Fig. 1
(a) Exophytic tumor of right tonsillar fossa. Tumor extends into the oropharyngeal airway but not into the palate, posterorlateral pharynx, or retromolar trigone. During physical exam, an index finger placed lateral to the tumor should confirm that the tumor is mobile and not adherent to the buccopharyngeal fascia or medial pterygoid fascia. (b) A left tonsil tumor showing endophytic growth pattern and submucosal invasion into soft palate. These tumors are often fixed and immobile. Palpation and imaging can help the surgeon discern the lateral boundaries of the tumor, but fixed tumor are not ideal tumors to approach transorally
Fixation of the tumor to the lateral or posterior pharyngeal wall, including deep extension to the parapharyngeal space
Significant involvement of the internal carotid artery as it passes through the parapharynx and retropharynx
Mandibular invasion
Skull base invasion
Distal extension down the lateral pharyngeal wall toward the parapharynx
Anterior extension from the tongue base toward the root of tongue
Extension lateral to the hyoid bone to involve the lingual artery and hypoglossal nerve (Fig. 2).
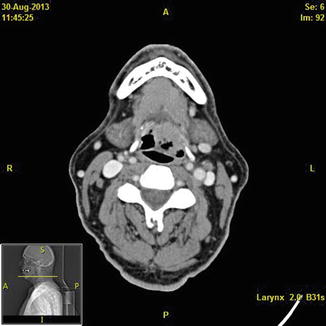
Fig. 2
Axial CT scan with contrast demonstrating a base of tongue tumor on the left side of the patient that extends lateral to the hyoid bone. Tumors in this area often involve the lingual artery, dorsal lingual artery, and hypoglossal nerve. If the surgeon is going to approach these tumors transorally, then dissection should proceed very cautiously in this area to avoid bleeding. Alternative approaches include transhyoid pharyngotomy or neck dissection prior to transoral tumor removal
Patient selection is vital for successful completion of TORS. If a patient would benefit from complete surgical removal of the tumor but is not a good candidate for TORS, the surgeon should also possess the ability to perform TLMS and open surgery through pharyngotomy or mandibulectomy in order to individualize the procedure to the patient and tumor and maximize the outcome.
Anesthesia
After proper patient selection and counseling, the patient arrives in the operating room and the airway needs to be secured. The tumor can impact the anesthesiologist’s access to the glottis and trachea. Communication with the anesthesiologist needs to be thorough and lucid, and it can be aided by a video of the nasopharyngoscopy of the airway. The communication includes a discussion of the choice of intubation technique and the tube, as well as the perioperative anesthesia issues of paralysis, pain control, blood pressure control, anticipated blood loss, length of procedure, and oxygen content of the anesthetic. The preoperative anesthesia briefing should include not only the preferred airway, but also a backup plan in the event of complications.
Approaches to securing the airway include intubation under general anesthesia or awake versus sedated intubation. Using either of these approaches, tracheal intubation can be achieved via a transoral, transnasal, or tracheostomy route. Our preferred intubation is a transoral route with the use of traditional laryngoscope or Glide scope (Verathon, Bothell, WA). The tube is secured to the lower lip on the side opposite the tumor with tape. Fiberoptic intubation can also be useful.
Deciding on the proper endotracheal tube entails choosing a tube that will not kink, is not readily flammable, and is of appropriate size and position to deliver anesthesia but not obstruct the surgeon’s view of the tumor. We typically use a wire-reinforced laser-safe endotracheal tube.
Taping the tube to the contralateral lower lip or suturing it to the contralateral lower dentition or labial mucosa can secure the tube. Airway fire is a risk in transoral surgery, and an airway fire can occur in the presence of the fire triad: a fuel source (the tube), an ignition source (cautery, laser), and an oxidizer (oxygen). Before beginning the procedure, the surgeon should communicate with the anesthesiologist that the FiO2 should be kept below 30 % throughout the transoral surgery to decrease the chance of a fire [18]. A suction placed in the mouth can remove excess plume and also decrease the accumulation of oxygen in the event of a cuff leak around the endotracheal tube. The patient should be paralyzed during the transoral portion of the case to allow maximal exposure. Prophylactic antibiotics should be given prior to the start of the operation.
