Purpose
To evaluate the long-term effect of a single-piece hydrophobic acrylic intraocular lens (IOL), AcrySof SN60AT (Alcon Laboratories, Fort Worth, Texas, USA), on the development of posterior capsule opacification (PCO) 5 years postoperatively.
Design
Prospective, observational, consecutive, case series.
Methods
setting : Iladevi Cataract and IOL Research Center, Ahmedabad, India. study population : Three hundred and ninety eyes with uncomplicated age-related cataract were included. Patients with diabetes mellitus, glaucoma, high myopia, pseudoexfoliation, traumatic cataract, subluxated cataract, previous ocular surgeries, and allergy to dilating drops were excluded. intervention procedures : Digital retroillumination photographic documentation was performed and analyzed for PCO using Evaluation of Posterior Capsule Opacification software. The scores and areas were calculated. main outcome measures : PCO development and the influence of the anterior capsule cover (total on and part on) on the IOL optic was studied within the capsulorrhexis margin and the central 3.0 mm optic area.
Results
There was a significant increase in PCO up to 3 years. No significant change in PCO was observed between 3 and 5 years within the capsulorrhexis margin and central 3.0 mm optic area. In the total on group, within the capsulorrhexis margin, significantly lower scores and areas were observed when compared with part-on scores and areas.
Conclusion
The increase in PCO up to 3 years was significant. Stabilization in PCO was observed between 3 and 5 years with no difference at 5 years. There was a low incidence of PCO in eyes with total anterior capsule cover over the IOL optic.
Although contemporary cataract surgery has made tremendous strides in technology, posterior capsular opacification (PCO) is still the most frequent long-term complication affecting visual outcomes after surgery. Neodymium–yttrium-aluminum-garnet (Nd:YAG) laser capsulotomy remains a frequent secondary intervention attributable to clinically significant PCO. Factors affecting the formation of PCO include patient characteristics, lens design, design of the edge of the lens, lens material, and surgical technique used. A complete 360-degree cover of the anterior capsulorrhexis over the intraocular lens (IOL) optic also influences PCO development.
Clinically, the introduction of the AcrySof IOL (Alcon Laboratories, Fort Worth, Texas, USA) in the early 1990s resulted in markedly reduced PCO rates. This triggered a discussion on whether it was the hydrophobic acrylic material or the sharp-edged lens design that acted as a key factor in reducing PCO. It remains unclear as to how far the modification of the haptic design and angulation of the single-piece AcrySof IOL will aid in PCO prevention in a prospective long-term observational study with objective PCO evaluation. The development of PCO is indisputably a dynamic process and a follow-up over a long-term duration is correlated with a higher incidence of PCO. However, the question as to whether PCO further increases, becomes stable, or regresses with the progress of time has not yet been answered. We decided to observe the degree of PCO development after implantation of a single-piece SN60AT hydrophobic acrylic IOL at 5 years after cataract surgery.
Subjects and Methods
This prospective, observational study comprised 390 eyes of 390 consecutive patients who underwent phacoemulsification from June 2005 to June 2006 at the Iladevi Cataract and IOL Research Center. The Institutional Review Board of Iladevi Cataract & IOL Research Center approved the study and care was taken to ensure that it adhered to the tenets of the Declaration of Helsinki. Informed consent was obtained from all the patients participating in the study. The study was registered with the website maintained by the National Institutes of Health, http://www.clinicaltrials.gov , identifier ID: NCT01820065 . Patients with uncomplicated age-related cataract, who were otherwise healthy, were included in the study. The exclusion criteria were as follows: patients with a history of diabetes mellitus, glaucoma, high myopia (axial length >27.0 mm), pseudoexfoliation, traumatic cataract, subluxated cataract, previous ocular surgeries, and allergy to dilating drops. All the patients underwent a thorough slit-lamp examination including intraocular pressure assessment. The fundus was examined using direct and indirect ophthalmoscopy. A single surgeon (A.R.V.) performed all the surgeries using the Infiniti Vision System (Alcon Laboratories). A single-piece hydrophobic acrylic (model SN60AT; Alcon Laboratories) IOL was implanted in the capsular bag. This SN60AT IOL has an optic diameter of 6.0 mm and an overall length of 13.0 mm. The height of the square edge of the optic was 0.20 mm and the height of the square edge of the haptics was 0.43 mm.
