16 SYSTEMIC DISEASE AND THE EYE
Diabetes Mellitus
Nicky R. Holdeman
ICD-9: 250.00
 THE DISEASE
THE DISEASE
Pathophysiology
Diabetes mellitus (DM) involves a complex sequence of events that often evolve over many years. It is characterized by a broad array of abnormalities and frequently results in serious, long-term vascular and neurologic complications. Pathologic changes may lead to systemic hypertension, renal failure, blindness, autonomic and peripheral neuropathy, amputations of the lower extremities, myocardial infarctions (MIs), peripheral artery disease, and strokes.
The most notable feature of the disease is a disordered metabolism of glucose leading to hyperglycemia. Elevation in blood glucose results from an absolute deficiency of insulin secretion, a reduction in its effectiveness (insulin resistance), or an increased hepatic glucose output. Chronic hyperglycemia is presumed to cause tissue damage by glycation of proteins, by altering cell signaling pathways, and/or excess production of polyol compounds from the metabolism of glucose.
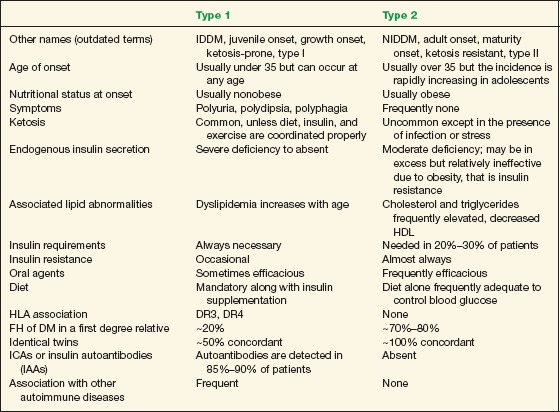
There are two major types of DM: type 1 (formerly juvenile onset, type I or insulin-dependent diabetes mellitus—IDDM) and type 2 (formerly adult onset, type II or noninsulin-dependent diabetes mellitus—NIDDM). Other less common forms of DM exist but are beyond the scope of this section. Each form of DM has a distinct etiology, clinical presentation, and course.
Note: The terms and acronyms IDDM and NIDDM are no longer used since the names are based on pharmacological treatment rather than causative considerations. The current classification system recognizes that DM results from different mechanisms, with the common endpoint being hyperglycemia.
Etiology
Type 1—Type 1 diabetes mellitus is an autoimmune disease in which pathologic, autoreactive T cells attack the insulin-secreting pancreatic islets of Langerhans, thereby leading to lifelong dependence on insulin for affected patients. This disease process often results in the production of autoantibodies, such as anti-islet cell autoantibodies (ICAs), autoantibodies to insulin (IAAs), and glutamic acid decarboxylase (GAD65). There is evidence that a precipitating event, such as a viral illness, may be required to trigger the destructive process. Other studies suggest that toxins in the environment, or diet supplementation with gluten-containing foods before age 3 months, might also initiate damage of genetically vulnerable beta cells. Whatever the etiology, clinical diabetes occurs when roughly 90% of the beta cells have been destroyed; this process can be quite rapid in infants and children and slower in adults. Circulating insulin in type 1 diabetes is virtually absent, thus these patients are dependent on exogenous insulin for survival. Ketosis is common in patients with poor glucose control and ketoacidosis can be life threatening.
Note: The type 1 Diabetes TrialNet studies (http://www.diabetestrialnet.org) are investigating the treatment of patients who have newly diagnosed type 1 diabetes, using immunologic agents (i.e., rituximab, mycophenolate mofetil, and daclizumab) to determine if these drugs can prevent further beta cell destruction. The studies may demonstrate that the incidence of type 1 diabetes can be reduced.
Type 2—The exact etiology of type 2 diabetes mellitus continues to be debated; however, development can often be correlated with obesity, in particular increases in visceral fat. Some patients may initially show normal insulin levels, but the natural history of type 2 diabetes reveals a gradual decrease in the number of insulin-producing beta cells, resulting in a relative insulin deficiency that worsens over time. At the point of diagnosis, a patient may have already lost 50% of their beta cell function, and he or she will typically lose 4% per year thereafter. Rising glucose levels further compromise insulin secretion and insulin action, a phenomenon known as “glucose toxicity.”
