Purpose
To characterize spontaneous dislocation of the fluocinolone acetonide sustained release implant (Retisert; Bausch & Lomb) drug pellet from its strut.
Design
Retrospective, observational case series.
Methods
The medical records of 224 patients in whom 407 fluocinolone acetonide implants were placed by either of 2 surgeons between 1998 and 2012 were reviewed for drug pellet–suture strut separation, observed before surgery in clinic or occurring at the time of implant replacement.
Results
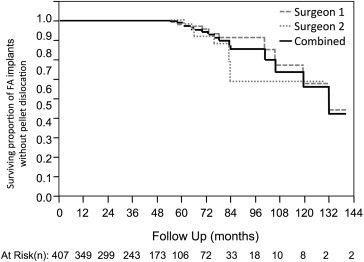
Seventeen (4.2%) of 407 drug pellets spontaneously dislocated away from the suture strut at a mean postimplantation time of 77.4 months (range, 33 to 132 months). The rate of spontaneous dislocation increased from 1 dislocation per 262 person years (95% confidence interval, 1 per 1269 to 1 per 89.6 person-years) before 60 months to 1 dislocation per 13.3 person-years (95% confidence interval, 1 per 24.4 to 1 per 7.9 person-years) afterward. Fourteen of 17 dislocated pellets were removed surgically. Of 77 exchanged fluocinolone acetonide implants without dislocated pellets, 11 (14.2%) pellets had separated, but not dislocated away, from the strut; time to exchange of dislocated or dissociated pellets was significantly longer than time to exchange of intact fluocinolone acetonide implants (70.5 vs 44.9 months; P < .001).
Conclusions
The fluocinolone acetonide drug pellet can dislocate spontaneously from the suture strut as a late event that typically occurs after 3 years. Pellets also may be dissociated from the strut without dislocation, when separation occurs at the time of surgical replacement. Removal of the dislocated pellet can be achieved safely. Time was the major risk factor identified for both events.
The fluocinolone acetonide intravitreal implant (Retisert; Bausch & Lomb, Rochester, New York, USA) is an implanted nonbiodegradable intravitreal sustained drug-delivery system that delivers a steady dose of fluocinolone acetonide for approximately 3 years. The sustained-release intravitreal implant is approved by the Food and Drug Administration for the treatment of chronic noninfectious uveitis, and it has been used off label for diabetic macular edema and cystoid macular edema secondary to retinal vein occlusion. The implant delivers the synthetic corticosteroid fluocinolone acetonide at rate of 0.6 μg/day for the initial month and then 0.3 to 0.4 μg/day for approximately 30 months. In a phase 3 trial that established its efficacy, the fluocinolone acetonide implant reduced the uveitis recurrence rate from 54.6% in the 34 weeks before insertion to 6.4% in the 34 months after placement. The most common side effects reported included intraocular pressure elevation in 67% and cataract in 93%.
The fluocinolone acetonide implant comprises 2 parts joined together. The pellet comprises 0.59 mg of fluocinolone enclosed in a silicone elastomer cup with a semipermeable release orifice; this assembly is attached to a polyvinyl alcohol suture tab, or strut, by a silicone-based adhesive. The dimensions of the suture strut are approximately 3 × 2 × 5 mm, and the pellet is 1.5 mm in diameter.
The drug-containing cup (hereafter called the pellet ) can dislocate spontaneously from the strut into the vitreous cavity, as described in 2 reports including a total of 5 patients. Two patients had complications of pellet dislocation, including 1 case of commotio retina and retinal tear and 1 case of anterior dislocation and corneal decompensation. All dislocation events occurred between 3 and 6 years after implantation, but no information regarding the rate of this occurrence was reported.
