Purpose
To define the cytologic composition of the double-layered epithelial lining of dacryops (lacrimal duct cyst), improve histopathologic diagnosis, and better understand pathogenesis.
Design
Clinicopathologic retrospective study with immunohistochemical studies of 15 lesions compared with normal lacrimal gland.
Methods
Clinical data from 14 patients were reviewed and microscopy was performed with routine stains and immunohistochemical probes for epithelial membrane antigen (EMA), gross cystic disease fluid protein-15 (GCDFP-15), cytokeratin 7 (CK7), and smooth muscle actin (SMA).
Results
The major lacrimal gland was involved in 13 lesions; 2 lesions arose in an accessory gland of Krause. One case was bilateral; the average age of the patients was 50.7 years. Neither visual acuity nor motility was disturbed. No lesion was discovered to have recurred after excision. Microscopically, in all dacryops specimens goblet cells and luminal pseudoapocrine apical cytoplasmic projections were identified. Lacrimal acinar cells immunoreacted with GCDFP-15 and CK7, whereas the normal ducts and the epithelium of the dacryops lesions reacted diffusely only with CK7. SMA-positive myoepithelial cells were found in the acini but not in the normal ducts or dacryops epithelium.
Conclusions
Negative GCDFP-15 staining ruled out apocrine metaplasia in dacryops. Normal ducts and dacryops showed no immunohistochemical evidence for the presence of myoepithelial cells. Pathogenetic theories of dacryops that implicate a failure of ductular “neuromuscular” contractility must therefore be revised. A dysfunction of the rich neural plexus around the ductules may play a role in the development of dacryops in conjunction with periductular inflammation and induced scarring.
Dacryops is a cystic condition of the ducts of the lacrimal glands that is of uncertain causation and bilateral in fewer than 10% of cases. It has been estimated to be responsible for 4% to 9% of all epithelial conditions of the gland and 5% of orbital cysts of all kinds. The term is of Greek origin (dacryo = tear, lacrimal gland or sac; ops = eye) and was first introduced into ophthalmology by Schmidt in 1803. An earlier term that has lost currency today was “ranula of the lacrimal gland” because of a resemblance to the cystic dilation of the sublingual or submaxaillary ducts in the floor of the mouth, which assumes a dome-shaped and bluish coloration mimicking the belly of a frog. The biologic properties of the cells constituting the linings of the lacrimal cysts have not been definitively elucidated.
In this report, after carefully analyzing the clinicopathologic features of 15 examples of dacryops in 14 patients, immunohistochemical investigations were conducted to clarify issues revolving around the cytologic composition of the cysts’ linings, which are composed of a double layer of cells. The foremost objectives of this study were to find cogent answers to the following 4 questions: (1) What is the nature of the inner cells of the double layer constituting the dacryops epithelial lining; (2) Is the outer epithelial layer of cells in dacryops a myoepithelium; (3) What causes the obstruction of the lacrimal ductular system leading to the formation of dacryops; and (4) How can improved cytologic knowledge lead to sounder microscopic diagnosis and management, as well as more reliable separation of dacryops from other cystic conditions that arise in the eyelids and anterior orbit?