Transoral Surgical Equipment
After securing the airway, and administering endotracheal anesthesia, the surgeon should take advantage of the relaxed patient to perform a last thorough oropharyngeal exam. The tumor should be thoroughly palpated to confirm its mobility, and the surgeon should make a mental map of the confines of the tumor and their relationship to visual landmarks. Proper exposure of the tumor requires an oral retractor. This is most commonly done with a Feyh–Kastenbauer (FK) laryngeal retractor (Gyrus ACMI, Southborough, Massachusetts), or a Crowe-Davis retractor.
The most commonly used robotic instruments are the 5 mm EndoWrist Shertel grasper, the 5 mm Maryland dissector, and the 5 mm monopolar cautery (Intuitive Surgical). The assistant at the head of the table commonly uses a laryngeal suction, a suction cautery, endoscopic graspers, and endoscopic lip appliers. The assistant has a vital role in maintaining a smoke-free, dry, and clean working field, and can also assist in retraction and tumor exposure.
TORS Approach
Once intubation is complete, with the patient lying supine, the operating table is positioned so that the head is turned 90° from the anesthesia team [19] (Fig. 3). The teeth are protected with moldable thermoplastic sheeting (WFR/Aquaplast Corp., Wyckoff, NJ). The oral retractor is placed, and sometimes a trial of several blades and multiple angles is attempted to obtain the absolute best exposure of the tumor possible. The surgeon uses a headlight during retractor placement, and care is taken not to injure the teeth and lips during opening of the retractor. The retractor is then suspended to the left side of the bed using a Storz Laryngoscope holder (Karl Storz, Tuttlingen, Germany). The robot is now docked on the right of the patient with the base even with the head and slightly angulated away from the operating table. Three robotic arms are used for TORS: the camera is placed with the middle arm, the left arm is used to hold a dissector/grasper, and the right arm is used to hold the monopolar cautery. A 0° camera is mounted for lateral oropharyngectomy, and the 30° binocular camera is used pointing up for base of tongue resection. The arms are positioned so that the pivot point is at the level of the oral commissures, and the arms are placed so that they have the least chance of colliding. A red rubber catheter is placed transnasally and hooked to suction for smoke evacuation. A surgical assistant with experience in transoral surgery sits at the head with access to a variety of instruments including suction cautery, suction cannulas of various sizes, laryngeal graspers, bipolar cautery which is long enough to reach the base of tongue, and endoscopic vascular clip appliers. The monitors should be placed so that the surgical assistant and/or nurse/tech has easy visualization of the robotic surgical field. The assistant should have the ability to audibly communicate during the procedure so that repositioning of the robotic arms or retractor, manipulation of the tissue, clearance of blood and hemostasis, and other appropriate collaboration can occur.
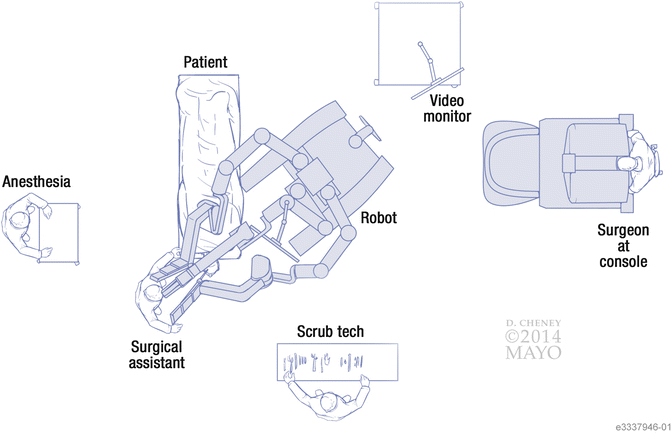

Fig. 3
Bird’s-eye view of the TORS setup in the operating room. Note the relationship of the TORS side cart to the edge of the bed, which allows proper positioning of the robotic arms. The surgical assistant sits at the head of the bed, while the surgeon sits at the console
Lateral Oropharyngectomy
Lateral oropharyngectomy can be performed with a headlight and electrocautery as has been described for decades, but it is enhanced by TORS by improved detail in the visualization and improved dexterity at the inferior extent of the tumor which often involves the base of tongue at the glossotonsillar sulcus [6]. With the tumor exposed and the robot docked, the procedure begins with a cut in the oral mucosa lateral and superior to the tumor near the retromolar trigone (Fig. 4). The incision is carried to the pterygomandibular raphe which serves as an anatomic landmark for the surgeon to develop the correct lateral dissection plane. Dissection is carried along the medial pterygoid fascia while retracting the tumor and the tonsil laterally. This action leads the surgeon into the plane between the superior pharyngeal constrictor, which will be taken with the specimen as a deep margin, and the buccopharyngeal fascia which will remain and separate the oral cavity from the parapharynx [16, 17, 20] (Fig. 5). Recall that the pterygomandibular raphe separates the superior pharyngeal constrictor from the buccinators muscle as it runs from the hamulus of the medial pterygoid plate and the mandible. At this point, it is helpful to make a medial incision through the palate mucosa and musculature and continue this incision inferiorly through the palatopharyngeus muscle (posterior tonsillar pillar) and pharyngeal mucosa and superior pharyngeal constrictor medially. Making these medial defining incisions at this point prevents the surgeon from continuing the dissection and excision too far medially as the tumor is rolled from lateral to medial during the final stages of removal. Inferior cuts can be made inferior to the tumor border and including the glossotonsillar sulcus and lingual tonsil as necessary for adequate tumor removal.