Ultimately, patients with type 2 diabetes develop a “dual defect” disease characterized by insulin resistance and a defect in compensatory insulin secretion. These individuals may also exhibit reduced numbers of insulin receptors, increased hepatic glucose production, impaired glycogen synthesis, paradoxical rise of glucagon in response to meals, decreased peripheral glucose uptake, abnormal patterns of insulin secretion, accelerated gastric emptying, or a combination of these conditions. Circulating endogenous insulin is usually sufficient to prevent ketoacidosis but inadequate to prevent hyperglycemia.
Note: It is now believed that about 2 million Americans currently diagnosed with type 2 diabetes may actually have latent autoimmune diabetes in adults (LADA), also referred to as type 1.5 DM. This condition appears to be a hybrid or “stealth” type of diabetes, which progresses slower in its attack and destruction of insulin-producing beta cells than type 1. Patients with LADA cannot normalize their blood glucose with oral antidiabetic agents and thus require exogenous insulin supplementation.
The Patient
Clinical Symptoms
Some cases of DM (especially type 2) are diagnosed at an asymptomatic stage as a result of a screening program, routine blood tests, or an eye examination. Compared with type 1 patients who commonly present with acute symptoms, individuals with type 2 diabetes are much more likely to have no symptoms or fewer symptoms, as the hyperglycemia develops gradually. Patients may report the classic syndrome of polyuria, polydipsia, polyphagia, unexplained weight loss, weakness, and fatigue; however, these symptoms are typically associated with marked hyperglycemia as seen in type 1 patients (Table 16-1).
TABLE 16-1 Clinical Features of DM*

*Note: While many of these signs and symptoms may occur in both type 1 and type 2 diabetes, those conditions indicated with a dagger (†) are most commonly detected in type 1 disease and associated with marked hyperglycemia.
Retinopathy may be detected in over 20% of type 2 patients at the time of initial diagnosis and may predate the diagnosis of diabetes by up to 6 years.
Early detection, improved delivery of care, and better self-management are key strategies for preventing diabetes-related complications. (For every 1% decrease in the A1c, there is approximately a 35% reduction in the risk of diabetic microvasculature complications.)
Clinical Signs
The physical findings will vary depending on the type of DM, the severity of insulin deficiency, and the duration of the disease. Examination may reveal systemic hypertension, neuropathy, periodontal disease, vaginitis, skin abnormalities, arteriopathy, blurred vision, retinopathy (Figs. 16-1 and 16-2), nephropathy, and/or increased susceptibility to infections. Diabetics also have a much higher risk for cardiovascular (CV) disease, abnormalities in lipid metabolism (dyslipidemia), and vascular thrombosis (see Table 16-1).
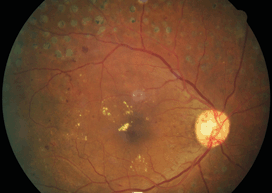
Figure 16-1. Proliferative DR with NVD and CSME. Note prior PRP scars in the periphery.
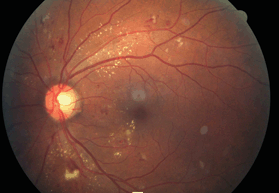
Figure 16-2. Moderate to severe nonproliferative DR with CSME.
Ocular manifestations of DM occur frequently and careful examination with appropriate treatment can often reduce the ophthalmic complications (Table 16-2). Clinicians should also be aware that the presence of any diabetic retinopathy (DR) signifies an excess risk of renal failure and of heart failure (HF) and that visual impairment, resulting from DR, predicts a high mortality rate, particularly from CV diseases and stroke.
TABLE 16-2 Ocular and Periocular Manifestations of DM

Patients >10 years of age with type 1 diabetes should have an initial dilated and comprehensive eye examination within 3 to 5 years after the onset of diabetes, and at least every year thereafter.
Patients with type 2 diabetes should have an initial dilated and comprehensive eye examination shortly after diagnosis, and at least every year thereafter.
Demographics
- The estimated lifetime risk of developing DM for individuals born in 2000 is 32.8% for males and 38.5% for females.
- DM is the sixth leading cause of death in the United States and is believed to be underreported both as a condition and as a cause of death. It is estimated that up to 17% of all deaths in the United States are related to diabetes.
- The incidence, target population, and genetics differ between type 1 and type 2 (Table 16-3).
- It is estimated that over 27 million people in the United States (>200 million worldwide) have DM and that the number of patients with diabetes will double every decade.
- About a fourth of U.S. adults, or 57 million people, have prediabetes. These patients have a substantially increased risk of CV disease and death.
