Topical Diagnosis of Optic Chiasmal and Retrochiasmal Lesions
Topical Diagnosis of Optic Chiasmal Disorders
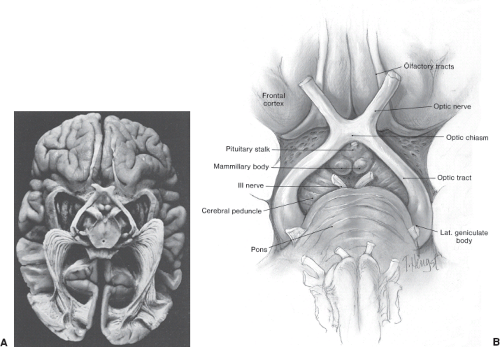
The optic chiasm is one of the most important structures in neuro-ophthalmologic diagnosis (Figs. 12.1 and 12.2). The arrangement of visual fibers in the chiasm accounts for characteristic defects in the visual fields caused by such diverse processes as compression, inflammation, demyelination, ischemia, and infiltration. In addition, damage to neurologic and vascular structures adjacent to the chiasm produces typical additional symptoms.
Visual Field Defects
Although there are many variations in the visual field defects caused by damage to the optic chiasm, the essential feature is a bilateral temporal (bitemporal) defect, the hallmark of damage to fibers that cross within the chiasm. The bitemporal defects may be superior, inferior, or complete, and they may be peripheral, central, or both and may be associated with additional defects related to associated damage to one or both optic nerves anteriorly and/or the optic tracts posteriorly.
From Damage to the Anterior Angle of the Optic Chiasm
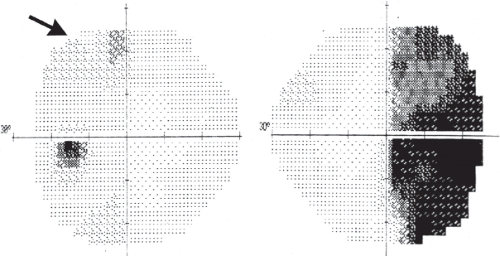
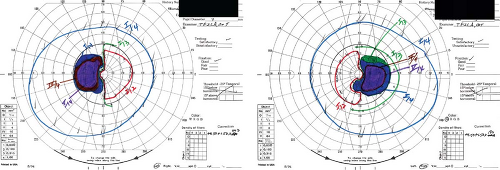
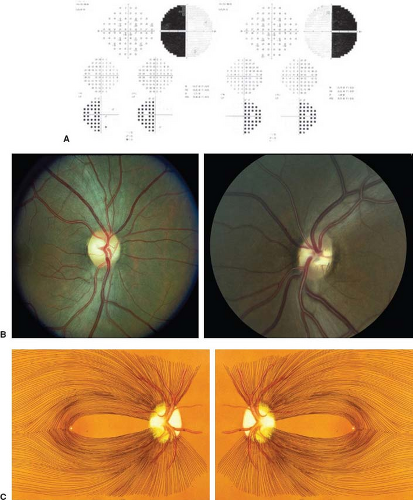
Where the optic nerve joins the chiasm, at the anterior angle of the chiasm, its particular fiber anatomy provides an opportunity for anatomic diagnosis. The crossed and uncrossed fibers are separated at this level but are quite compact, and a small lesion affecting either the crossed or the uncrossed fibers may produce a unilateral hemianopic defect. Such a defect is called a “junctional scotoma.” In such cases, it is not uncommon to find an asymptomatic scotoma in the upper temporal field of the opposite eye (Fig. 12.3). This scotoma results from damage to ventral fibers originating from ganglion cells located inferior and nasal to the fovea that, upon reaching the distal end of the optic nerve, have been thought to loop anteriorly about 1 to 2 mm into the contralateral optic nerve (Wilbrand’s knee, see Chapter 2). There is now some evidence that Wilbrand’s knee is not a normal anatomic structure but rather an artifact that develops when there is atrophy of the optic nerve. Although this may be true, Wilbrand’s knee clearly exists from a clinical standpoint; that is, a patient with evidence of an optic neuropathy in one eye and an isolated superior temporal defect in the visual field of the opposite eye has a lesion in the region of the anterior optic chiasm.
