Purpose
To evaluate the influence of Valsalva maneuver on the morphology and thickness of the choroid at the macular area.
Design
Prospective interventional case series.
Methods
Institutional setting. Nine healthy volunteers performed macular spectral-domain optical coherence tomography using enhanced-depth imaging at rest and during a Valsalva maneuver. Horizontal and vertical B-scans centered on the fovea were acquired. Subfoveal and average choroidal thickness in the central 3 mm were compared in the resting position and during the Valsalva maneuver using manual and semiautomatic measuring tools. Changes in choroidal thickness were evaluated.
Results
There was no statistically significant difference in choroidal thickness at rest or during Valsalva maneuver in any of the compared groups. The subfoveal thickness difference was −4.1 μm on horizontal scans ( P = .28) and 1.4 μm on vertical scans ( P = .75). The mean choroidal thickness difference in the central 3000 μm was 8.5 μm on horizontal scans ( P = .73) and −5.3 μm on vertical scans ( P = .41).
Conclusions
Valsalva maneuver does not change choroidal thickness at the posterior pole. The increase in ocular pressure caused by this maneuver cannot be explained by an increase in choroidal thickness in this portion of the uveal tract.
The Valsalva maneuver occurs when a forced exhalation is performed against a closed glottis. This causes an increase in intrathoracic venous pressure and a sustained decrease of the venous flow into the thoracic cavity, leading to an increase in the venous pressure in the head and neck. The increase in intrathoracic venous pressure is transmitted through the jugular, orbital, and vortex veins to the choroid. This has been postulated to cause choroidal vascular engorgement and an increased choroidal volume that ultimately leads to an increase in intraocular pressure (IOP). Valsalva maneuver has been shown to transiently increase IOP in special circumstances such as weight lifting and isometric exercises. In people who repeatedly perform Valsalva maneuvers, such as musicians who play wind instruments, visual field loss has been reported and attributed to this repeated phenomenon.
The mechanism for IOP rise has not been completely explained. One hypothesis is that the increase in systemic venous pressure may lead to a decrease of the ocular venous flow through the vortical veins and a consequent increase in choroidal volume and intraocular pressure. Others have related this IOP increase with an increase in episcleral venous pressure.
Shuman and associates demonstrated that, in patients performing Valsalva maneuver while playing high-resistance wind instruments, there was an increase in thickness of the choroid that is situated right posterior to the pars plana of the ciliary body. Measurements were performed with ultrasonic biomicroscopy at the posterior part of the ciliary body before and during wind instrument blowing. The authors hypothesized that this increase in the uveal tract would occur throughout the whole globe. This increase in the uveal thickness would lead to an increase in the IOP.
Recently, with the advent of spectral-domain optical coherence tomography (OCT), the ability to image the choroid at the posterior pole of the eye has increased. Spaide and associates described a technique called “enhanced depth imaging” using the Heidelberg Spectralis spectral-domain OCT imaging system (Heidelberg Engineering, Heidelberg, Germany). By pushing the OCT closer to the eye, one is able to obtain an inverted image that penetrates deeper into the choroidal tissue, allowing a cross-sectional image of the full choroidal thickness at the posterior pole.
With this technique it is possible to investigate choroidal thickness changes that occur at the posterior pole in various physiologic and pathologic conditions. The choroidal thickness is increased in diseases such as Vogt-Koyanagi-Harada disease and central serous chorioretinopathy and after the intake of sildenafil.
Using this method, the authors studied the effect of the Valsalva maneuver on choroidal thickness at the posterior pole in order to better understand the mechanisms of IOP rise associated with the maneuver.
Methods
This was a prospective interventional case series performed in an institutional setting. Eighteen eyes of 9 young, healthy volunteers were included in this study.
The volunteers had no history of systemic or ocular diseases (except for refractive error). However, as high myopia has been shown to influence choroidal thickness, no one with myopia greater than 6.0 diopters was accepted as a volunteer. Subjects were excluded if they had any physical disability that would prevent performing the Valsalva maneuver. All participants underwent a full ophthalmic examination including visual acuity, slit-lamp evaluation, IOP measurement by Goldmann tonometry, and macular and optic disc examination to exclude ocular pathology.
Each volunteer was submitted to 2 perpendicular (horizontal and vertical) OCT B-scans, centered on the fovea, with the Spectralis Heidelberg apparatus.
The Spectralis scans were single 30-degree B-scans centered on the fovea averaged 100 times. These B-scans were then used for reference, using the “follow-up” function of the Spectralis, for the following scans.
An enhanced depth imaging scan averaged 100 times was performed in both perpendicular B-scans in a resting position. Finally, the volunteer engaged in a Valsalva maneuver by blowing forcefully against his hand while occluding the nose with the index finger and thumb. After 15 seconds of expiratory pressure, the enhanced depth imaging scans averaged 100 times were repeated on the same choroidal section. The Valsalva maneuver was maintained while the scan was being acquired by keeping the expiratory pressure against the hand. After a brief rest, the volunteer engaged in a second Valsalva maneuver and the remaining perpendicular scan was acquired. All the scans were carried out in the sitting position.
Only images with a signal quality greater than 25 dB were included in the study.