- Type 1 diabetes affects 1 in 250 persons in the United States. Approximately 10,000 to 15,000 new cases are reported each year, and the disease appears to be on the rise.
- Type 1 diabetes is less common among black and Asian populations.
- Type 2 diabetes accounts for approximately 90% to 95% of all cases of diabetes among American adults.
- Type 2 diabetes has an overall prevalence of approximately 9% in the United States, but it is estimated that by 2010, over 10% of the population will have type 2 diabetes. One and a half million new cases are reported each year; approximately 25% to 30% of patients are unaware of their disorder at the time of diagnosis.
- Type 2 diabetes is more common in Native Americans, African Americans, and Hispanic/Latino Americans. This same population has a three- to sixfold higher incidence of diabetes-related end-stage renal disease than seen in whites.
- Type 2 diabetes increases with age, lack of physical activity, and obesity. Individuals who have a body mass index (BMI) greater than 30 are at 10 to 20 times greater risk. Approximately 28% of U.S. adults aged 20 years and older are obese.
- Type 2 diabetes occurs more frequently in women with prior gestational DM; these women have a 20% to 50% chance of developing diabetes over 5 to 10 years.
- DR is the leading cause of new cases of blindness in patients 25 to 74 years of age. In 2007, DR affected more than 5.5 million Americans, and it is predicted that the number of cases will at least triple by 2050.
- Each year, 12,000 to 24,000 people lose their sight due to diabetes. African Americans are twice as likely to suffer from diabetes-related blindness.
- Type 1 diabetes is less common among black and Asian populations.
TABLE 16-3 Comparison of the Essential Features of Type 1 and Type 2 DM
Significant History
- Age (type 2 DM is more common in people greater than 45 years of age; however, it is becoming increasingly more common in younger individuals, even adolescents)
- Family history (FH) of DM in a first-degree relative; if FH is positive for DM, determine age of onset, whether it was associated with obesity, and form of treatment. FH is much stronger in type 2 (70% to 80%) versus type 1 (20%)
- Diabetic patients with family members who have had severe retinopathy are more likely than other diabetics to have more severe retinal disease
- Unexplained weight loss (type 1) or visceral obesity and increased BMI (type 2)
- Physical inactivity
- Polyuria, polydipsia, polyphagia may be reported with marked hyperglycemia
- Chronic or recurrent infections
- Gestational diabetes, or having delivered a baby weighing greater than 9 lb
- Long or highly irregular menstrual cycles
- Member of a high-risk ethnic population
- Systemic hypertension, smoking, dyslipidemia, use of oral contraceptives, or other factors that increase cardiac risk
Ancillary Tests
The risk of developing type 2 diabetes is increased with plasma glucose (PG) levels well below the current cut-point for defining impaired fasting glucose (IFG), which is 100 to 125 mg/dL. Fasting plasma glucose (FPG) levels, even as low as 90 mg/dL, have been shown to be associated with an increased risk for type 2 diabetes. However, diabetes risk associated with fasting glucose should be viewed in the context of other risk factors such as systemic hypertension, dyslipidemia, and obesity. While the definition or cut-point of DM may change with time, the conventional criteria are as follows:
A diagnosis of DM, in nonpregnant adults, is appropriate in one of the following circumstances. Each must be confirmed on a subsequent day unless unequivocal symptoms of hyperglycemia are present:
- Symptoms of diabetes plus casual PG concentration ≥ 200 mg/dL (11.1 mmol/L). Casual is defined as any time of day without regard to time since last meal. The classic symptoms of diabetes include polyuria, polydipsia, and unexplained weight loss.
- FPG ≥ 126 mg/dL (7.0 mmol/L). Fasting is defined as no caloric intake for at least 8 hours. If the FPG level is over 126 mg/dL on more than one occasion, further evaluation of the patient with a glucose challenge is unnecessary.
- 2-h PG ≥ 200 mg/dL during an oral glucose tolerance test.
Note: The International Expert Committee and the American Diabetes Association (ADA) have revised the recommendations listed above to include an A1c value of 6.5% or greater in the diagnosis of diabetes. A1c tests are now reproducible, standardized between labs, and are more convenient for patients as the test does not require overnight fasting. Compliance with screening may therefore be improved using the A1c test, which can be determined from a single nonfasting blood sample. However, A1c testing is more expensive than FPG, and values may vary with age, ethnicity, anemia, renal insufficiency, and certain infections. A cut-point of ≥6.5% was selected to diagnose diabetes, in that it tightly correlates with the risk of developing retinopathy.
