| None (6) | 2.67 (2.67) | 1.84–3.74 | .0001 |
| Focal (32) | 3.25 (4.14) | 1.59–4.78 | |
| Banded (27) | 11.7 (12.58) | 8.34–17.24 | |
| Diffuse (40) | 8.26 (10.42) | 4.57–13.10 | |
| Undetermined (4) | 2.58 (2.39) | 1.04–3.75 |
FAF patterns were also associated with location of atrophy (foveal vs extrafoveal; P = .03) and familial history of AMD ( P = .05), but not with other variables ( P ≥ .06). The complete set of bivariate analyses is provided in Supplemental Table 3 (for the relationship with the main outcome, GA growth) and Supplemental Table 4 (for the relationship between FAF patterns and other independent variables) (Supplemental Material available at AJO.com ).
Stratified Analysis
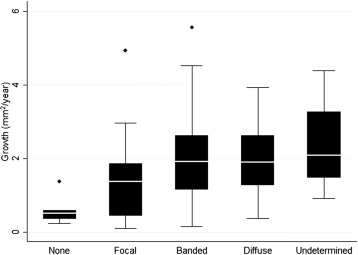
Within FAF patterns, median GA growth increased with increasing baseline area of atrophy size for all pattern groups ( Figure 5 ). Eyes with the pattern none had the smallest progression, while eyes with other FAF patterns experienced a relatively similar distribution of median rate of growth within quartiles of baseline area of atrophy.
Multivariable Analysis
The results of multivariable analysis are shown in Table 3 . Baseline area of atrophy (β = 1.02), FAF patterns (β = 0.54), and follow-up time (β = 1.62) were independently associated with GA growth. On the other hand, age, pseudophakia, extrafoveal location of atrophy, and CRT, which were associated with GA growth on bivariate analyses, were no longer significant.
| Variables | Coefficients (β) | SE | 95% CI | P Value |
|---|---|---|---|---|
| FAF patterns | 0.54 | 0.20 | 0.15–0.94 | .007 |
| Time (y) | 1.62 | 0.13 | 1.36–1.89 | <.0001 |
| Time 2 (y 2 ) | −0.03 | 0.05 | −0.12 to 0.06 | .52 |
| BAA | 1.02 | 0.01 | 0.99–1.05 | <.0001 |
| Familial history | −0.14 | 0.20 | −0.53 to 0.26 | .51 |
| Sex | 0.30 | 0.20 | −0.08 to 0.69 | .12 |
| Foveal atrophy | 0.10 | 0.15 | −0.19 to 0.38 | .51 |
| Hypertension | 0.10 | 0.19 | −0.28 to 0.47 | .61 |
| Smoking | −0.04 | 0.35 | −0.72 to 0.64 | .90 |
| Antioxidant use | 0.09 | 0.21 | −0.33 to 0.50 | .68 |
| Lens status | 0.13 | 0.21 | −0.28 to 0.54 | .53 |
| Concomitant disease | −0.03 | 0.19 | −0.39 to 0.34 | .89 |
| Constant | −0.86 | 0.34 | −1.54 to −0.19 | .01 |
a Model includes age, body mass index, best-corrected visual acuity, and central retinal thickness as random effects (not shown).
Figure 6 shows the hypothetical relationship between FAF patterns, baseline area of atrophy, and GA growth that served as the basis for analysis of mediation. The purpose of mediation analysis was to understand the relationship between FAF patterns and GA growth, considering baseline area of atrophy, and to determine the relative contribution of each variable to the enlargement of GA. The analysis consists of 2 parts. First, all associations among the variables must be statistically significant ( Table 4 ): Step 1: FAF patterns were associated with GA growth without inclusion of baseline area of atrophy in the model ( P = .002); Step 2: Baseline area of atrophy was associated with GA growth independently of FAF patterns ( P < .001); and Step 3: FAF patterns were associated with baseline area of atrophy ( P < .001). This suggests that the relationship shown in Figure 6 is a possible scenario. Then, the percent change in the coefficient of FAF patterns when baseline area of atrophy was included in the model was evaluated; it decreased from 3.30 (in Step 1 on mediation analysis, representing its total effect) to 0.54 (in the multivariable analysis in Table 3 , representing its direct effect only). This implies strong confounding of baseline area of atrophy on the relationship between FAF patterns and GA growth. A large percentage of the effect of FAF patterns on GA growth (83.6%, [(3.30 − 0.54)/3.30] × 100) was presumably mediated (caused) through baseline area of atrophy.


Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