From Damage to the Body of the Optic Chiasm
Lesions that damage the body of the optic chiasm characteristically produce a bitemporal defect that may be quadrantic or hemianopic and that may be peripheral, central, or both, with or without so-called “splitting of the macula” (Fig. 12.4). In most cases, visual acuity is normal. When the lesion compresses the chiasm from below, such as occurs with a pituitary adenoma, the field defects are typically superior; however, they often are unequal in the two eyes. In some patients, a suprasellar, infrachiasmatic lesion may elevate the chiasm and optic nerves such that the superior aspect of the nerves is compressed by the dural (falciform) folds extending from the intracranial end of the optic canals, thus producing inferior field defects. Thus, patients with bitemporal field defects that are denser below may not necessarily have a suprasellar, suprachiasmatic process. Having made this point, it certainly is true that suprasellar, suprachiasmatic compressive lesions—such as tuberculum sellae meningiomas, craniopharyngiomas, aneurysms, and dolichoectatic anterior cerebral arteries—tend to damage the superior fibers of the chiasm. The defects in the visual fields in such cases are still bitemporal, but are denser inferiorly or are entirely in the inferior fields rather
than the superior fields of both eyes. Papilledema, which is quite unusual in patients with suprasellar, infrachiasmal lesions, is somewhat more common in suprachiasmal lesions because such lesions can extend into and occlude the third ventricle.
than the superior fields of both eyes. Papilledema, which is quite unusual in patients with suprasellar, infrachiasmal lesions, is somewhat more common in suprachiasmal lesions because such lesions can extend into and occlude the third ventricle.
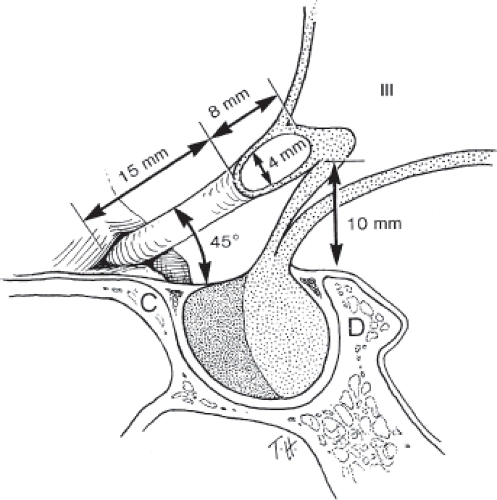 Figure 12.2 Artist’s drawing of the relationships of the optic nerves and optic chiasm to the sellar structures and third ventricle (III). C, anterior clinoid; D, dorsum sellae. |
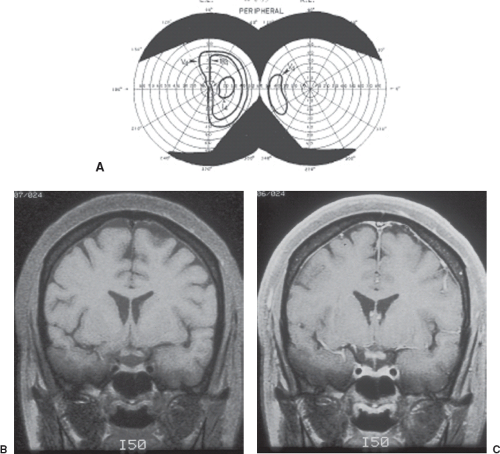
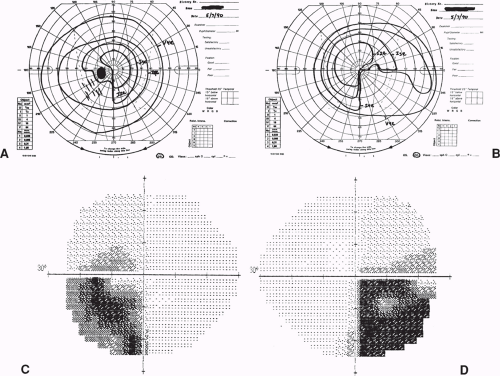
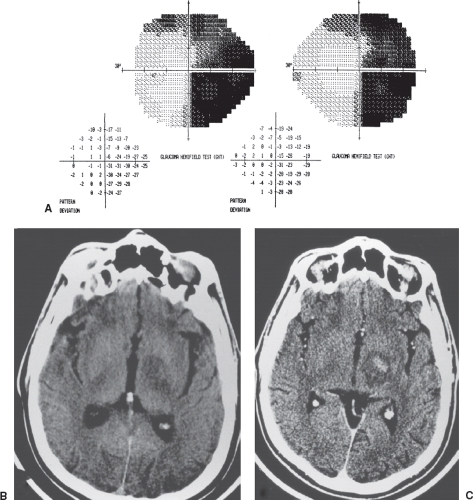
Infiltrating tumors, such as gliomas, germ cell tumors, and lymphomas, as well as inflammatory and demyelinating lesions that affect the body of the optic chiasm (e.g., sarcoid, multiple sclerosis), may produce typical bitemporal field defects (Fig. 12.5). In such cases, the field defects may be denser superiorly, inferiorly, or of equal density above and below the horizontal midline.