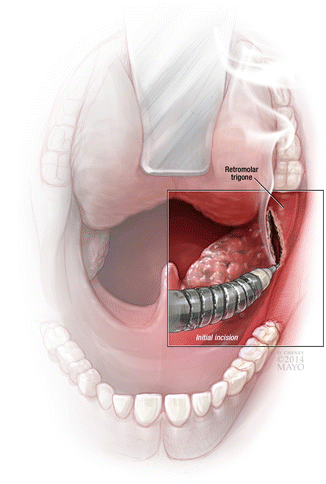
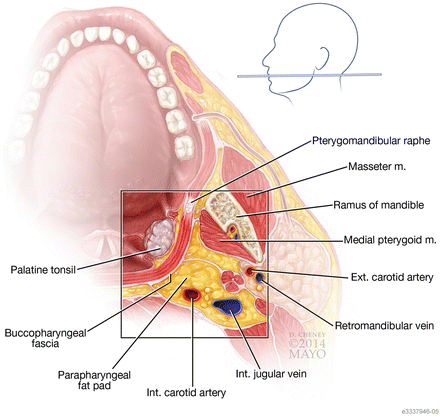

Fig. 4
Initial transoral oropharyngectomy cut performed lateral to tumor allows dissection down to the medial pterygoid fascia and allows proper plane of dissection onto buccopharyngeal fascia

Fig. 5
Axial cut showing the important anatomical relationships the head and neck surgeon needs to be aware of as he or she begins to dissect out the tumor. Special attention should be paid to the relationship of the buccopharyngeal fascia, the parapharyngeal fat, and the internal carotid artery
The most critical portion of tumor removal is the lateral dissection, as this is where the majority of the vascular supply to the tonsil from the lingual and facial arteries enters the specimen [16] (Fig. 6). The correct plane of dissection is critical at this portion of the operation. After establishing the lateral border of dissection as the buccopharyngeal fascia and the medial specimen with the palatoglossus, palatopharyngeus, and superior pharyngeal constrictor muscles, the surgeon dissects along the buccopharyngeal fascia, often bluntly with the spatula of the cautery. Dissection continues inferiorly with some medial tension on the specimen provided by the grasping instrument. Often this instrument is used gently as a retractor, as grasping a friable tumor multiple times will just lead to more bleeding and fragmentation of the specimen. Proceeding inferiorly the obliquely oriented styloglossus muscle and the vertically oriented stylopharyngeus muscle are encountered. The glossopharyngeal nerve may be visualized anterolaterally between these muscles. Numerous vascular contributions from the lingual, facial, and ascending pharyngeal vessels of various diameter may be encountered at this location and can be controlled by vascular clips applied by the assistant. If the facial or lingual arteries are encountered, they should be managed by multiple vascular clips. If bleeding is encountered that obscures vision, the surgeon can grasp the vessel with the robotic grasper to tamponade the vessel, and the assistant can apply pressure to the external neck at the level of the greater cornu of the hyoid bone. The styloglossus and stylopharyngeus muscles superiorly provide a medial barrier between the surgeon and internal carotid artery, and its pulsations and that of the external carotid artery can be seen transmitted through these structures and the parapharyngeal fat deep to the buccopharyngeal fascia. Inferiorly, the styloglossus and stylopharyngeus muscles can be transected t where they travel between the superior and middle pharyngeal constrictor to free the specimen. Blunt dissection at this level to free the muscles can prevent injury to the lingual artery. As the specimen is freed by final inferior cuts at the base of tongue and medial cuts at the medial superior pharyngeal constrictor, the surgeon and assistant should confirm orientation of the specimen that is not lost when removing the tumor through the mouth. It should be recognized that oncologic effectiveness is not necessarily improved by intact removal of the specimen. If at any time the volume of tumor prevents proper exposure of the operative area, the tumor can be transected and removed in sections, as is common in TLMS. The assistant grasps the tumor and the surgeon releases it. The tumor is then removed with orientation intact and placed on a towel and marked appropriately to prepare it for examination by the pathologist. The surgeon should visually inspect and palpate the specimen to confirm complete tumor removal and recognize any areas where the margins of resection may not be complete. The oropharynx is then carefully inspected for bleeding vessels, communication with the neck through the buccopharyngeal fascia, and any suspicious areas that might contain residual tumor. The robot arms are removed and the oral retractor is completely released to relieve venous congestion of the tongue while the tumor is being inspected by the pathologist.