The pellet also can become separated from the strut, but not dislocated into the vitreous cavity; this occurrence usually is seen at the time of fluocinolone acetonide implant exchange, and less commonly on preoperative examination. Nicholson and associates analyzed a consecutive series of 27 fluocinolone acetonide implant exchanges and found that implants in which the pellet became separated from the strut during the exchange had resided in the eye longer than those that remained intact on removal (47.4 vs 32.5 months, respectively; P = .0032). The authors also identified 1 case of retinal tear and 1 limited suprachoroidal hemorrhage that resulted from the retrieval of a dissociated pellet that fell posteriorly during exchange. In the present study of a large series of implants performed by 2 surgeons (G.J.J., D.C.), we further characterized the rate and clinical significance of dislocation and separation without dislocation of the pellet from the strut in eyes implanted with sustained-release fluocinolone acetonide implants.
Methods
Approval for this 2-center study was obtained from the Duke University Institutional Review Board for the subjects treated by one of the authors (G.J.J.), who managed patients at Duke University, and from the Western Institutional Review Board for the remaining subjects, who were treated by one of the other authors (D.C.) at the Texas Retina Associates center. This study was a retrospective, observational case series. The charts of all completed cases of fluocinolone acetonide implant insertions and exchanges performed by 2 surgeons (G.J.J., D.C.) between 1998 and 2012 were reviewed. Demographic information, indication for surgery, date of implantation, location of implant, prior vitrectomy, additional combined procedures performed contemporaneously with fluocinolone acetonide implant insertion, and available follow-up were recorded for all cases. The follow-up date for each implant was established as either date of surgical removal from the eye or at the last documented retinal examination when the implant was visualized. Patients without at least 1 month of follow-up were excluded from analysis.
Patient charts were reviewed for any evidence of separation of the pellet from the strut on clinical examination; these events were defined as pellet dislocation. Surgical reports of all fluocinolone acetonide implant exchanges also were reviewed. All cases in which the pellet–strut separation was identified during surgery (which could include those instances when the separation occurred, but was not observed before surgery, or when it occurred during implant removal) were defined as pellet dissociation.
For all cases of pellet dislocation, further information was collected, including timing of the dislocation, initial visual acuity on diagnosis of dislocation, associated ocular complications, management, and final visual acuity. For eyes that underwent surgical removal of the dislocated implant, the surgical technique was reviewed. For all cases of pellet dissociation, operative complications were reviewed based on surgical reports and postoperative examinations.
Statistical Analysis
Eyes with spontaneous pellet dislocation were compared with those without spontaneous pellet dislocation, and eyes with pellet dissociation were compared with those that underwent exchange without dissociation or dislocation. Categorical variables were analyzed with the Fisher exact test and continuous variables were analyzed with a 2-tailed t test. Visual acuity as measured by Snellen or Early Treatment Diabetic Retinopathy Study vision charts were converted to the logarithm of the minimal angle of resolution scale to calculate means, and visual acuities at 2 different time points were compared using a paired t test. Kaplan-Meier survival curves were constructed to demonstrate the occurrence of spontaneous dislocations in all patients. A Cox proportional hazard model was used to adjust for available follow-up when comparing hazard ratios of various risk factors for pellet dislocation. All calculations were completed using JMP Clinical software (SAS Institute, Cary, North Carolina, USA).
Results
Patients
A total of 407 fluocinolone acetonide implants in 224 patients were included for analysis ( Table 1 ). Two patients were excluded because of insufficient follow-up. Patients were predominantly white 145 (65%) and women 158 (70%). The indication for fluocinolone acetonide implant placement was most commonly posterior noninfectious uveitis 338 (83%), although a substantial percentage was inserted for the treatment of cystoid macular edema after retinal vein occlusion 32 (14%), retinitis pigmentosa 4 (1%), or diabetes 4 (1%).