An A1c level of approximately 5% indicates the absence of diabetes.
Values between 5.7% and 6.4% are considered “prediabetic” as they are likely at the highest risk for progression to diabetes, as are patients with IFG and impaired glucose tolerance (IGT). Like other diagnostic tests for diabetes, an abnormal value should be confirmed with a repeat test.
If a diagnosis of DM is made, other baseline tests, procedures, and examinations should be performed. These assessments may reveal coexistent disorders, document preexisting target organ damage (TOD), and help guide future management. Evaluations include
- Fasting plasma triglycerides
- Total cholesterol, HDL cholesterol, LDL cholesterol
- Electrocardiography (ECG)
- Renal function studies
- Peripheral pulses (to detect peripheral artery disease or PAD)
- Neurologic, podiatric, and eye examinations
Self-monitoring of blood glucose (SMBG) is sometimes employed in the management of diabetes. Most would agree that SMBG is associated with better glycemic control, especially in insulin-treated diabetics or when adding to or modifying oral therapy.
For most patients with type 1 diabetes, SMBG is recommended three or more times daily. The optimal frequency and timing of SMBG for patients with type 2 diabetes on oral therapy is not known but may be incorporated in order to facilitate reaching glucose goals. However, for well-controlled patients with noninsulin treated type 2 diabetes, home monitoring of blood glucose is of questionable value, is expensive, and may increase the risk of depression.
The accuracy of data obtained by any glucose monitoring system requires proper instruction of the patient in sampling and measuring procedures as well as proper calibration of the instruments. Audible glucose monitoring devices, in both English and Spanish, are available for diabetics who are visually impaired.
Daily blood glucose levels should be less than 110 mg/dL before eating breakfast (preprandial) and less than 140 mg/dL 2 hours after eating a meal (postprandial). In addition, measurement of 1,5-anhydroglucitol (1,5-AG) levels in serum can be used to monitor postprandial glycemic control in patients with moderately well-controlled diabetes. The 1,5-AG serum assay (GlycoMark) can be used to measure postprandial glucose levels over 1 to 2 weeks.
Continuous glucose monitoring devices, which measure interstitial glucose via subcutaneous sensors, may be particularly beneficial for diabetics who are at risk for nocturnal hypoglycemia. These devices have alarms that can awaken a patient and prevent a seizure. However, this monitoring system is expensive, has some accuracy limitations, and requires patient motivation. Although this method is appealing theoretically, the extent to which it will improve long-term clinical outcomes remains to be determined.
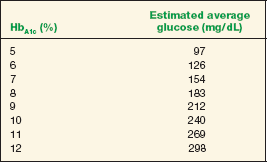
Glycosylated hemoglobin (HbA1c or A1c) reflects metabolic control and state of glycemia over the preceding 8 to 12 weeks and is directly proportional to the concentration of free glucose in the blood (Table 16-4). This test, while now approved for the initial diagnosis of diabetes, also provides a unique method of assessing and monitoring diabetic management. Each 1-point reduction in the A1c reduces the risk for DR by 39% for type 1 diabetes and 25% for type 2. Three to four A1c measurements a year are usually sufficient. A reasonable goal to achieve and maintain is near normoglycemia (~7.0%); however, this level should be individualized based on patient characteristics. Once clinicians and patients have chosen an individualized, optimal A1c, they should design a management regimen to achieve that target. Intensive control, yielding A1c levels below 7.0%, is not effective in reducing macrovascular events, appears to have no additional beneficial effect on retinopathy, and increases patient burden, cost, and the risk for hypoglycemia.
TABLE 16-4 HbA1c Percentage and Corresponding Estimated Average Glucose Level
Urine strips are a precise and convenient method to detect glucosuria (Diastix are sensitive to as little as 0.1% glucose in urine). However, the renal threshold for PG is approximately 180 mg/dL, thus urine glucose is not the method of choice for monitoring diabetic control.
Urine testing for ketones (Acetest or Ketostix) is important for type 1 diabetics when they are ill, under stress, or when their blood glucose exceeds 240 mg/dL.
Note: A noninvasive imaging device that identifies elevated levels of flavoprotein autofluorescence in retinal tissue, which is caused by increased metabolic stress, may provide evidence of diabetes or diabetic eye disease before neuronal loss is evident. This technology may allow clinicians to diagnose diabetes earlier and to initiate glycemic control before extensive ocular tissue damage occurs.