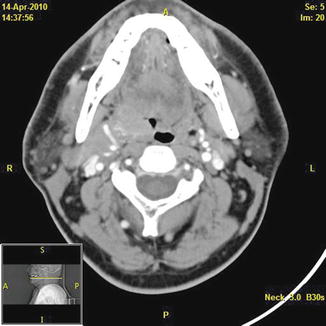

Fig. 6
Axial CT scan with contrast demonstrating a right-sided tonsillar carcinoma and the adjacent facial artery with multiple vascular feeders lateral to the tumor. The surgeon will encounter the most robust vascular supply to the tumor at its lateral borders, and these vessels usually come from the ipsilateral facial and lingual artery
Base of Tongue Resection
It is important for the surgeon to perform a careful physical examination of the BOT tumor prior to transoral excision, as it allows the surgeon to create a 3D image of the tumor and correlate it with preoperative imaging if available, as well as assess the tumor for mobility. This should be done before the retractor is placed, as the anatomy of the oropharynx can become distorted. With retractor placement, one should try to provide the widest tumor exposure, as this step is crucial for successful tumor removal (Fig. 7). The placement of a tongue stitch or applying tongue retraction with a sponge before mouth gag or laryngoscope insertion can help the surgeon with tongue retraction in order to obtain deeper access. Both the anterior edge and most of the inferior extent of the tumor should be visualized, and adjustments may need to be performed throughout the procedure to maintain maximum exposure [11].
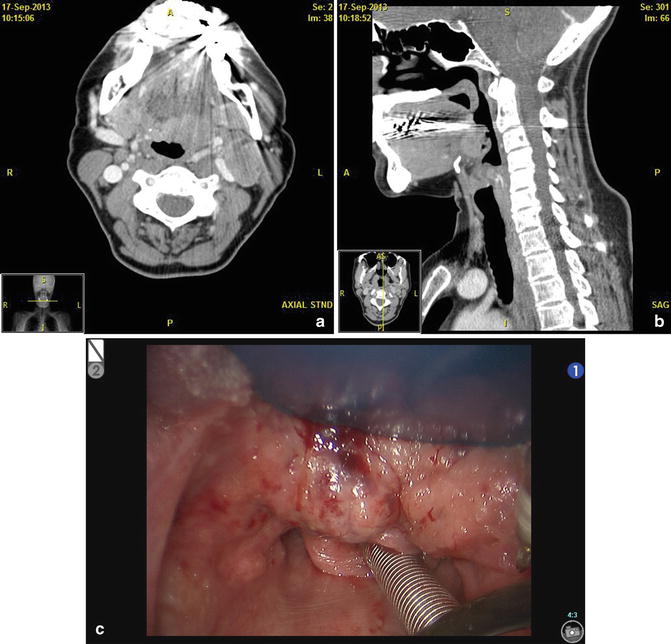

Fig. 7
The surgeon can use both physical exam and imaging to help create a mental image of the tumor to aid with operative planning. (a) Axial CT scan with contrast showing a left well-encapsulated lingual mass abutting the facial artery. (b) Sagittal CT scan showing that the tumor does not extend beyond the hyoid bone, making it a good TORS candidate. (c) Intraoperative view of the tumor
With exposure maximized, the surgeon can make a vertical cut through the tumor, exposing its greatest depth. This is useful since tangential cuts (to the tumor) across the BOT can fail to include the full tumor depth and unintentionally leave tumor within the deep intrinsic tongue musculature (Fig. 8). For the first step, the initial mucosal cuts are made anteriorly, allowing the tumor to fall posteriorly into the operative field. In order to recognize the inferior extent of the tumor throughout dissection, the first inferior cut should be made at the level of the vallecula. For counter-traction along the plane of dissection, the surgeon can retract along the healthy mucosa of the anterior margin. Tumor extent helps to guide the placement of both the medial and lateral cuts. Again, if the tumor is too large to see the borders for these cuts, transection of the tumor and removal in sections can be performed. Dissection is then carried out either in an anterior to posterior, or superior to inferior direction through the mucosa, lymphoid tissue, and tongue musculature, while making sure to preserve a cuff of normal tissue encircling the tumor. Once the tumor is freed, it is grasped by the surgical assistant, oriented, and passed along to pathology for frozen section analysis. The surgeon should again inspect the defect for bleeding and suspicious tissue, and then release the retractor while the specimen is being evaluated.