When trauma damages the optic chiasm, the most common field defect is a complete bitemporal hemianopia. The reason is that in such cases, the chiasm may actually be transected (Fig. 12.6).
From Damage to the Posterior Angle of the Optic Chiasm
Lesions that damage the posterior aspect of the optic chiasm produce characteristic defects in the
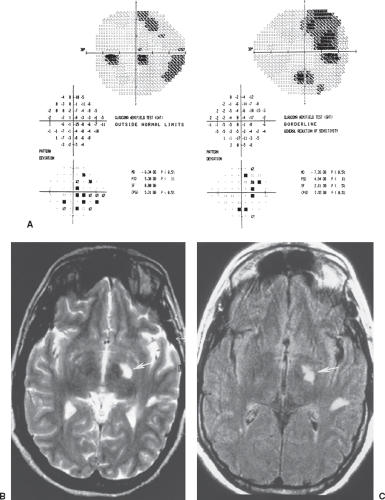
visual fields: bitemporal hemianopic scotomas. These defects may be superior, inferior, or central in location (Figs. 12.7 to 12.9). When central, such defects may be mistaken for cecocentral scotomas and attributed to a toxic, metabolic, or even hereditary process rather than to a tumor; however, true bitemporal hemianopic scotomas almost always are associated with normal visual acuity and color perception, whereas cecocentral scotomas invariably are associated with reduced visual acuity and acquired dyschromatopsia.
visual fields: bitemporal hemianopic scotomas. These defects may be superior, inferior, or central in location (Figs. 12.7 to 12.9). When central, such defects may be mistaken for cecocentral scotomas and attributed to a toxic, metabolic, or even hereditary process rather than to a tumor; however, true bitemporal hemianopic scotomas almost always are associated with normal visual acuity and color perception, whereas cecocentral scotomas invariably are associated with reduced visual acuity and acquired dyschromatopsia.
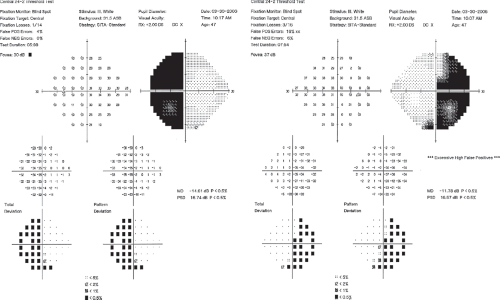 Figure 12.4 Body of the optic chiasm syndrome. Automated static perimetry demonstrates a complete bitemporal hemianopia. The patient’s visual acuity was 20/20 OU. |
Lesions that damage the posterior aspect of the optic chiasm also may damage one of the optic tracts, thus producing a homonymous field defect that is
combined with whatever field defect has occurred from damage to the optic chiasm.
combined with whatever field defect has occurred from damage to the optic chiasm.