Methods
With prior Institutional Review Board approval to retrospectively review patient data, the routine and consultation files of the David G. Cogan Laboratory of Ophthalmic Pathology at the Massachusetts Eye and Ear Infirmary were evaluated for the period July 1, 2008 through March 30, 2012 for cases diagnosed as dacryops, lacrimal gland duct cyst, and conjunctival cyst. After careful review of hematoxylin–eosin-stained glass slides, 15 lesions in 14 patients were selected for inclusion in this study based on the presence of lobules of lacrimal gland parenchyma in intimate association with the fibrous walls of nonkeratinizing epithelial cysts. Paraffin-embedded tissue for preparation of 4- to 6-μm-thick sections was available in all cases for additional staining. Histochemical staining was performed with periodic acid–Schiff (PAS) (with and without diastase), Masson trichrome, mucicarmine, alcian blue, and Prussian blue for iron. Immunohistochemical probes were employed for identifying the biomarkers listed in Table 1 . With respect to the findings obtained with smooth muscle actin (SMA) for the identification of myoepithelial cells, these were rechecked by immunostaining 2 dacryops specimens with muscle-specific actin, calponin, glial fibrillary acidic protein (GFAP), S100, and smooth muscle myosin to prove the reliability and reproducibility of the SMA results. The immunoperoxidase method was used on the paraffin-embedded tissue sections with diaminobenzidine serving as the chromogen and hematoxylin as the counterstain. In addition to the cases of dacryops that included lobules of lacrimal gland tissue, 3 control specimens of normal lacrimal tissue were evaluated (2 represented negative biopsies for sarcoidosis and 1 was a prolapsed gland). Conjunctival epithelium present in 5 dacryops excisions and 3 other normal conjunctival specimens was examined with the same histochemical stains and immunohistochemical probes employed on the dacryops specimens. Clinical data pertaining to the 14 patients with dacryops were collected from hospital files, office records, or referring doctors, or, when necessary and possible, from direct telephonic interviews with the patients to confirm or complete the clinical demographics. Signs and symptoms, review of pertinent medical history and drug regimens, and the discovery of clinical outcomes were determined and tabulated. All available clinical photographs and, when possible, the actual computed tomographic scans (not merely the written reports) were retrieved and reviewed.
| Probe a | Cell Specificities | Staining Pattern |
|---|---|---|
| Gross cystic disease fluid protein-15 (GCDFP-15) | Breast tissue with apocrine metaplasia; apocrine glands (auditory ceruminous and eyelid Moll glands); apocrine tumors and cysts; secretory coils of cutaneous eccrine and apocrine glands; serous acinar cells of lacrimal, submandibular, sublingual, and minor salivary glands; absent in ducts of exocrine glands | Cytoplasmic (Golgi zone) and membranous |
| Alpha smooth muscle actin (α-SMA) b | Smooth muscle cells; myoepithelial cells; myofibroblasts; leiomyoma; leiomyosarcoma | Cytoplasmic |
| Cytokeratin-7 (CK7) | Epithelia of simple glands; secretory portions of apocrine and eccrine glands but not ducts; conjunctival epithelium and lacrimal acini and ducts | Cytoplasmic |
| Epithelial membrane antigen (EMA) | Glandular epithelial cells; luminal cuticles of lacrimal acini and ducts; apocrine and eccrine gland secretory coils and ducts | Membranous |
| Carcinoembryonic antigen (CEA) | Epithelial cells of endodermal derivation; luminal cuticles of apocrine and eccrine gland ducts and of lacrimal ducts | Membranous |
a All monoclonals supplied by Ventana Medical Systems, Oro Valley, Arizona, USA.
b To confirm the reliability of the SMA results, 2 lesions of dacryops were studied using other markers for myoepithelial cells: MSA (muscle-specific actin); calponin (actin-associated regulatory protein); desmin (intermediate filament in smooth and striated muscle); smooth muscle myosin (nonsarcomeric contractile protein in smooth muscle); S100 (for calcium flux regulator in Schwann, melanocytic and myoepithelial cells, among others); and glial fibrillary acidic protein (for glial cytoplasmic filaments that cross-react with smooth muscle actin).