The Treatment
Diabetes care is complex and requires that many health-related issues, beyond glycemic control, be addressed. A complete medical evaluation should be performed to detect the presence of diabetic complications, to formulate a management plan, and to provide a basis for continuing care. Currently, the treatment objectives for both type 1 and 2 are similar and that is to maintain glycemic control as close to normal as possible, while avoiding undue risk for adverse events or placing an unacceptable burden on the patient. Treatment strategies should be individualized based on the type of diabetes, comorbidities, life expectancy, and the specific requirements of each patient.
The management of DM involves lifestyle adaptations such as diet and exercise, to improve glucose levels, blood pressure (BP), lipid levels, and weight control. Bariatric surgery is a very effective treatment option for type 2 diabetics who are morbidly obese. Studies show improved survival from heart disease and cancer in patients following bariatric surgery for morbid obesity, but also report a 73% remission in type 2 diabetes compared with nonsurgical treatments.
Standard medications include several classes of oral antidiabetic agents (such as sulfonylureas, biguanides, alpha-glucosidase inhibitors, glitazones, meglitinides, and centrally acting dopamine receptor agonists) (Table 16-5) as well as various parenteral insulin preparations (Table 16-6). For most patients with type 2 diabetes, the initial treatment should include lifestyle intervention and use of metformin, which has the best profile of benefit to risk and is the only medication proven to reduce mortality rates. If this first strategy (tier 1, step 1) fails to achieve or maintain target glycemic goals, other medications should be added rapidly. It is important to note that many type 2 patients who start on oral agents will eventually require insulin to maintain adequate glycemic control. Insulin plus metformin is a particularly effective means to lower glycemia while limiting weight gain. Insulin therapy may also abate the expense, side effects, and inconvenience of taking multiple oral drugs several times a day.
TABLE 16-5 Oral Antidiabetic Drugs
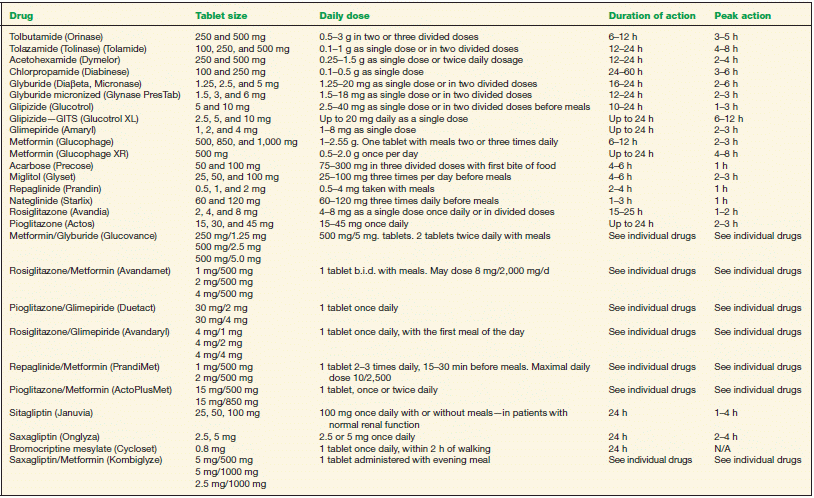
TABLE 16-6 Insulin Preparations
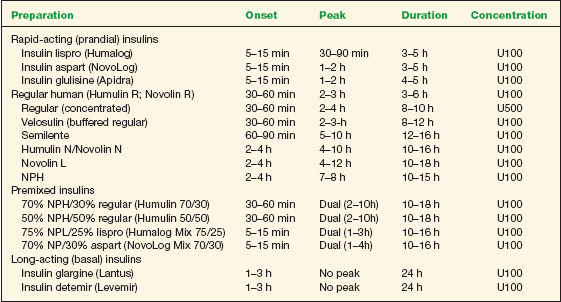
The time course of action of any insulin may vary in different individuals, or at different times in the same individual. Because of this variation, times indicated above should be considered general guidelines only.
There are a number of different types of injected insulin, which can be used to comprise a regimen that can approximate the normal response of the beta cell.
Rapid-acting insulins can be used at meals to control postprandial glucose. Longer-acting insulins, including NPH, glargine, and detemir, provide basal or background insulin. NPH is intermediate-acting insulin given twice daily; glargine and detemir have 24-hour profiles and are given once daily.