Visual Field Defects Caused by Lesions That Damage the Optic Chiasm After Initially Damaging the Optic Nerve or Optic Tract
If there is extension of a lesion from the optic nerve or the optic tract to the optic chiasm, the blind eye usually is on the side of the lesion. If, for example, a patient with a blind right eye exhibits a defect in the temporal field of the left eye, the lesion obviously is on the right. Similarly, if there has been a left homonymous hemianopia from a right optic tract lesion and if there is extension of the lesion to affect the optic chiasm, blindness develops in the right eye, or if not blindness, an extensive field defect. Conversely, if a lesion of the optic chiasm that has produced a bitemporal hemianopia extends to the right optic nerve, it will eventually produce blindness or near-blindness of the right eye. Similarly, if a chiasmal lesion extends into the right optic tract, there is again blindness or near-blindness of the right eye. In other words, when there is extension
of a lesion from an optic nerve or optic tract to the optic chiasm, the blind (or near-blind) eye is always on the side of the original lesion, and when there is extension of a lesion from the optic chiasm to the optic nerve or to the optic tract, the blind (or near-blind) eye is always on the side of the extension of the lesion.
of a lesion from an optic nerve or optic tract to the optic chiasm, the blind (or near-blind) eye is always on the side of the original lesion, and when there is extension of a lesion from the optic chiasm to the optic nerve or to the optic tract, the blind (or near-blind) eye is always on the side of the extension of the lesion.
Neuro-Ophthalmologic Signs and Symptoms Associated with the Optic Chiasmal Syndrome
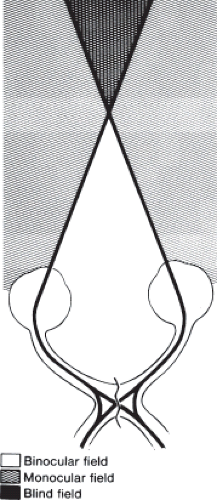
The most frequent symptoms of patients with lesions that damage the optic chiasm are progressive loss of central acuity and dimming of the visual field, particularly in its temporal portion. In addition, bitemporal field defects, whether complete or scotomatous, may produce two other types of visual symptoms. One type consists of a disturbance of depth perception. Patients with this symptom have difficulty with near tasks, such as threading needles, sewing, and using precision tools. In such patients, convergence results in crossing of the two blind temporal hemifields. This produces a completely blind triangular area of field with its apex at fixation (Fig. 12.10). The image of an object posterior to fixation falls on blind nasal retinas and thus disappears.
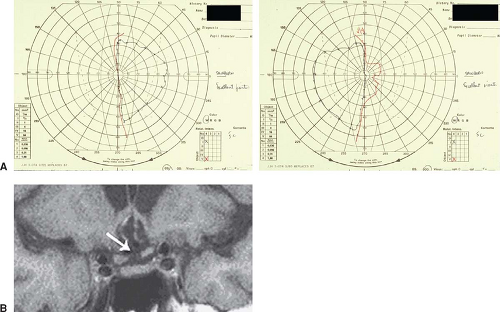
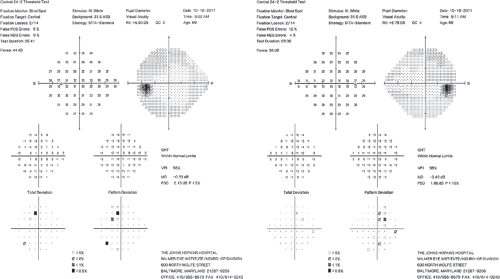 Figure 12.9 Posterior optic chiasmal syndrome. Subtle, primarily superior bitemporal hemianopic scotomas in a patient with a pituitary adenoma. |
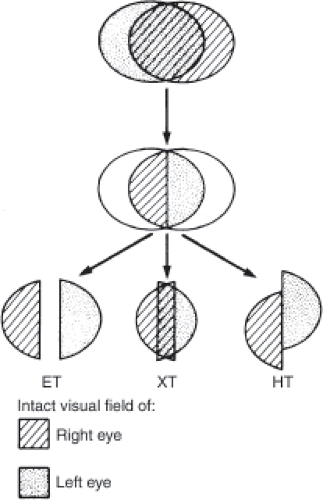
Patients with bitemporal field defects also may experience diplopia or difficulty reading caused by a horizontal or vertical deviation of images unassociated with evidence of an ocular motor nerve paresis, the hemifield slide phenomenon. Such patients have difficulty reading because of doubling or loss of printed letters or words. The problems encountered by these patients result from loss of the normal partial overlap of the temporal field of one eye and the nasal field of the contralateral eye. This overlap normally permits fusion of images and helps stabilize ocular alignment in patients with vertical or horizontal phorias. Because their remaining visual fields represent only the temporal projection from each eye, patients with a bitemporal hemianopia do not have a physiologic linkage between the two remaining hemifields. In such patients, there is loss of fusion, and a pre-existing phoria becomes a tropia, causing diplopia or other sensory difficulties (Fig. 12.11).