Results
Clinical Findings
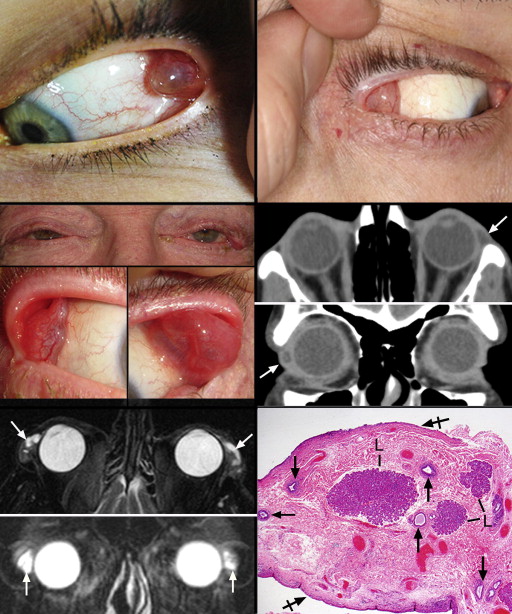
Table 2 summarizes the demographic data, clinical symptoms, and findings on ocular examination for the 14 patients included in this study. The left and right orbits were equally involved. There were 7 male and 7 female patients; the mean age was 50.7 years and the youngest patient was 4 years old. Thirteen patients had unilateral cysts ( Figure 1 , Top left and Top right). Eleven patients had cysts located in the palpebral lobe of the major lacrimal gland (7 right and 6 left); a twelfth had a deeper orbital lobe lesion. One palpebral lobe case was bilateral ( Figure 1 , Middle left, top and bottom panels). The last 2 patients had a cyst of a superior forniceal accessory Krause gland, 1 of which encroached on the anterior orbit from the nasal portion of the superior fornix. Visual acuity was unaffected in all patients and none had a motility disturbance, proptosis, or displacement of the globe. Three patients manifested superolateral eyelid fullness in the straight-ahead primary gaze position. All patients on the eversion of the upper eyelids had a visible cyst protruding into the interpalpebral zone from either the superolateral fornix or lateral canthus. Besides eyelid fullness, symptoms of mild pain and ocular irritation were noted in 7 cysts, whereas the rest of the lesions were completely asymptomatic, including that of 1 patient whose lesion was discovered during levator surgery. Imaging studies obtained on 3 patients in the axial plane disclosed that the cysts of the palpebral lobes resided anterior to the orbital rim ( Figure 1 , Middle right, top and bottom panels). Two cysts involved the anterior orbit; 1 of these was a superomedial cyst that caused some remodeling of the adjacent bone without osteolysis. In coronal sections the palpebral lesions were characteristically situated in the horizontal meridian and contoured to the globe medially and adopted somewhat of an overall triangular shape with the apex pointing laterally. In the bilateral case there appeared to be multiloculation. In this case T2-weighted magnetic resonance imaging with fat suppression demonstrated high signal intensity of the lesions, which appeared multiloculated and isointense with the vitreous ( Figure 1 , Bottom left, top and bottom panels). Simple excision was performed in all cases without any known recurrence and no patient complained postoperatively of dry eye symptoms.
| # | Age/Sex | Laterality | Duration of Symptoms | Symptoms/Complaints | Examination | Systemic Medications | Recurrence | Length of F/U (mo) |
|---|---|---|---|---|---|---|---|---|
| 1 | 64/F | Right | 3 mo | Chronic irritation; history of cosmetic eyelid surgery 5 years earlier on upper and lower eyelids | Conjunctival or lacrimal gland cysts and scar tissue | NA | No | 60 |
| 2 | 53/F | Right | 3 mo | Lump in upper eyelid | 10 × 12 mm large cyst involving superolateral orbit and conjunctiva | Dyazide, Synthroid, Zantac, atenolol | No | 48 |
| 3 | 39/M | Right | 1 wk | Recurrence of right upper eyelid swelling after removal of lesion by another ophthalmologist | Superotemporal conjunctival large cyst (6 mm across) just superior to lateral canthal tendon; little inflammation and no associated mass | NA | No | 36 |
| 4 | 64/M | Left | NA | Upper eyelid swelling and foreign body sensation | Lacrimal gland lesion pressing on the eyelid | Crestor, aspirin, dronedrarone, multivitamin lisinopril, metoprolol, vitamin D, zolpidem | No | 36 |
| 5 | 59/M | Right | 3 mo | Chronic irritation OD; cosmetic upper eyelid surgery 5 years earlier and on upper and lower eyelids 15 years earlier | Cystic lesion involving superotemporal conjunctiva and anterior orbit | NA | No | 24 |
| 6 | 68/M | Right | 1 y | Ptosis following cataract surgery | Palpebral lobe cyst found at levator surgery | Warfarin, vitamin D, multivitamin, Prinivil, aspirin | No | 15 |