Premixed insulins are a fixed combination of a long-acting and rapid-acting insulin and may be given twice daily. These premixed formulations actually show an asymmetric peak, with a skewing toward the longer acting insulin.
Continuous infusion of insulin (insulin pump) usually achieves slightly better glycemic control than does an optimized-injection regimen. However, it is not clear whether insulin pumps are cost effective and they are usually reserved for patients in whom injection regimens do not provide adequate control.
Note: There is some controversy that the glitazone drugs (i.e., rosiglitazone and pioglitazone) may increase the risk of developing macular edema (ME) in some diabetic patients. The most current studies (ACCORD) show that these drugs appear to be safe to use in diabetic patients without contributing to this important complication of the disease.
Several newer drugs include:
Exenatide (Byetta) and liraglutide (Victoza) are incretin mimetics (a glucagon-like peptide or GLP-1 agonist) used as adjunctive therapy to improve glycemic control in patients with type 2 diabetes who have not achieved adequate control with metformin, a thiazolidinedione, and/or a sulfonylurea. These drugs may be well suited for patients with hazardous jobs that would make hypoglycemia particularly dangerous and in those that need to lose weight and in whom the HbA1c level is close to target (<8%). The drugs are available as a prefilled pen injector device.
GLP-1 agonists work by stimulating insulin secretion, normalizing release of glucagon, slowing gastric emptying, and improving satiety. Compared to other antidiabetic agents, these drugs do not cause hypoglycemia, and they are the first glucose-lowering drugs to demonstrate substantial and sustained weight loss. Nausea is a common problem and other side effects are possible.
Sitagliptin (Januvia) and saxagliptin (Onglyza) are oral dipeptidyl peptidase-4 (DPP-4) inhibitors and may be appropriate for select patients with type 2 diabetes. Blocking the action of DPP-4 prolongs the activity of the incretin proteins, leading to better insulin release, suppressing the release of glucagon, and delaying gastric emptying, which collectively results in better blood glucose control.
Note: Neither GLP-1 agonists nor DPP-4 inhibitors cause hypoglycemia unless used with other agents known to increase risk.
Pramlintide (Symlin) is a synthetic analog of the β cell co-secreted pancreatic hormone amylin. This medication is injected subQ just before meals with insulin, in a different syringe from the insulin syringe. Symlin lowers postprandial blood glucose by slowing gastric emptying, by suppressing glucagon secretion, and by modulating the appetite, which may lead to weight loss.
The Food and Drug Administration (FDA) approved Exubera, an inhaled insulin, in January 2006. Unfortunately, due to the poor level of adoption by both physicians and patients, the manufacturer removed it from the market in October 2007. Other formulations of inhaled insulin and their delivery systems appear very promising (i.e., technosphere insulin—Afresa).
Coexistent disorders such as smoking, obesity, arterial hypertension, and/or dyslipidemia must be addressed as they may increase the risk and accelerate the progression of diabetic complications (i.e., retinopathy, neuropathy, nephropathy, and peripheral vascular disease). If no contraindications exist, enteric-coated aspirin (75 to 162 mg/d) is reasonable for older diabetic adults who are at increased CV risk and/or in patients with symptomatic CV disease. The risk of ischemic heart disease is two to four times greater in diabetics, occurs at a younger age, and is much higher in women with diabetes.
DR, even in its mildest form, is an independent predictor of kidney disease and of the risk for HF. A careful cardiac assessment is recommended for patients with DR to allow detection of subclinical HF or left ventricular dysfunction.
The Diabetes Control and Complications Trial (DCCT) demonstrated that “tight” metabolic control, geared toward maintaining near normal glucose levels, had a definite and beneficial effect on the development and progression of long-term diabetic microvascular complications in type 1 diabetics. In addition, the Epidemiology of Diabetes Interventions and Complications (EDIC) study showed a reduction in the risk of both microvascular and macrovascular events resulting from intensive therapy, which persisted for years, despite increasing levels of blood glucose.
Patients with type 2 DM were not included in the DCCT or EDIC studies. However, the United Kingdom Prospective Diabetes Study (UKPDS) showed that intensive therapy in younger patients with new onset type 2 diabetes did indeed reduce diabetes-related microangiopathic organ damage by 25%. Surprisingly, the treatment of systemic hypertension and dyslipidemia proved more successful in reducing CV mortality than reducing the patients HbA1c levels. This apparent contradiction was thought to be due to the many potential mechanisms of increased CV damage in diabetes, including systemic hypertension, abnormal clotting function, platelet adherence, abnormal vascular reactivity, and abnormal lipid profiles. The Steno 2 study, which addressed many of these suspected risk factors, confirmed that appropriate management of glycemia, systemic HTN, dyslipidemia, microalbinuria, and risk of thrombosis significantly reduced both micro and macrovascular complications. There appear to be impressive and definitive long-term benefits from multiple risk factor intervention.