Patients with chiasmal syndromes may or may not have ophthalmoscopically apparent nerve fiber layer or optic disc atrophy when first examined; however, when atrophy is present in patients with a complete bitemporal hemianopia unassociated with evidence of a unilateral or bilateral optic neuropathy, the pattern of both retinal nerve fiber layer and optic nerve atrophy is quite specific. In such cases, degeneration occurs in fibers from peripheral and macular ganglion cells located nasal to the fovea. Axons from peripheral ganglion cells located nasal to the disc (and, thus, nasal to the fovea) attain the disc directly, entering its nasal aspect. Fibers from macular ganglion cells nasal to the fovea but temporal to the disc (i.e., about half the fibers that comprise the papillomacular bundle) also attain the optic disc directly but enter its temporal aspect. Finally, fibers from peripheral ganglion cells located nasal to the fovea but temporal to the optic disc—which make up a small portion of the superior and inferior arcuate bundles along with a larger number of fibers from peripheral temporal ganglion cells—enter the superior and inferior aspects of the disc. Thus, when there is atrophy of nasal fibers (nasal to both the fovea and disc or nasal to the fovea but temporal to the disc), the normal striations of the nerve fiber layer are lost both nasal and, to a large extent, temporal to the disc in the papillomacular region, and the optic disc shows corresponding atrophy at its nasal and temporal regions with relative sparing of the superior and inferior portions where the majority of spared temporal fibers (subserving nasal field) enter (Fig. 12.12). The optic atrophy occupies a more or less horizontal band across the disc, wider nasally than temporally, so-called “band” or “bow-tie” atrophy.
It is perhaps clinically more useful when atrophy is absent in patients with chiasmal (or optic nerve) compression than when it is present. Although patients with significant retinal nerve fiber bundle defects and optic atrophy can have impressive return of both acuity and field when successful decompression is obtained, patients with normal-appearing fundi should have near complete or complete return of visual function. Thus, it is crucial that decompression be carried out as soon as possible in these patients.
Although the presence or absence of optic disc pallor is predictive of visual recovery in patients with chiasmal compression, the thickness of the peripapillary retinal nerve fiber layer (PRNFL) as assessed by optical coherence tomography (OCT) is even more so. The normal average thickness of the PRNFL is about 100 μm. Patients with absolute optic atrophy (i.e., no light perception), whether glaucomatous or nonglaucomatous, have a PRNFL average thickness of 35 to 40 μm. Thus, there is a range of about 65 μm between normal and complete loss. It has been shown that patients with less than 50% reduction in the average thickness of the PRNFL; that is, ≥75 μm, have a high likelihood of regaining both acuity and field after successful decompression. Again, however, patients with more than a 50% reduction in average PRNFL thickness can still experience visual improvement following successful decompression.