| 7 | 63/M | Bilateral | 2-3 mo | Patient was unaware of lesions; primary ophthalmologist noted them | Lacrimal gland palpebral lobe: multiloculated cysts, nontender, no inflammation; everted laterally due to mass effect of cysts; floppy upper eyelid; blepharitis and dermatochalasis both eyes | Colchicine, allopurinol, aspirin, atenolol, Cialis, Lasix, insulin, Lipitor, Ativan, losartan, pentoxifylline, triamterene/hydrochlorothiazide, verapamil | No | 9 |
| 8 | 49/M | Left | 3 mo | Mass in superolateral conjunctival fornix causing eye irritation | Eversion of upper eyelid disclosed a cystic mass prolapsing within the lacrimal gland | Trazodone, lorazepam, and doxycycline | No | 5 |
| 9 | 58/F | Left | 1.5 mo | Enlarging “bump” in the lateral canthal region of left upper eyelid, changing from clear to red; visible as a lump in the upper eyelid; some foreign body sensation / pulling sensation | Cystic subconjunctival mass in lateral aspect of inner left upper lid and fullness of lateral upper lid; freely mobile; mildly injected; nontender | Niacin, aspirin, fish oil, and other vitamins | No | 2 |
| 10 | 45/F | Left | 2 mo | Swelling of the upper eyelid | Upper eyelid full laterally; eversion of the upper eyelid revealed a large clear cyst | None | No | 2 |
| 11 | 43/F | Right | 1 wk | Foreign body sensation and redness at lateral canthus | Superotemporal orbital/lacrimal gland cyst with area of focal injection over temporal bulbar conjunctiva; pain on right gaze of right eye | Motrin and Zantac | No | 1 |
| 12 | 35/F | Right | 3 d | “Preseptal cellulitis” with swelling and redness | Mild resistance to retropulsion of the right eye; CT showed an ovoid lesion in the superomedial orbit causing bone remodeling; appears unrelated to the preseptal cellulitis | Augmentin, Lexapro | No | 1 |
| 13 | 66/M | Left | 3-4 d | Foreign body sensation | Multiple left upper eyelid tarsal concretions and cysts causing irritation | Aspirin, simvastatin, Plavix, metoprolol | No | 1 |
| 14 | 4/M | Left | 6 mo | Lateral conjunctival lesion OS present for 6 months that became more irritating 1 month prior to presentation; mother reports frequent eye rubbing | 2-3 mm cystic lesion in lateral canthus | None | No | 0 |
Cytoarchitecture of the Normal Lacrimal Gland
The specimens of dacryops that included areas of undisturbed lacrimal gland units, and 5 nondiseased controls of orbital biopsies that contained lacrimal gland tissue in them, were studied with the same routine and immunohistochemical stains as the dacryops ( Table 1 ) to delineate the normal microanatomic structure of the gland. The lobules of acinar tissue, the intralobular and interlobular ducts, and the main excretory (final confluent) ducts were all specifically evaluated.
Thirteen of the 15 lacrimal gland specimens were all derived from the palpebral lobes. Multiple small lobules with adjacent excretory ducts in favorable sections were found beneath the nonkeratinizing conjunctival squamous epithelium, which harbored scattered goblet cells ( Figure 1 , Bottom right). In contrast to the secretory acinar cells in the lobules, the draining ducts had more conspicuous lumens that became progressively patulous as one proceeded from intralobular to interlobular ducts and finally to the main excretory ducts, which often adopted a spiraling configuration ( Figure 2 , Top left). The lumens of the interlobular and excretory ducts in all specimens focally exhibited apical cytoplasmic expansions or projections that did not appear to actually decapitate into the lumens as cytoplasmic debris (consequently, these can be referred to as pseudoapocrine snouts). Rather, the lumens possessed stringy mucoid or acellular eosinophilic secretory material ( Figure 2 , Top right) that was PAS-, alcian blue–, and mucicarmine-positive ( Figure 2 , Middle left, and inset). The adlumenal apical cytoplasmic projections of the duct structures were particularly intensely PAS-positive and resisted diastase pre-digestion ( Figure 2 , Top right, inset).
