Purpose
To compare the effects of trimethoprim-sulfamethoxazole vs placebo in reducing the risk of recurrences of Toxoplasma gondii retinochoroiditis.
Design
Single-center, prospective randomized double-masked clinical trial.
Methods
A total of 95 patients from Campinas, Brazil, with active recurrent Toxoplasma gondii retinochoroiditis were included. The initially active toxoplasmosis lesions were successfully treated in all cases using trimethoprim-sulfamethoxazole (800 mg/160 mg) twice daily for 45 days. Subsequently, 5 patients dropped out of the study. The remaining patients were randomized to Group 1 (trimethoprim/sulfamethoxazole tablet every 2 days) or Group 2 (identical placebo tablet every 2 days). Randomization was 1:1, was stratified by sex, and used block sizes of 4. The primary outcome was recurrent toxoplasmosis retinochoroiditis within 1 year, and the secondary outcome was a 1-year change in best-corrected visual acuity (BCVA) (ETDRS chart).
Results
The incidence of recurrent toxoplasmosis retinochoroiditis within 12 months was 0 of 46 (0%) and 6 of 47 (12.80%) in the trimethoprim-sulfamethoxazole and placebo groups, respectively ( P = .026). Visual acuity improvements in the 2 groups were similar. No treatment-limiting toxicity was observed.
Conclusions
Trimethoprim/sulfamethoxazole therapy resulted in a 100% reduction in the recurrence of Toxoplasma gondii retinochoroiditis over 1 year of treatment.
Toxoplasmosis is most likely the most common cause of posterior uveitis in immunocompetent individuals. This disease can affect all age groups and both sexes, and ocular involvement can occur without any significant evidence of systemic disease. During the chronic phase of toxoplasmic infections, short and usually self-limiting periods of parasite reactivation regularly occur and cause the typical presentation of acute recurrent toxoplasmic retinochoroiditis.
Visual acuity and the number of chorioretinal lesions are commonly used as measures of the long-term burden of ocular toxoplasmosis. Antibiotic treatment primarily aims to reduce the risk of permanent visual loss, recurrent retinochoroiditis, and the severity and duration of acute symptoms. There is uncertainty about the effectiveness of antibiotic treatment.
There is no universally accepted treatment approach for ocular toxoplasmosis, and one of the current options is the sulfonamide group of drugs, which is effective against tachyzoite forms but which has little effect on bradyzoites, resulting in latent foci of the disease that are responsible for its recurrence.
Despite the fact that newly developed drugs decrease the number of tissue cysts in experimental and animal studies, prevention of recurrent attacks of ocular toxoplasmosis has not yet been achieved.
In an open-label trial, Silveira and associates suggested that intermittent treatment with trimethoprim-sulfamethoxazole could reduce the rate of recurrent toxoplasmic retinochoroiditis. The patients were randomized to treatment with 1 tablet of trimethoprim-sulfamethoxazole every 3 days or to observation without treatment and were followed monthly for up to 20 consecutive months for clinical signs of disease recurrence. Recurrences developed in 4 treated patients (6.60%) and in 15 controls (23.80%). The weaknesses of their study were the facts that control subjects did not receive placebo and that subjects were not examined in a masked fashion; these factors may affect the ability to interpret drug side effects, and may also affect follow-up and introduce bias during the examinations.
The purpose of this double-masked trial was to compare the effects of trimethoprim-sulfamethoxazole vs placebo in reducing the risk of recurrences of Toxoplasma gondii retinochoroiditis.
Methods
This study was a single-center, prospective randomized double-masked clinical trial. Ethics committee (Institutional Review Board) approval was obtained, and all participants gave informed consent (National Bioethics Commission of Brazil identifier: 0613.0.146.000-10). The trial was registered in October 2011 and began in November 2011 (Influence of Trimethoprim-Sulfamethoxazole for the Recurrence of Ocular Toxoplasmosis; clinicaltrials.gov identifier: NCT01449877; http://clinicaltrials.gov/show/NCT01449877 ).
A total of 100 patients were recruited from a public hospital in the city of Campinas, in Brazil. All patients were followed up for 12 months. Inclusion criteria were active recurrent Toxoplasma gondii retinochoroiditis (a new focus of necrotizing retinochoroiditis with active inflammation either adjacent to or remote from preexisting retinochoroidal scars, with positive IgG for toxoplasmosis). Patients who were under 18 years of age, immunocompromised (eg, acquired immunodeficiency syndrome), or undergoing immunosuppressive treatments, or who had concomitant retinochoroiditis from other causes (eg, tuberculosis), were excluded.