Follow-up Examination and Image Acquisition
The patients were asked to return for postoperative follow-up visits at 1 month and 1, 3, and 5 years. Digital retroillumination photographic documentation was performed at all the follow-up visits, after maximal pupil mydriasis, at a fixed illumination and magnification. We used a digital camera (Nikon, Tokyo, Japan) mounted on a slit lamp (Haag-Streit, Switzerland) with an external light and flash light source, which provided coaxial illumination from the flash pack through a fiber optic cable attached to the camera. Thereafter, at every follow-up visit, photographic documentation of the posterior capsule was performed on a widely dilated pupil using retroillumination. The images of the posterior capsules of each patient taken at every follow-up visit were saved in a separate folder. These images provided information about the onset and progression of PCO. The eye was considered to have total cover if the capsulorrhexis rim completely overlapped the anterior surface of the IOL optic for 12 clock hours. If the overlap was between 1 and 9 clock hours, the eye was considered to have partial cover.
Posterior Capsule Opacification Image Interpretation and Analysis
All the digital images were analyzed for PCO using the Evaluation of Posterior Capsule Opacification 2000 program (a software program developed by Manfred Tetz and Christophe Nimsgern, Universitats-Augenklinik Charite, Berlin, Germany). Posterior capsule opacification was evaluated for the entire IOL optic (within the capsulorrhexis margin) and for the central 3 mm optic zone. The boundaries and each opaque area of the posterior capsule were drawn on the stored images using a computer mouse so that the fraction of the opaque area could be calculated using the Evaluation of Posterior Capsule Opacification software. The Evaluation of Posterior Capsule Opacification program is a morphologic assessment of PCO, in which the geographic extent and density of backscatter on retroilluminated images determine the overall score. The density of opacification was clinically graded as 0 (none), 1 (minimal), 2 (mild), 3 (moderate), or 4 (severe). Individual PCO values for each image were calculated by multiplying the density of opacification by the fraction of the capsular area involved behind the IOL optics (within the capsulorrhexis margin) and the central 3 mm optic zone. The value was interpreted in terms of Evaluation of Posterior Capsule Opacification score and area. Evaluation of Posterior Capsule Opacification score (PCO density × area) was expressed on a scale of 0–4. Evaluation of Posterior Capsule Opacification area (mm 2 ) was rated between 0% and 100%.
Observations
The primary observations included an assessment of the overall development of PCO at different time points. We also assessed the impact of the anterior capsule relationship on the IOL optic at different time points: from 1 month to 1 year, from 1 to 3 years, and from 3 to 5 years. All these observations were made within the capsulorrhexis margin and also within the central 3 mm area of the optic. Nd:YAG capsulotomy rates were also assessed.
Sample Size Calculation and Statistical Analysis
Assuming a difference of 4% between 3 and 5 years, 330 eyes were necessary to carry out a paired comparison with 80% power. After adding 20% to this number to factor in the expected dropout rate, we decided to recruit 396 eyes for the study. This was the required sample size for carrying out a paired comparison at different time points. The data were analyzed as paired data. However, as the data were not normally distributed, as confirmed by the Shapiro-Wilk test, nonparametric tests were used to check for differences. The Mann-Whitney U test and Freidman test were applied to compare differences in the development of PCO at different time points and also to assess the influence of the anterior capsule relationship on PCO.
Results
The mean age of the 213 male and 177 female patients (total 390 patients) was 58.3 ± 7.4 years with a mean follow-up of 59.6 ± 2.3 months. The mean axial length was 23.8 ± 1.5 mm.