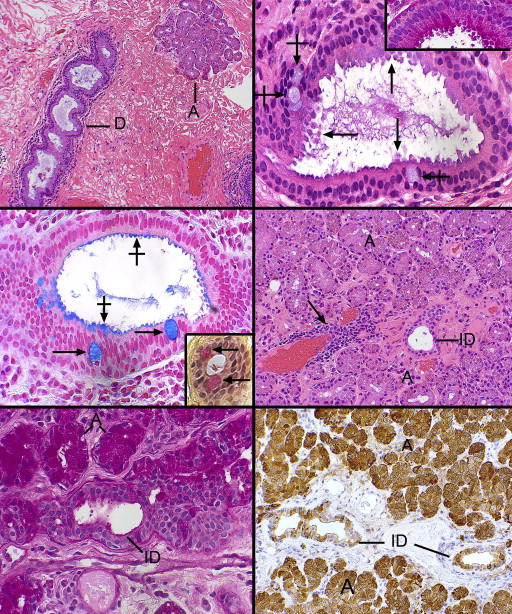
The apical cell membrane and its villiform projections were also PAS-, alcian blue–, and mucicarmine-positive, as were goblet cells, which were ubiquitously present in the epithelial walls of the interlobular (but not intralobular) and excretory ducts ( Figure 2 , Upper right, Middle left and inset). In addition to a small number of intralobular ducts, the majority of the lobular subunits of the lacrimal gland were composed mostly of eosinophilic, granular cells with pyramidal or cuboidal epithelial cells manifesting inconspicuous lumens ( Figure 2 , Middle right). The PAS stain disclosed that the cytoplasmic (zymogen) granules were intensely positive and resisted digestion from pretreatment with diastase ( Figure 2 , Bottom left). The intralobular ducts, with their more obvious lumens, lacked these cytoplasmic granules. The ducts failed to stain for iron with Prussian blue. Scattered between the acini were many plasma cells and rare lymphoid aggregates without a follicular organization, which had a predilection for periductular or perivascular locations ( Figure 2 , Middle right).
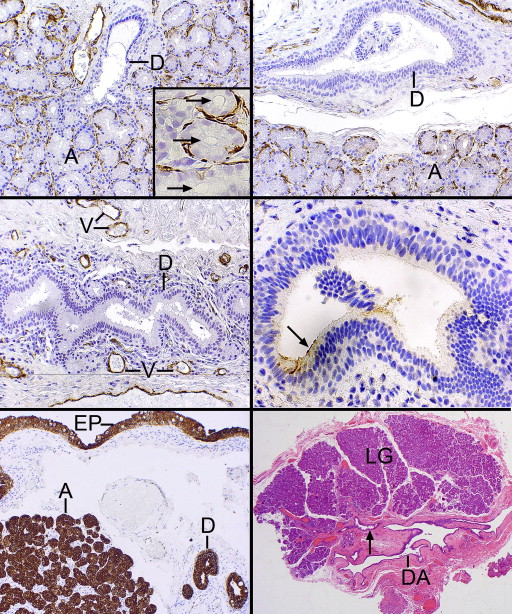
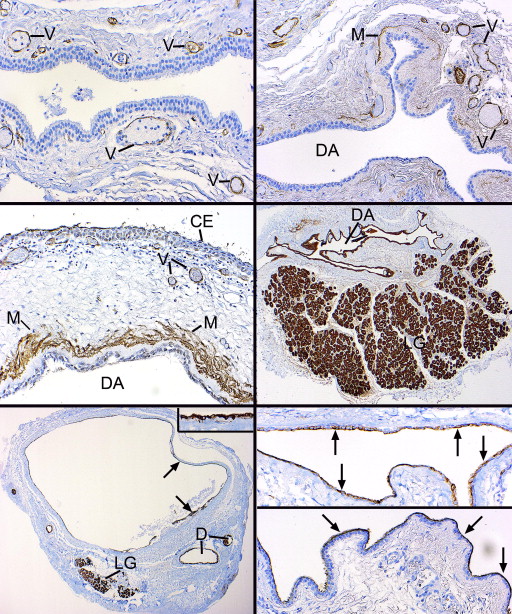
Immunohistochemical staining with a monoclonal antibody–directed gross cystic disease fluid protein-15 (GCDFP-15) demonstrated that the acini were diffusely positive but that the intralobular and early interlobular ducts were only spottily positive in their cytoplasms ( Figure 2 , Bottom right). Antibodies against SMA revealed that the intralobular ( Figure 3 , Top left), interlobular ( Figure 3 , Top right), and main excretory ( Figure 3 , Middle left) ducts were bereft of an outer, positively staining myoepithelial layer, whereas the acini all displayed a compressed outer myoepithelium intimately applied to the basal region of the acinar cells ( Figure 3 , Top left and inset and Top right). The critical finding of SMA negativity of all ducts was confirmed by employing other immunohistochemical stains on 2 specimens for muscle differentiation in the basilar ductal cells, all of which were also negative (but positive in the acinar myoepithelium): S-100, calponin, and smooth muscle myosin. GFAP and desmin were not identifiable in either the dacryops or acini. The larger interlobular and main excretory ducts were also completely