Note: Intensive regulation of blood glucose is difficult at best and glycemic controls should always be realistic. In some patients (e.g., small children, the elderly, those with advanced renal disease), the criteria for optimal control may need to be adjusted upward as the detrimental risks of hypoglycemia may outweigh the benefits of tight glycemic control.
In addition, several recent trials (ADVANCE, ACCORD, and VADT) are consistent in showing no significant benefit of very tight glycemic control (HbA1c ≤ 6%) on macrovascular or microvascular events in older type 2 diabetics with established disease. However, the 10-year follow-up of the UKPDS suggested a definite benefit (or “legacy effect”) of tight glycemic control in patients with type 2 diabetes, which extended beyond a finite period of intensive management. The observations from the more recent trials suggest that intensive glucose-lowering therapy may hold more promise for younger, newly diagnosed type 2 diabetics—the analysis of data is ongoing.
In general, it is reasonable to conclude that multifactorial treatments such as glucose control that minimizes hypoglycemia (HbA1c ~7%), controlling BP (<140/80 mm Hg), and other risk factors (diet, exercise, smoking cessation, and lipid lowering) should be considered in most patients with diabetes, regardless of their age, in order to reduce the risk of CV mortality and all-cause mortality.
Transient worsening of retinopathy (and neuropathy) during the first 6 to 12 months of intensive therapy has been noted in several clinical trials and is referred to as “treatment-induced diabetic retinopathy or neuropathy.”
Ophthalmic management of retinal disease involves the use of focal or grid laser treatment for clinically significant macular edema (CSME) (see Table 16-2) and pan-retinal photocoagulation for retinopathy of high-risk characteristics. High-risk characteristics include
- Neovascularization of the disc (NVD) greater than l/4 the disc area in size, on or within 1 DD of the optic disc
- Any amount of NVD if accompanied by a fresh preretinal or vitreous hemorrhage
- Neovascularization elsewhere (NVE) greater than 1/2 the disc area, with a fresh vitreous or preretinal hemorrhage
Appropriate glycemic control, blood lipid control, and BP control in type 1 and type 2 diabetes remain the cornerstone for prevention of DR and its progression. Secondary measures include laser photocoagulation, vitrectomy, and intravitreal triamcinolone. Intravitreal implants and intravitreal injections of antiangiogenesis (anti VEGF) agents are also effective in selected patients.
One should keep in mind that DR may progress more rapidly in some patients and that various risk factors may increase the chances for the development of retinal complications. The risk of retinal complications in diabetics increases with the following:
- Duration of disease
- Uncontrolled systemic hypertension
- Abnormal lipid profiles
- Elevated levels of hemoglobin (increased progression of PDR and diabetic macular edema [DME])
- Pregnancy
- Cataract surgery or YAG capsulotomy (intravitreal injection of bevacizumab at the conclusion of cataract surgery may reduce the risk of progression of DR in diabetic patients undergoing phacoemulsification).
- Carotid disease
- Poor glycemic control (9 years exposure to an A1c of 8% yields the same risk of retinopathy as 2.5 years exposure to an A1c of 11%.)
- Proteinuria
- Strenuous exercise (proliferative DR is a relative contraindication to strenuous exercise, since strenuous activity could potentially precipitate a vitreous hemorrhage)
- Proteinuria
The International Clinical Disease Severity Grading Scale for Diabetic Retinopathy and Diabetic Macular Edema represents a simplified version of the ETDRS classification (Tables 16-7 and 16-8):
TABLE 16-7 International Clinical DR Disease Severity Scale

TABLE 16-8 International Clinical DME Scale

*Hard exudates are a sign of current or previous ME. DME is defined as retinal thickening, which requires a three-dimensional assessment best performed by a dilated examination using slit lamp biomicroscopy and/or stereo fundus photography. CSME can be documented and followed by noninvasive optical coherence tomography.