Mean Evaluation of Posterior Capsule Opacification (Evaluation of Posterior Capsule Opacification Score and Area) Within the Capsulorrhexis Margin and the Central 3.0 mm Optic Area
The PCO (Evaluation of Posterior Capsule Opacification score and area) within the capsulorrhexis margin and the central 3.0 mm optic area at 1 month and 1, 3, and 5 years postoperatively are given in Table 1 ( Figure 1 ). The PCO (Evaluation of Posterior Capsule Opacification score and area) differ significantly within the capsulorrhexis margin ( P < .001) and within the central 3.0 mm optic area ( P < .001) when compared at 1 month and 1, 3, and 5 years, respectively ( Table 1 ).
| Within the Capsulorrhexis Margin | ||||||||
|---|---|---|---|---|---|---|---|---|
| Evaluation of Posterior Capsule Opacification Score | Evaluation of Posterior Capsule Opacification Area | |||||||
| 1 Month | 1 Year | 3 Years | 5 Years | 1 Month | 1 Year | 3 Years | 5 Years | |
| N (eyes) | 390 | 371 | 367 | 380 | 390 | 371 | 367 | 380 |
| 0.01 ± 0.03 | 0.06 ± 0.16 | 0.19 ± 0.43 | 0.20 ± 0.38 | 0.01 ± 0.03 | 0.06 ± 0.14 | 0.13 ± 0.22 | 0.13 ± 0.22 | |
| P < .001 a , b | P < .001 a , b | |||||||
| Central 3 mm Optic | ||||||||
|---|---|---|---|---|---|---|---|---|
| 1 Month | 1 Year | 3 Years | 5 Years | 1 Month | 1 Year | 3 Years | 5 Years | |
| N (eyes) | 390 | 371 | 367 | 380 | 390 | 371 | 367 | 380 |
| 0.00 ± 0.00 | 0.01 ± 0.07 | 0.06 ± 0.23 | 0.05 ± 0.21 | 0.00 ± 0.00 | 0.01 ± 0.07 | 0.05 ± 0.16 | 0.04 ± 0.14 | |
| P < .001 a , b | P < .001 a , b | |||||||
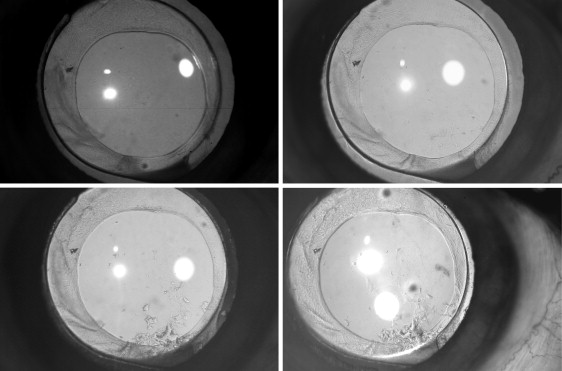
When we observed the paired comparisons within the capsulorrhexis margin, a significant difference was observed in PCO (Evaluation of Posterior Capsule Opacification score and area) between 1 month and 1 year ( P < .001) and between 1 and 3 years ( P < .001). However, the paired difference was not significant from 3 to 5 years. Similarly, within the central 3 mm optic, the paired comparisons showed significant differences in PCO (Evaluation of Posterior Capsule Opacification score and area) between 1 month and 1 year ( P < .001) and between 1 and 3 years ( P < .001). However, the paired difference was not significant from 3 to 5 years ( Table 2 ).