All patients were treated for active toxoplasmic retinochoroiditis with 1 tablet of trimethoprim-sulfamethoxazole (800 mg/160 mg) 2 times daily for 45 days, and all lesions were healed after this treatment. Subsequently, 5 patients dropped out of the study. The remaining patients were randomly assigned to Group 1 (1 trimethoprim-sulfamethoxazole tablet every 2 days) or Group 2 (1 identical placebo tablet containing starch without any active ingredients every 2 days). Randomization was 1:1 and was stratified by sex, and block sizes of 4 were used. One nurse generated the random allocation sequence, and another nurse enrolled and assigned the participants in the interventions in a masked fashion.
The trial was sponsored by Fundação de Amparo a Pesquisa do Estado de São Paulo, protocol 2010/15980-2. Trimethoprim/sulfamethoxazole tablets cost US$0.31 each and placebo tablets cost US$0.15.
Data were collected by means of a medical history form completed by the physician at the time of the first medical examination. Best-corrected visual acuity (BCVA) with Early Treatment Diabetic Retinopathy Study (ETDRS) charts, biomicroscopy, tonometry, indirect ophthalmoscopy, recurrence of Toxoplasma gondii retinochoroiditis, and medical events were recorded monthly on a standardized form by a member of the medical staff in a masked fashion.
The primary outcome was recurrent toxoplasmosis retinochoroiditis within 12 months ( Figure 1 ), and the secondary outcome was a change in BCVA (ETDRS chart) over 12 months.
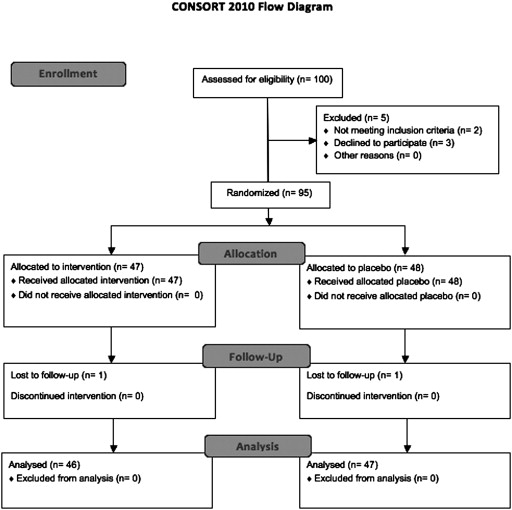
Sample size calculations based on presumed recurrence rates of 0% and 15% in the treatment and placebo groups, respectively, indicated that a sample size of 35 in each group would be significant for detecting a difference of this magnitude with a power of 80% and type 1 error probability of 5%. However, to account for losses in follow-ups and based on feedback obtained after the initiation of the study, 95 patients were enrolled.
Descriptive statistics were calculated. Continuous data were expressed as the mean values, standard deviation (SD), and ranges. Between-group differences of continuous variables were compared using the Mann-Whitney U test, and categorical variables were compared using χ 2 test or Fisher exact test when appropriate. Analyses were conducted using SPSS version 21 (IBM Corporation, Armonk, New York, USA) and SOFA version 1.3.5 (Statistics Open For All; AGPL3 Free Software, accessed at http://www.sofastatistics.com/downloads.php ). P values are 2-tailed. Statistical significance was considered at the .05 level.
Results
Between August 24, 2011, and August 28, 2012, 95 patients were enrolled at a public hospital in Campinas, Brazil, and were randomized to trimethoprim/sulfamethoxazole (47 patients) or placebo (48 patients) therapy. A total of 93 patients completed the 12 months of follow-up (1 lost to follow-up in each group; Figure 1 ). The mean age was 33 (SD 13) years (median 33), and 53 patients were female. The age and sex distributions were similar between the groups ( Table 1 ).
| Trimethoprim-Sulfamethoxazole (n = 47) | Placebo (n = 48) | P | |
|---|---|---|---|
| Age (y): mean (SD), median | 34 (13), 33 | 33 (14), 33 | .78 a |
| Male/female ratio | 20/27 | 22/26 | .75 b |
| Baseline BCVA | 58 (SD 20) letters (20/80) | 52 (SD 18) letters (20/100) | .17 a |
| 12-month BCVA | 79 (SD 16) letters (20/25) | 74 (SD 18) letters (20/32) | .17 a |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