lacking any cytoplasmic staining for GCDFP-15, although an interrupted or attenuated cuticle-like deposit was often observed along the adlumenal plasma membrane ( Figure 3 , Middle right), perhaps representing a deposit derived from some component of the tears. Cytokeratin 7 (CK7) immunoreacted with the conjunctival epithelium, the ducts, and acini with equal intensity ( Figure 3 , Bottom left). No SMA-positive cells were discovered within the conjunctival basilar epithelium, nor was there any suggestion of patchy differentiation detected within the conjunctival epithelial cells themselves.
Histopathologic and Immunohistochemical Features of Dacryops
In 12 of the 14 cases the dacryops cavity was collapsed and microscopically appeared as many serpiginous passageways with variably prominent adjacent lobules of lacrimal gland parenchyma ( Figure 3 , Bottom right and Figure 4 , Top left). The outermost reaches of the labyrinthine outlines radiated toward the many separate lacrimal gland lobules. In 2 cystic specimens an overall rounded outline was offered along with a prominent fibrous wall, 1 containing a focus of active fibroblasts ( Figure 4 , Top right). Another case of dacryops of a superior forniceal accessory gland of Krause ( Figure 4 , Middle left) also had a rounded configuration. Both lacrimal gland lobules and dilated ducts were present next to the dominant cyst. Inflammation in the fibrous wall of the dacryops was generally not observed, although it was present in the Krause dacryops ( Figure 4 , Middle left), probably indicative of indigenous conjunctival lymphoid aggregates. Occasional collections of small lymphocytes without follicle formation in a periductular location were identified as in the normal lacrimal gland ( Figure 4 , Middle right). Three of the cystic spaces lacked a well-defined fibrous wall ( Figure 4 , Middle right), which typically was highlighted with the Masson trichrome stain. This stain also failed to reveal any intensely reddish cytoplasm with longitudinal filamentation that might suggest the possibility of a myoepithelial role for the outer ductular epithelial cells. Inflammation in the lobules of lacrimal tissue above the usual presence of scattered interstitial lymphocytes and plasma cells was detected in 3 cases as a mild fibrosing dacryoadenitis with early loss of acinar elements.