| Within the Capsulorrhexis Margin | |||||
|---|---|---|---|---|---|
| Evaluation of Posterior Capsule Opacification Score | |||||
| 1 Month | 0.01 ± 0.03 | 1 Year | 0.06 ± 0.16 | 3 Years | 0.19 ± 0.43 |
| 1 Year | 0.06 ± 0.16 | 3 Years | 0.19 ± 0.43 | 5 Years | 0.20 ± 0.38 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
| Evaluation of Posterior Capsule Opacification Area | |||||
| 1 Month | 0.01 ± 0.03 | 1 Year | 0.06 ± 0.14 | 3 Years | 0.13 ± 0.22 |
| 1 Year | 0.06 ± 0.14 | 3 Years | 0.13 ± 0.22 | 5 Years | 0.13 ± 0.22 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
| Central 3 mm Optic | |||||
|---|---|---|---|---|---|
| Evaluation of Posterior Capsule Opacification Score | |||||
| 1 Month | 0.00 ± 0.00 | 1 Year | 0.01 ± 0.07 | 3 Years | 0.06 ± 0.23 |
| 1 Year | 0.01 ± 0.07 | 3 Years | 0.06 ± 0.23 | 5 Years | 0.05 ± 0.21 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
| Evaluation of Posterior Capsule Opacification Area | |||||
| 1 Month | 0.00 ± 0.00 | 1 Year | 0.01 ± 0.07 | 3 Years | 0.05 ± 0.16 |
| 1 Year | 0.01 ± 0.07 | 3 Years | 0.05 ± 0.16 | 5 Years | 0.04 ± 0.14 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
Comparison of Evaluation of Posterior Capsule Opacification (Evaluation of Posterior Capsule Opacification Score and Area) With Anterior Capsule Relationship
Total on
In the total on group, there was a statistically significant increase in PCO (Evaluation of Posterior Capsule Opacification score and area) at every follow-up within the capsulorrhexis margin ( P < .001) and within the central 3.0 mm optic area up to 5 years ( P < .001) ( Table 3 ). Within the capsulorrhexis margin and central 3 mm area of the optic, PCO (Evaluation of Posterior Capsule Opacification score and area) significantly increased from 1 month to 1 year ( P < .001) and from 1 to 3 years ( P < .001). However, no significant difference was observed between 3 and 5 years ( Table 4 ).
| Total On Cover of the Anterior Capsule | ||||||||
|---|---|---|---|---|---|---|---|---|
| Within the Capsulorrhexis Margin | ||||||||
| Evaluation of Posterior Capsule Opacification Score | Evaluation of Posterior Capsule Opacification Area | |||||||
| 1 Month | 1 Year | 3 Years | 5 Years | 1 Month | 1 Year | 3 Years | 5 Years | |
| Eyes | 271 | 255 | 259 | 264 | 271 | 255 | 259 | 264 |
| 0.00 ± 0.01 | 0.04 ± 0.13 | 0.18 ± 0.45 | 0.17 ± 0.39 | 0.00 ± 0.01 | 0.04 ± 0.12 | 0.11 ± 0.22 | 0.11 ± 0.22 | |
| P < .001 a , b | P < .001 a , b | |||||||
| Central 3 mm Optic | ||||||||
|---|---|---|---|---|---|---|---|---|
| 1 Month | 1 Year | 3 Years | 5 Years | 1 Month | 1 Year | 3 Years | 5 Years | |
| Eyes | 271 | 255 | 259 | 264 | 271 | 255 | 259 | 264 |
| 0.00 ± 0.00 | 0.01 ± 0.07 | 0.06 ± 0.26 | 0.05 ± 0.23 | 0.00 ± 0.00 | 0.01 ± 0.07 | 0.05 ± 0.17 | 0.04 ± 0.14 | |
| P < .001 a , b | P < .001 a , b | |||||||
| Part On Cover of the Anterior Capsule | ||||||||
|---|---|---|---|---|---|---|---|---|
| Within the Capsulorrhexis Margin | ||||||||
| Evaluation of Posterior Capsule Opacification Score | Evaluation of Posterior Capsule Opacification Area | |||||||