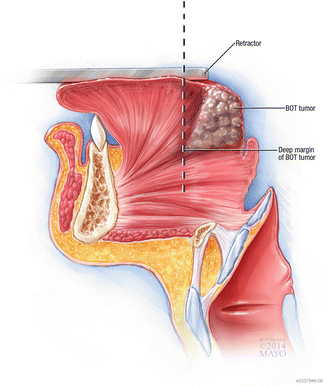

Fig. 8
The initial mucosal cut is made anteriorly in BOT lesions, allowing the tumor to fall posteriorly. This provides proper visualization throughout the procedure. It is important to palpate the deepest extent of the BOT tumor before removal. The placement of the retractor can limit tumor excision depending on the tumor’s extent, and if this is the case, an open surgical technique may be required over a transoral one
During the procedure, the surgeon should be aware of the dorsal lingual artery laterally along with contributions of the lingual artery. This is especially true for tumors that extend deeply and laterally beyond the hyoid bone, reaching the hyoglossus muscle, where both the lingual artery and hypoglossal nerve are in close proximity (Fig. 9). For adequate hemostasis, these vessels should be clipped with two to three clips on the patient’s side, and one clip on the tumor side. As described above for bleeding from the lingual artery during lateral oropharyngectomy, external pressure in the area of the greater cornu of the hyoid bone can be applied to slow blood flow until a clip can be applied. For tumors with this type of lateral extent, it may be safer to proceed with an external approach and allow for more confident resection of margins.
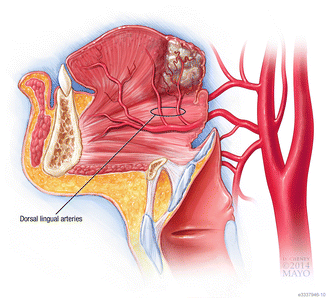

Fig. 9
Sagittal view showing the important vasculature that the head and neck surgeon can encounter during the excision of a BOT lesion, with the dorsal lingual artery and its branches being the most relevant
Margin Analysis
The entire oncologic goal of TORS for OP SCCa is complete microscopic margin clear resection of the cancer. As has been affirmed for decades, the most common cause of patient mortality from cancer is the failure to completely eradicate the tumor at its primary source [21]. The best opportunity to accomplish this momentously important task is at the primary source. If the surgeon waits for final pathology confirmation and finds that the permanent margins are positive, it can be difficult, if not impossible, to correlate that positive margin with its original location in the oropharynx. Consequently, it is mandatory that an experienced frozen section pathologist is available as a member of the operative team. Equally important is the ability to communicate with that pathologist directly. The frozen section pathology result is only as good as the weakest length in that communication chain. The specimen needs to be removed, oriented, transported to the pathology lab with no loss of orientation, inked at the appropriate border, cut, placed on the chuck, cut with a microtome, placed on the slide, stained, and then examined by the pathologist for tumor at the cut margin. This multistep process needs to be performed for the medial, lateral, superior, inferior, and deep edges of the specimen (Fig. 10). Any of these specimens that have tumor at the margin need to be correlated to the appropriate location in the operative bed, and a new resection margin needs to be excised and the process repeated. Obviously there are multiple hazards in this process including loss of orientation, failures in communication, and errors in inking and cutting that could result in a false positive or negative margin. In our institution, the surgeon who removes the specimen examines it grossly and transports it to the pathologist directly and has a face-to-face communication with the pathology team to ensure that the communication link is as strong as possible. In this manner, we have been able to achieve a 99 % correlation with frozen and final pathology and a 99 % local control rate for OP SCCa resected by transoral surgery. The surgeon should continue to clear margins by repeated resection until all margins are microscopically clear of tumor, or a structure is encountered that cannot be safely resected.