For additional information:
American Diabetes Association (ADA): www.diabetes.org/
National Diabetes Information Clearinghouse: www.niddk/nih.gov/health/diabetes/nidc.htm
Systemic Hypertension
Nicky R. Holdeman
ICD-9: 401.1
 THE DISEASE
THE DISEASE
Pathophysiology
Systemic hypertension (HTN) is a multifactorial disease produced by an array of pathogenetic mechanisms. Essential (or primary) hypertension appears to involve a complex interplay between susceptibility genes, environmental factors, the central nervous system, the kidney, and the renin-angiotensin-aldosterone system (i.e., the systems that influence vascular tone, heart contractility, and fluid volume).
Systolic hypertension, in the elderly is due to age-related reductions in the elasticity (stiffening or fibrosis) of large and medium-sized arteries, leading to a progressive rise in the systolic BP. The diastolic blood pressure (DBP), on the other hand, tends to peak around age 50 and then begins to decrease with advancing years. This increasing difference between the systolic and diastolic BP causes the familiar widening of pulse pressure with age.
Etiology
In essential HTN, which accounts for 90% to 95% of cases, no specific etiology has been established. In addition to the potential factors listed above, chronic systemic inflammation and oxidative stress may also play a role in the development of essential HTN.
Causes of secondary HTN (those 5% to 10% of cases with an identifiable etiology) include:
- Obesity and obstructive sleep apnea
- Drug induced or drug related (i.e., NSAIDs, steroids, oral contraceptives, cyclosporine, cocaine, alcohol)
- Pregnancy (gestational hypertension)
- Renal parenchymal disease
- Renovascular hypertension (i.e., renal artery stenosis)
- Coarctation of the aorta
- Hyperaldosteronism (Conn’s syndrome)
- Cushing’s disease
- Hyperparathyroidism
- Hyperthyroidism
- Hypercalcemia
- Pheochromocytoma
- Acromegaly
Blood pressure (BP) rise and hypertension are not necessarily an inevitable consequence of aging; however, isolated systolic hypertension (ISH) frequently occurs in individuals over 60 years of age. ISH is defined as a systolic blood pressure (SBP) of 140 mm Hg or more and a DBP less than 90 mm Hg. ISH is related to loss of arterial elasticity (or compliance) of large arteries due to atherosclerosis and is often more resistant to treatment than essential hypertension. ISH is the most frequent form of uncontrolled hypertension in the United States. For individuals older than 50 years of age, the SBP is a more important predictor of CV disease than the diastolic pressure. Conversely, hypertensive individuals, younger than 50, may have isolated diastolic hypertension. Therefore, clinicians should focus equally on the management of an elevated systolic or diastolic reading.
The Patient
Clinical Symptoms
Probably, the greatest challenge for physicians treating high BP is the nonadherence to therapy as the condition is largely asymptomatic until there is end-organ damage. The major target organ damage (TOD) caused by systemic hypertension occurs in the heart, brain, kidney, peripheral arteries, and the retina. While uncommon, elevated BP may produce vague complaints such as suboccipital headaches, palpitations, facial flushing, lightheadedness, or fatigue. Accelerated hypertension may cause visual disturbances.
Clinical Signs
The threshold for the diagnosis of arterial hypertension is when the SBP exceeds 139 mm Hg or when the DBP is higher than 89 mm Hg. However, this definition is arbitrary, and in actuality, there is a continuous relationship with CV risk down to a level of 115/75 mm Hg.
Clinical findings will depend on the etiology of the hypertension, duration, severity, and total effect on target organs. The physical examination should detect evidence of known causes of high BP, assess the presence and extent of TOD and CV disease, and identify other risk factors or concomitant disorders. One should look for the following signs or symptoms (see also Fig. 16-3):
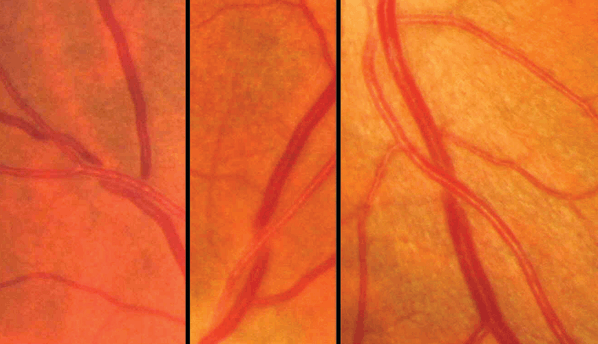
Figure 16-3. Retinal A/V crossing defects (concealment, banking, tapering, and deviation) in patient with systemic hypertension.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