| 1 Month | 1 Year | 3 Years | 5 Years | 1 Month | 1 Year | 3 Years | 5 Years | |
| Eyes | 119 | 116 | 108 | 114 | 119 | 116 | 108 | 114 |
| 0.02 ± 0.05 | 0.11 ± 0.20 | 0.23 ± 0.36 | 0.26 ± 0.38 | 0.02 ± 0.05 | 0.10 ± 0.17 | 0.17 ± 0.22 | 0.19 ± 0.23 | |
| P < .001 a , b | P < .001 a , b | |||||||
| Central 3 mm Optic | ||||||||
|---|---|---|---|---|---|---|---|---|
| 1 Month | 1 Year | 3 Years | 5 Years | 1 Month | 1 Year | 3 Years | 5 Years | |
| Eyes | 119 | 116 | 108 | 114 | 119 | 116 | 108 | 114 |
| 0.00 ± 0.00 | 0.01 ± 0.08 | 0.05 ± 0.15 | 0.06 ± 0.17 | 0.00 ± 0.00 | 0.01 ± 0.08 | 0.04 ± 0.13 | 0.05 ± 0.16 | |
| P < .001 a , b | P < .001 a , b | |||||||
| Total On Cover of the Anterior Capsule | |||||
|---|---|---|---|---|---|
| Within the Capsulorrhexis Margin | |||||
| Evaluation of Posterior Capsule Opacification Score | |||||
| 1 Month | 0.00 ± 0.01 | 1 Year | 0.04 ± 0.13 | 3 Years | 0.18 ± 0.45 |
| 1 Year | 0.04 ± 0.13 | 3 Years | 0.18 ± 0.45 | 5 Years | 0.17 ± 0.39 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
| Evaluation of Posterior Capsule Opacification Area | |||||
| 1 Month | 0.00 ± 0.01 | 1 Year | 0.04 ± 0.12 | 3 Years | 0.11 ± 0.22 |
| 1 Year | 0.04 ± 0.12 | 3 Years | 0.11 ± 0.22 | 5 Years | 0.11 ± 0.22 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
| Central 3 mm Optic | |||||
| Evaluation of Posterior Capsule Opacification Score | |||||
| 1 Month | 0.00 ± 0.00 | 1 Year | 0.01 ± 0.07 | 3 Years | 0.06 ± 0.26 |
| 1 Year | 0.01 ± 0.07 | 3 Years | 0.06 ± 0.26 | 5 Years | 0.05 ± 0.23 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
| Evaluation of Posterior Capsule Opacification Area | |||||
| 1 Month | 0.00 ± 0.00 | 1 Year | 0.01 ± 0.07 | 3 Years | 0.05 ± 0.17 |
| 1 Year | 0.01 ± 0.07 | 3 Years | 0.05 ± 0.17 | 5 Years | 0.04 ± 0.14 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
| Part On Cover of the Anterior Capsule | |||||
|---|---|---|---|---|---|
| Within the Capsulorrhexis Margin | |||||
| Evaluation of Posterior Capsule Opacification Score | |||||
| 1 Month | 0.02 ± 0.05 | 1 Year | 0.11 ± 0.20 | 3 Years | 0.23 ± 0.36 |
| 1 Year | 0.11 ± 0.20 | 3 Years | 0.23 ± 0.36 | 5 Years | 0.26 ± 0.38 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
| Evaluation of Posterior Capsule Opacification Area | |||||
| 1 Month | 0.02 ± 0.05 | 1 Year | 0.10 ± 0.17 | 3 Years | 0.17 ± 0.22 |
| 1 Year | 0.10 ± 0.17 | 3 Years | 0.17 ± 0.22 | 5 Years | 0.19 ± 0.23 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
| Central 3 mm Optic | |||||
| Evaluation of Posterior Capsule Opacification Score | |||||
| 1 Month | 0.00 ± 0.00 | 1 Year | 0.01 ± 0.08 | 3 Years | 0.05 ± 0.15 |
| 1 Year | 0.01 ± 0.08 | 3 Years | 0.05 ± 0.15 | 5 Years | 0.06 ± 0.17 |
| P value a | <.001 b | P value a | <0.001 b | P value a | NS |
| Evaluation of Posterior Capsule Opacification Area | |||||
| 1 Month | 0.00 ± 0.00 | 1 Year | 0.01 ± 0.08 | 3 Years | 0.04 ± 0.13 |
| 1 Year | 0.01 ± 0.08 | 3 Years | 0.04 ± 0.13 | 5 Years | 0.05 ± 0.16 |
| P value a | <.001 b | P value a | <.001 b | P value a | NS |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