Most patients with lesions that affect the optic chiasm have no disturbances of ocular alignment or motility except for the hemifield slide phenomenon (see above); however, some lesions in the parasellar/suprasellar region can damage the ocular motor nerves, causing double vision in addition to visual loss. When the ocular motor nerves are damaged within the cavernous sinus, there also may be pain, evidence of a trigeminal sensory neuropathy, or both, in the territory of the first and/or second divisions of the trigeminal nerve. The third division of the nerve, which does not pass through the cavernous sinus, is not affected by such lesions, and a motor neuropathy thus is never present unless the lesion extends posteriorly. The oculosympathetic fibers also may be damaged by a lesion in the suprasellar or parasellar region, resulting in a postganglionic Horner syndrome characterized by ipsilateral ptosis and a small reactive pupil (see Chapter 15). Horner syndrome often is associated with an ipsilateral abducens nerve palsy because the postganglionic oculosympathetic fibers briefly join the abducens nerve within the cavernous sinus. When both the oculomotor nerve and oculosympathetic fibers are affected, the clinical picture is one of an oculomotor nerve paresis with a small pupil that is normally reactive if the paresis is pupil-sparing or poorly or nonreactive if the paresis involves the pupillomotor fibers. Single or multiple ocular motor nerve pareses in a patient with a bitemporal field defect suggests a process extrinsic to the chiasm rather than an infiltrative intrinsic lesion.
The unusual phenomenon of seesaw nystagmus may occur in patients with tumors of the diencephalon and chiasmal regions (Video 12.1). This condition is characterized by synchronous alternating elevation and intorsion of one eye and depression and extorsion of the opposite eye. The cause of seesaw nystagmus is unknown, but it may be related to damage to the interstitial nucleus of Cajal or adjacent structures by the tumor.
Lesions causing a chiasmal syndrome may arise from or extend to the hypothalamus. Patients with this presentation may develop diabetes insipidus and hypothalamic hypopituitarism. Such patients may have excessive fluid intake that may not be obvious unless they are specifically asked how many glasses of water, etc. they drink a day. In addition, prepubertal children may suffer from both growth retardation and delayed sexual development, and young children and infants may have severe failure to thrive (Russell syndrome) and present with severe emaciation.
Topical Diagnosis of Retrochiasmal Visual Pathway Disorders
Unilateral lesions of the visual sensory pathway beyond the optic chiasm—the optic tract, lateral geniculate body (LGB), optic radiation, or striate cortex (see Fig. 12.13A)—produce homonymous visual field defects without loss of visual acuity. When such defects are complete, they do not allow topical diagnosis. In such instances, the clinician must rely on other ocular findings, other symptoms and signs of neurologic disease (or their absence!), and/or on neuroimaging to determine the area and extent of damage as well as the etiology of the lesion.
Homonymous defects in the visual fields characteristically develop slowly when they are caused by compression and rapidly when they are caused by hemorrhage, ischemia, or inflammation. Regardless of the cause, however, such defects include complete homonymous quadrantanopias and hemianopias, incomplete homonymous quadrantanopias and hemianopias with varying degrees of congruity, and homonymous paracentral scotomas.
Almost 90% of patients with isolated homonymous hemianopia have occipital lobe lesions, usually caused by vascular disease in the territory of the posterior cerebral arteries, whereas patients with lesions of the postchiasmal visual pathway anterior to the occipital cortex may or may not have neurologic symptoms and/or signs (see below). In some cases, a relative afferent pupillary defect (RAPD) on the side
of the hemianopia in the absence of evidence of an optic neuropathy or other cause for the defect helps localize the location of the lesion to the contralateral optic tract, whereas asymmetric responses to optokinetic (OKN) stimulation with the poorer responses observed when targets are moved toward the side opposite the hemianopia (i.e., toward the side of the lesion) generally indicate a lesion of the optic radiations in the parietal lobe (see below). In all cases, however, neuroimaging, preferably MRI, should be used to verify or determine the location and etiology of a homonymous hemianopia.
of the hemianopia in the absence of evidence of an optic neuropathy or other cause for the defect helps localize the location of the lesion to the contralateral optic tract, whereas asymmetric responses to optokinetic (OKN) stimulation with the poorer responses observed when targets are moved toward the side opposite the hemianopia (i.e., toward the side of the lesion) generally indicate a lesion of the optic radiations in the parietal lobe (see below). In all cases, however, neuroimaging, preferably MRI, should be used to verify or determine the location and etiology of a homonymous hemianopia.