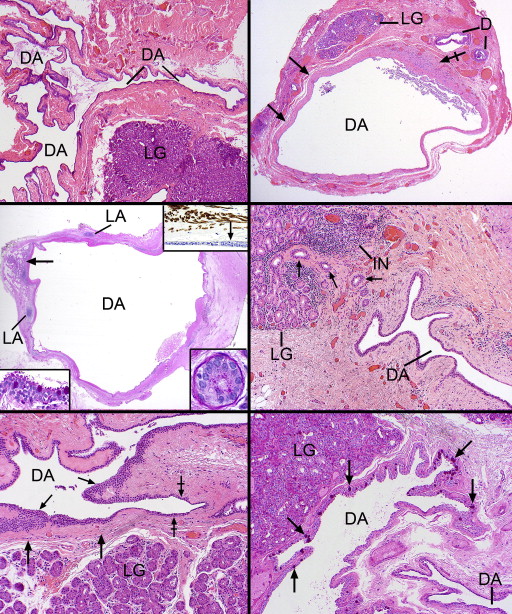
The lining of the dacryops spaces consisted generally of a double layer of cuboidal cells (rarely focally a single layer), which in oblique sections could appear to be multilaminar ( Figure 4 , Bottom left). No papillations or zones of squamous metaplasia or hyperkeratosis were found in the linings. In all specimens PAS-positive goblet cells were generously distributed among the inner cells ( Figure 4 , Bottom right and Figure 5 , Top left). All epithelial linings in focal areas displayed adlumenal cells with apical snouts ( Figure 5 , Top right), which had not dehisced into the secretions. These apical zones stained intensely positively with the PAS stain (resistant to diastase), in contrast with the rest of the nonstaining cytoplasm, except for the diffuse cytoplasmic positivity of the admixed goblet cells. The dacryops linings did not possess discrete PAS-positive granules (thereby distinguishing them from acinar cells), nor was there any evidence of the presence of iron in the cytoplasm as demonstrated with the Prussian blue stain. In 2 specimens, spheroidal or globoid bodies were discovered in the lumen of the dacryops and were GCDFP-15 positive, in contrast to the negativity of the dacryops linings themselves ( Figure 5 , Middle left and inset). A trail-off of the dacryops spaces into tortuous dilated ducts was clearly detected in approximately half the specimens ( Figure 5 , Middle right, top and bottom panels).
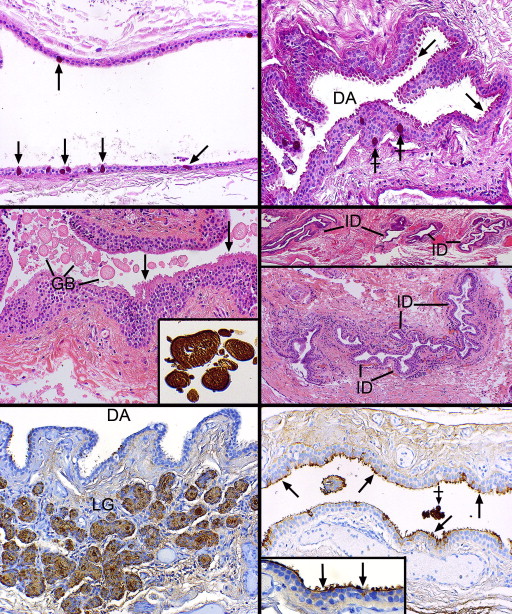
Immunohistochemical investigations disclosed that the double layer of cells lining the dacryops spaces were GCDFP-15-negative, whereas the acini of the lobular lacrimal tissue was uniformly positive ( Figure 5 , Bottom left). A finding in three-quarters of the dacryops cases was a cuticular membrane or deposit of GCDFP-15 positivity on the apical surfaces of the adlumenal cells ( Figure 5 , Bottom right). Small amounts of preserved luminal secretion were also positive. SMA failed to demonstrate any positivity among the basilar lining cells of the dacryops cavity, as would be expected if they were a myoepithelium ( Figure 6 , Top left); thin positive circular outlines delineating the muscular component of small vessels served as a positive internal control. In the fibrous walls of the dacryops of 4 specimens, dispersed positive thin myofibroblastic cells were observed and were always separated from the negative dacryops basilar lining cells by a delicate mantle of collagen ( Figure 6 , Top right). One palpebral lobe lacrimal gland lesion with a dominant rounded cyst exhibited a multilaminar zone of interweaving myofibroblastic cells separated from the lining cells by the interposition of collagen ( Figure 6 , Middle left). CK7 immunostained the dacryops linings and the lobules of the lacrimal gland ( Figure 6 , Middle right) as well as any dilated ducts nearby the lesion ( Figure 6 , Bottom left). Epithelial membrane (EMA) reacted with the apical and lateral cell borders of most of the lining cells ( Figure 6 , Bottom right, top panel), while carcinoembryonic membrane antigen (CEA) positivity was more unreliable, irregular, and generally found only on the apical membrane as a thin lamina or cuticle ( Figure 6 , Bottom right, bottom panel).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