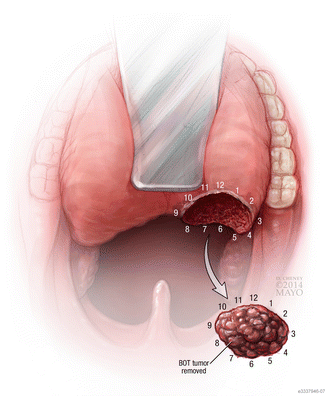

Fig. 10
For both tonsillar and BOT tumors, it is important that the surgeon maintain proper orientation of the specimen as it is passed on to pathology for frozen section. This ensures that any margins that are positive can be accurately re-excised
It has been a point of contention as to how much normal tissue should be excised along with the primary tumor, with suggested margins ranging from 1 mm to up to 10 mm of normal tissue [22]. Although in theory these values may ring true, practically they are less plausible. Oropharyngeal tissue contracts as much as 50 % after being removed and thus a 5 mm margin can become only a 2 mm margin once it reaches the pathologist. If one were to take a deep margin any greater than 2.4 mm in the tonsillar fossa, one would sacrifice the major vessels within the parapharyngeal space. Hinni analyzed 128 tonsillar carcinomas that were treated transorally where the average deep margin was only 1.98 mm, but he still found the 5-year local control rate to be 99 % [21]. We agree with Hinni that a clear microscopic margin is the goal, and that this goal holds up to equal oncologic effectiveness for transoral resection of OP SCCa whether the margin is 1 mm or 10 mm.
Revision Surgery
The decision to use TORS as a treatment option goes back to the initial description of patient candidacy. In the author’s experience the issue with TORS as salvage therapy is twofold:
1.
One needs to be able to obtain margins for adequate resection. One can imagine the primary previously untreated tumor as being contained in a box. Thus during resection, when negative margins are obtained, one can be fairly certain the tumor in its entirety has been removed. Once a patient has had chemoradiation, the tumor is no longer confined to a “box,” instead there are now islands of tumor leading to the possibility of obtaining negative margins around one island, but leaving residual tumor behind. A similar issue is seen when tumors begin to invade lymphatics or neurovascular structures and begin to spread outside of its “box.”
2.
The majority of TORS defects are left to heal by secondary intention. Some authors advocate vascularized flaps in patients who have been chemoradiated, but again this somewhat defeats the purpose of the minimally invasive approach. Patients with a history of prior chemoradiation have an increased risk of developing non-healing wounds and wound infections. In patients with TORS as the primary treatment, the chance of a cure approaches 90 % (14), whereas in patients where TORS is a salvage treatment, the chance of a cure without significant morbidities drops to 70 % [15].
Neck Dissection
Neck dissections are often part of the management for oropharyngeal malignancies, and either ipsilateral or bilateral neck dissections can be performed. If the patient has N0-N2b or N3 disease, they typically undergo an ipsilateral select neck dissection (levels II–IV). The submandibular gland can be retained without any loss of oncologic effectiveness in the neck disease, and this structure can be very helpful in reinforcing the thin tissue between the oropharyngeal dissection and the neck dissection to reduce fistula [23]. The retropharyngeal nodes can be involved in oropharyngeal SCCa if there is significant invasion of the tumor into the soft palate or posterior pharyngeal wall. If these areas are involved, then careful review of the radiographs and palpation of this area should ensue. In the presence of radiographic or clinical evidence of retropharyngeal nodal adenopathy, then the retropharyngeal nodes should be removed. These nodes lie just medial to the internal carotid artery and are often in contact with its medial border. They usually number between 1 and 3 [24]. The nodes can be removed directly through the transoral defect where they are often readily accessible, or the surgeon can dissect up through the neck dissection along the medial surface of the internal carotid artery while retracting the hypoglossal nerve and digastric muscle superiorly. Dissection of the retropharyngeal nodes can lead to a communication between the neck and oropharynx that may need to be closed by suturing the retropharyngeal fascia to the edge of the remaining superior pharyngeal constrictor muscle.
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