| All Implants | Spontaneous Dislocation | Nondislocated | Dissociated Pellets at Exchange | Intact Implants at Exchange | P Value | ||
|---|---|---|---|---|---|---|---|
| Dislocated Versus Nondislocated | Dissociated Versus Intact | ||||||
| Total no. of implants | 407 | 17 | 390 | 11 | 66 | ||
| Total no. of patients | 224 | 15 | 221 | 10 | 47 | ||
| Demographics | |||||||
| Male | 66 (30) | 6 (40) | 60 (27) | 2 (20) | 15 (32) | .371 | .796 |
| White | 145 (65.0) | 12 (80) | 133 (60) | 6 (60) | 38 (80.8) | .173 | .213 |
| Age (SD) at implantation (y) | 50.2 (16.5) | 38.5 (16.9) | 50.7 (16.3) | 42.3 (16.9) | 50.4 (14.3) | .003 a | .094 |
| Surgery factors | |||||||
| Right eyes | 194 (47.7) | 9 (52.9) | 185 (47.4) | 6 (55) | 34 (51.5) | .805 | 1.00 |
| Uveitis as indication | 338 (83.0) | 13 (76.5) | 326 (83.5) | 9 (81.8) | 57 (86.4) | .502 | .653 |
| Vitrectomized eye | 107 (26.3) | 4 (23.6) | 103 (26.4) | 2 (18.2) | 14 (21.2) | 1.00 | 1.00 |
| Inferonasal location | 215 (52.6) | 8 (47.1) | 207 (52.8) | 7 (63.6) | 37 (56.1) | .805 | .749 |
| Inferotemporal location | 168 (41.3) | 8 (47.1) | 160 (41.0) | 4 (36.4) | 24 (36.4) | .624 | 1.00 |
| Time to (mo) | Final follow-up | Dislocation | Final follow up | Exchange | Exchange | ||
| Mean (SD) | 44.4 (28.0) | 77.4 (24.9) | 43.0 (27.3) | 65.9 (20.7) | 44.9 (15.6) | <.001 a | <.001 a |
| Median | 41.5 | 72.7 | 40.4 | 67.2 | 40.2 | ||
| Minimum to maximum | 1.1 to 140.2 | 33.9 to 131.9 | 1.1 to 140.2 | 37.5 to 104.8 | 5.5 to 89.9 | ||
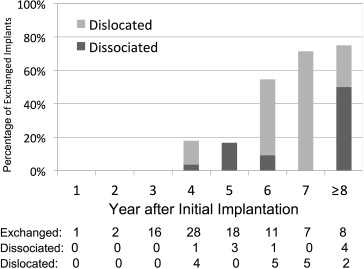
Spontaneous Pellet Dislocations
A total of 17 (4.2%) dislocated pellets in eyes of 15 patients were identified ( Table 2 ). Dislocation occurred at a mean of 77.4 months after implantation (range, 33.9 to 131.9 months; Figure 1 ). The most common indication for fluocinolone acetonide implant placement in these cases was posterior uveitis (12 [70.6%]). Mean logarithm of the minimal angle of resolution and median Early Treatment Diabetic Retinopathy Study or Snellen visual acuity at diagnosis were 0.69 (approximately 20/100) and 20/126, respectively. Visual acuity ranged from 20/25 to hand movements, and visual acuity was better than 20/60 in 7 eyes (41%). Patients observed a large floater in 12 of 17 cases.
| Patient No. | Indication for FA Implant | Age at Insertion (y) | Gender | Time to Dislocation (mo) | Clinical Course | Vision at Diagnosis | Indication for Surgery | Time to Surgery (mo) | Time Observed (mo) | Final Vision |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Uveitis | 38.6 | M | 64.5 | Immediate surgery a | 20/60 | Symptomatic | 0.86 | — | 20/80 |
| 2 | Uveitis | 38.1 | F | 75.3 | Immediate surgery a | 20/200 | Symptomatic | 0.20 | — | 20/200 |
| 3 | Uveitis | 44.4 | F | 65.8 | Immediate surgery a | 20/400 | Symptomatic | 0.03 | — | 20/200 |
| 4 | Uveitis | 11.4 | F | 60.8 | Immediate surgery a | 20/400 | Symptomatic | 0.43 | — | 20/200 |
| 4 | Uveitis | 11.9 | F | 82.9 | Immediate surgery a | 20/30 | Symptomatic | 0.95 | — | 20/25 |
| 5 | Uveitis | 46.8 | M | 83.3 | Immediate surgery a | 20/25 | Recurrence | 0.49 | — | 20/25 |
| 6 | Uveitis | 34.5 | F | 105.5 | Immediate surgery a | 20/160 | Symptomatic, recurrence | 1.09 | — | 20/160 |
| 6 | Uveitis | 37.5 | F | 69.8 | Immediate surgery a | 20/160 | Symptomatic, recurrence | 1.09 | — | 20/160 |
| 7 | Uveitis | 29.8 | F | 100.4 | Delayed surgery b | 20/100 | Symptomatic, alternate surgery c | 26.28 | — | 20/100 |
| 8 | CME, RP | 67.1 | M | 131.9 | Observed | 20/800 | — | — | 7.89 | 20/800 |
| 9 | Uveitis | 14.0 | F | 54.2 | Immediate surgery a | 20/25 | Symptomatic | 2.11 | — | 20/20 |
| 10 | Uveitis | 40.3 | F | 72.7 | Observed | 20/50 | — | — | 48.19 | 20/50 |
| 11 | Uveitis | 36.9 | F | 60.0 | Immediate surgery a | 20/25 | Recurrence | 0.3 | — | 20/32 |
| 12 | CME, CRVO | 70.2 | M | 33.9 | Delayed surgery b | 20/160 | Symptomatic, recurrence | 7.80 | — | 20/160 |
| 13 | Uveitis | 42.5 | F | 77.9 | Delayed surgery | HM | Alternate surgery, c recurrence | 24.61 | — | LP |
| 14 | CME, CRVO | 31.2 | M | 57.3 | Observed | 20/40 | — | — | 5.99 | 20/50 |
| 15 | CME, BRVO | 59.0 | M | 94.1 | Delayed surgery b | 20/126 | Recurrence | 25.20 | — | 20/160 |
a Immediate surgical removal within 3 months of diagnosis.
b Delayed surgical removal after 3 months of diagnosis.
c Surgical pellet removal combined with alternate surgery that was clinically indicated.
Fourteen (82.3%) of 17 dislocated pellets were removed surgically at a mean of 6.5 months after diagnosis (range, 0 to 26.3 months), most commonly because of patient symptoms or recurrence of inflammation or cystoid macular edema ( Figure 2 ). Ten were removed within 3 months of diagnosis, whereas the eyes of the remaining 7 patients initially were observed. Of these 7, 4 implants eventually were removed surgically at time points ranging from 7 to 26 months after the initial diagnosis of pellet dislocation. Visual acuity at the final examination was not statistically different than preoperative visual acuity in any group.
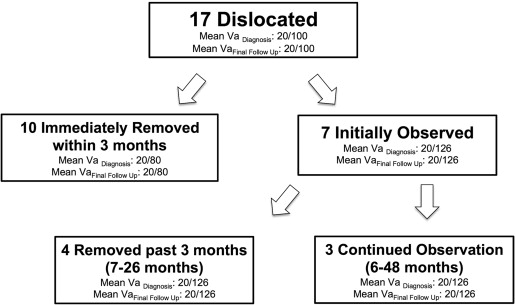
Observation of Dislocated Pellets
A total of 7 eyes with pellet dislocations were observed for at least 6 months after pellet dislocation diagnosis. Three dislocated pellets were observed on average for 20.7 months and were not surgically removed. In 2 cases, the dislocated pellet remained lodged in the peripheral vitreous and the patient remained asymptomatic with stable visual acuity, better than 20/50 in the eye. In the third case, the dislocated pellet was freely mobile in the vitreous cavity; however, the visual acuity in that eye was poor (20/800) and the patient was asymptomatic.
An additional 4 eyes with dislocated pellets were observed for at least 6 months (range, 7 to 26 months; mean, 21.2 months) before pellets were removed surgically. Two eyes, one diagnosed with branch retinal vein occlusion and another with central retinal vein occlusion, developed recurrent cystoid macular edema, which prompted removal of the dislocated pellet and placement of a new fluocinolone acetonide implant. In the third eye, after 24 months, the pellet dislodged from the peripheral vitreous into the central vitreous cavity causing a symptomatic floater. The pellet then was removed surgically and a glaucoma tube was inserted to improve control of intraocular pressure. In the fourth eye, an eye with multiple sclerosis-associated panuveitis and hand movements visual acuity, the pellet remained in the peripheral vitreous, but the eye developed recurrent cystoid macular edema and a subluxated intraocular lens. These events prompted removal of the dislocated pellet and suture strut, placement of a new fluocinolone acetonide implant, and removal of the intraocular lens.
There were no retinal tears, detachments, or other forms of retinal injury seen over the period during which the dislocated pellets were observed. In 2 of 7 eyes, visual acuity declined by 1 to 2 lines. However, in those 2 eyes, it was unclear whether the visual acuity decline was related directly to the dislocated pellet or to macular edema that recurred because the implant was drug depleted.
Surgical Removal of Dislocated Pellets
Removal of dislocated pellets was accomplished by 1 of 2 techniques. In the first approach, an infusion chandelier first was placed inferiorly, opposite the side of the original fluocinolone acetonide insertion site, and a 20-gauge microcannula was inserted superotemporally through the pars plana, on the same side as the infusion chandelier. The infusion was begun, a scleral incision was created at the site of the original implant, and then the original suture strut to which the pellet had been attached before it dislocated was removed. The scleral incision then was closed partially with 7-0 Vicryl (Ethicon, Somerville, New Jersey), leaving enough space for a 20-gauge vitrectomy probe. A wide-field viewing system was used to view the posterior segment. A vitrectomy then was performed; the vitrectomy probe was inserted through the partially closed scleral incision. A soft-tipped aspiration cannula was inserted through the superior microcannula, and a 20-gauge diamond-dusted forceps then was inserted through the partially closed scleral incision. The soft-tipped aspiration cannula then was used to elevate the dislocated pellet, which was passed to the diamond-dusted forceps held in the other hand. As the pellet was brought toward the scleral incision, the infusion pressure was raised to maintain the globe pressure, and the assistant then cut and removed the preplaced 7-0 Vicryl suture. Next, the pellet was removed from the eye with the diamond-dusted forceps. Finally, the scleral wound was then closed with interrupted 9-0 nylon sutures and the knots were buried.
For the second technique, a 23-gauge infusion line was placed inferiorly, opposite the original implant site, and one other 23-gauge microcannula was placed in a superior quadrant opposite the implant site. A scleral incision was created at the site of the original implant, and the strut was removed as described above. The scleral incision then was closed temporarily with a single 7-0 Vicryl suture. Pars plana vitrectomy was performed with a 23-gauge vitrectomy probe and a separate endoilluminating light pipe. The Binocular Indirect Ophthalmoscope Microscope (BIOM, Oculus Surgical, Port St. Lucie, Florida) system was used to view the posterior segment during this procedure. A 23-gauge soft-tipped aspiration cannula was used to elevate the dislocated pellet safely into the pupillary plane, at which point the Binocular Indirect Ophthalmoscope Microscope lens was rotated out of the surgical field and the pellet was observed directly with the operating microscope. The pellet then was grasped with a 20-gauge diamond-dusted foreign body forceps through the scleral incision and removed as described above. Finally, the scleral wound was closed with interrupted 9-0 nylon sutures.
The method to elevate the dislocated fluocinolone acetonide implant with a soft-tipped cannula was adopted when initial attempts to grasp the dislocated pellet directly with forceps were unsuccessful because of rotation of the pellet away from the forceps. For both techniques, infusion was maintained during all surgical maneuvers.
No posterior segment complications occurred during removal of dislocated pellets. By 3 months, visual acuity returned to preoperative levels in 11 of 14 eyes. No eyes had a 3-line or more visual acuity loss.
Pellet Dissociations
A total of 91 fluocinolone acetonide implants were exchanged in 64 patients at a mean of 52.0 months after initial implantation (range, 5.5 to 119.3 months; Figure 3 ). In addition to the 14 implants known to have pellet dislocations by clinical examination before exchange, in 11 eyes, the pellet–strut separation was identified only during surgery. In this latter instance, termed dissociation , toothed forceps usually were placed through the wound to grasp and remove the pellet. Westcott scissors were used to excise any vitreous attached to the pellet that presented through the wound. In one case, the pellet was left lodged into the vitreous base. No cases occurred of posterior dislocation of the pellet during removal of the dissociated pellet or other surgical complications, such as retinal tear or detachment.