Choroidal thickness was compared in each eye before and during the Valsalva maneuver both in vertical and horizontal scans using a semiautomatic method and a manual method.
In the semiautomatic algorithm, the images were exported from the Spectralis as .tiff files and analyzed using NIH software ImageJ ( http://fiji.sc/wiki/index.php/Fiji , Accessed November 2011). Choroidal scans were cropped into standard 32-bit gray-level images. Manual segmentation was performed under a 200% magnification by placing an average of 30 points sequentially under the hyperreflective line of the retinal pigment epithelium. Additional points were placed sequentially at the inner scleral boundary ( Figure 1 ). Single subfoveal points were also selected. The xy coordinates were extracted and a polynomial regression (a + bx + cx + dx + yx ) using the least squares method was performed for each set of user-defined points. We obtained a function (f(x)) for the external boundary of the retinal pigment epithelium and a second function (f(x′)) for the inner scleral boundary. Normal and Valsalva maneuver segmented limits were graphically plotted using the obtained polynomial equations in Microsoft Excel. Subfoveal thickness, total thickness (f[th] = [f(x) − f(x′)]), and thickness variation (v[th] = [f(th1) − f(th2)]) of the central 3000 μm were calculated for resting scans and scans acquired during Valsalva maneuver. This method of measurements was classified as semiautomatic. In order to test the reproducibility of the semiautomatic algorithm, the method was repeated 5 times for the same image. To test the reproducibility of the Valsalva maneuver, a volunteer performed 5 successive Valsalva maneuvers.
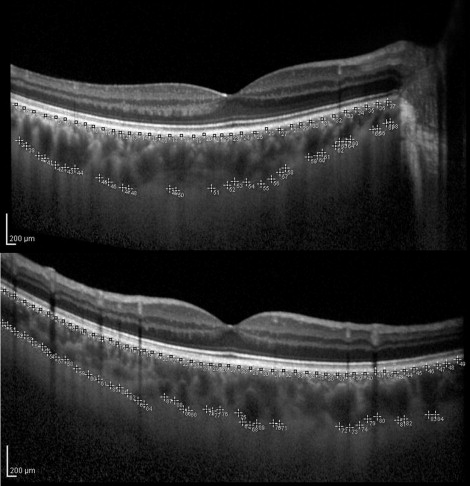
Subfoveal manual measurements were performed using the calipers provided in the Spectralis Heidelberg software. These were compared to the semiautomatic subfoveal points.
Investigators performing manual and semiautomatic choroidal thickness measurements were masked regarding the Valsalva maneuver status of the B-scan being evaluated.
Statistical analysis was performed with PASW Statistics (version 18.0 for MAC; IBM Corporation, Chicago, Illinois, USA). Changes in choroidal thickness were evaluated using paired sample t tests. Statistical significance was considered when P < .05. The intraclass correlation coefficient (ICC) of reproducibility was used as a summary statistic to quantify the test-retest reproducibility. A Bland-Altman plot was used to assess the inter-test variability between subfoveal semiautomatic and subfoveal manual measurements of choroidal thickness.
Results
Eighteen eyes of 9 healthy volunteers were analyzed. Volunteers were aged 25 to 38; 7 subjects (78%) were male.
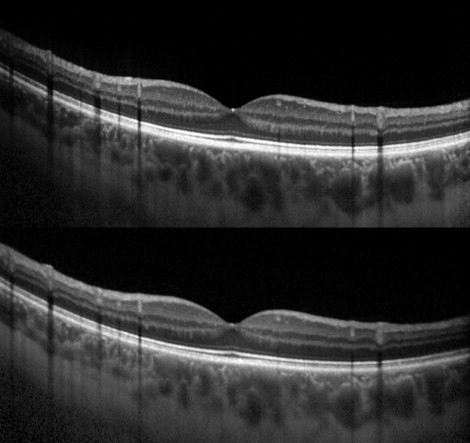
Valsalva maneuver did not have any significant influence on macular choroidal thickness. This was independent of the orientation of the scan and of the eye measured ( Figure 2 ).
When comparing all 36 baseline subfoveal choroidal thicknesses with the respective Valsalva maneuvers, using the semiautomatic method, the mean recorded change was −1.34 ± 16.9 μm ( P = .64). The subfoveal thickness difference was −4.1 μm on horizontal scans ( P = .28) and 1.4 μm on vertical scans ( P = .75). The mean choroidal thickness difference in the central 3000 μm was 8.5 μm on horizontal scans ( P = .73) and −5.3 μm on vertical scans ( P = .41). The main results are outlined in the Table .
| Choroidal Thickness (μm) | Semiautomatic Subfoveal | Manual Subfoveal | Mean Choroidal Thickness b (Central 3 mm) | |||
|---|---|---|---|---|---|---|
| Rest | Valsalva | Rest | Valsalva | Rest | Valsalva | |
| Horizontal | 323.9 ± 121.4 | 319.8 ± 113.9 | 312.9 ± 123.1 | 307.2 ± 120.2 | 301.2 ± 115.2 | 292.7 ± 111.8 |
| Vertical | 334.3 ± 129.3 | 335.7 ± 137.3 | 329.1 ± 116.7 | 324.8 ± 114.2 | 323.1 ± 114.4 | 328.4 ± 106.4 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