Topical Diagnosis of Optic Tract Lesions
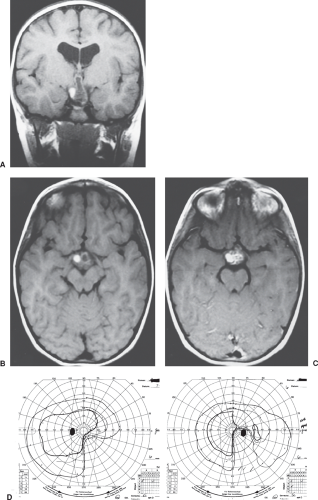
Although lesions affecting the optic tracts are infrequent, they are of great importance because they are located in the first region beyond the optic chiasm where lesions produce a homonymous visual field defect (Fig. 12.13). The causes are varied and include tumors, vascular processes, demyelinating disease, and trauma (Figs. 12.14 and 12.15).
As noted above, patients with optic tract lesions often have a specific finding that permits the determination of the location of the lesion on clinical grounds
alone. All patients with a complete or near-complete homonymous hemianopia caused by an isolated optic tract lesion (i.e., no evidence of an associated optic neuropathy) have an RAPD in the eye contralateral to the side of the lesion (i.e., the eye on the side of the hemianopia and that has a temporal field defect). This occurs because (1) the temporal visual field is considerably larger than the nasal field. Therefore, there are more crossing nasal fibers than noncrossing temporal fibers; (2) the pupillomotor fibers within the visual sensory pathway hemidecussate in the optic chiasm along with or as part of the visual axons; and (3) the pupillomotor fibers are present within the optic tract for most of its extent. Because the temporal visual field is 60% to 70% larger than the nasal field, there is a disparity with respect to light input from the two eyes to the mesencephalic pupillary center. A complete lesion of one optic tract thus preferentially reduces input from the contralateral eye, and the result is an RAPD unassociated with any evidence of an optic neuropathy or retinopathy.
alone. All patients with a complete or near-complete homonymous hemianopia caused by an isolated optic tract lesion (i.e., no evidence of an associated optic neuropathy) have an RAPD in the eye contralateral to the side of the lesion (i.e., the eye on the side of the hemianopia and that has a temporal field defect). This occurs because (1) the temporal visual field is considerably larger than the nasal field. Therefore, there are more crossing nasal fibers than noncrossing temporal fibers; (2) the pupillomotor fibers within the visual sensory pathway hemidecussate in the optic chiasm along with or as part of the visual axons; and (3) the pupillomotor fibers are present within the optic tract for most of its extent. Because the temporal visual field is 60% to 70% larger than the nasal field, there is a disparity with respect to light input from the two eyes to the mesencephalic pupillary center. A complete lesion of one optic tract thus preferentially reduces input from the contralateral eye, and the result is an RAPD unassociated with any evidence of an optic neuropathy or retinopathy.
Another pupillary phenomenon that sometimes can be elicited in patients with lesions of the optic tract that produce a complete or nearly complete homonymous hemianopia is pupillary hemiakinesia (aka hemianopic pupillary reaction or Wernicke pupil). Because the pupillary afferents are present in this section of the visual system, light that is projected onto the “blind” retinal elements (subserving the nasal visual field in the ipsilateral eye and the temporal visual field in the contralateral eye), results in either no pupillary reaction or a markedly reduced reaction. However, when light is projected onto the intact retinal elements (subserving the temporal visual field in the ipsilateral eye and the nasal visual field in the contralateral eye), a normal pupillary reaction is observed. This phenomenon is best observed with a bright, very focused beam of light, such as that from a slit lamp biomicroscope.
Optic tract lesions do not cause loss of visual acuity nor do they affect color vision unless they also damage the optic chiasm or the intracranial portions of one or both optic nerves. Thus, patients with lesions confined to an optic tract that produce a complete or near-complete homonymous hemianopia have normal visual acuity and color vision, and they have no sensation of reduced light brightness in the eye despite an RAPD. On the other hand, when lesions of the optic tract also damage the ipsilateral optic nerve or chiasm, reduced visual acuity, dyschromatopsia, and an RAPD are present on the side of the lesion.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree