Purpose
To investigate relationships among vitreous fluid levels of soluble vascular endothelial growth factor receptor-2 (sVEGFR-2), vascular endothelial growth factor (VEGF), and soluble intercellular adhesion molecule 1 (sICAM-1) in patients with branch retinal vein occlusion (BRVO) and macular edema or patients with idiopathic macular hole.
Design
Retrospective case-control study.
Methods
setting: Tokyo Women’s Medical University and Eguchi Eye Hospital. patient population: Forty-nine Japanese patients who underwent unilateral vitrectomy (27 with BRVO and 22 with macular hole). observation procedures: Vitreous fluid samples were obtained during vitreoretinal surgery to measure the levels of sVEGFR-2, VEGF, and sICAM-1. Retinal ischemia was evaluated from capillary nonperfusion on fluorescein angiography. Macular edema was examined by optical coherence tomography. main outcome measures: Vitreous fluid levels of the 3 molecules and severity of macular edema.
Results
BRVO patients had a significantly higher vitreous fluid level of sVEGFR-2 (median, 1670 pg/mL; interquartile range [IQL], 1205 to 2225 pg/mL) than macular hole patients (median, 1265 pg/mL; IQR, 731 to 1800 pg/mL; P = .017), as was the case for VEGF (median, 237 pg/mL; IQR, 42.2 to 1305 pg/mL; vs median, 15.6 pg/mL; IQR, 15.6 to 15.6 pg/mL; P < .001) and sICAM-1 (median, 10.1 ng/mL; IQR, 6.3 to 22.5 ng/mL; vs median, 4.1 ng/mL; IQR, 3.3 to 6.0 ng/mL; P < .001). In BRVO patients, there was a significant positive correlation between vitreous fluid levels of sVEGFR-2 or VEGF and sICAM-1, but not between sVEGFR-2 and VEGF. Vitreous fluid levels of all 3 molecules were correlated significantly the with severity of macular edema in BRVO patients.
Conclusions
sVEGFR-2 may induce an increase of vascular permeability together with or via sICAM-1, or both with and via sICAM-1, in BRVO patients with macular edema.
Branch retinal vein occlusion (BRVO) is a common retinal vascular disease that often leads to macular edema, which is the most frequent cause of visual impairment in these patients. We previously reported that the intraocular levels of both vascular endothelial growth factor (VEGF) and interleukin 6 were correlated significantly with the severity of macular edema in BRVO patients, suggesting that increased vascular permeability may have an important role in the development of macular edema associated with BRVO. Intravitreal injection of bevacizumab (a monoclonal antibody targeting VEGF) or ranibizumab (an Fab fragment that binds and neutralizes all isoforms of VEGF-A) has been reported to improve macular edema in patients with BRVO. However, some patients experience persistent macular edema despite intravitreal treatment with bevacizumab or ranibizumab. This suggests that other mechanisms, such as inflammatory molecules like intercellular adhesion molecule 1 (ICAM-1), VEGF receptor (VEGFR), or both, may contribute to macular edema in BRVO patients as well as VEGF.
We recently reported that the aqueous humor level of soluble ICAM-1 (sICAM-1) was correlated significantly with the severity of macular edema in BRVO patients and that there was a significant correlation between aqueous levels of sICAM-1 and VEGF in these patients. It has been reported that signaling via VEGFR-2, but not VEGFR-1, is responsible for the increase of vascular permeability mediated by VEGF. However, the relations among the vitreous levels of VEGFR-2, VEGF, and ICAM-1 in BRVO patients with macular edema have not been investigated. Therefore, we performed the present study to investigate the influence of soluble VEGFR-2 (sVEGFR-2), VEGF, and sICAM-1 on macular edema in BRVO patients.
Methods
Subjects
This was a retrospective case-control study of 49 Japanese patients who underwent vitrectomy in 1 eye (27 with BRVO and 22 with idiopathic macular hole [MH]) to treat macular edema. During surgery, we obtained vitreous fluid samples to measure the levels of sVEGFR-2, VEGF, and sICAM-1. Pars plana vitrectomy was performed at Tokyo Women’s Medical University or Eguchi Eye Hospital. Forty-two consecutive patients with BRVO who sought treatment at these hospitals between January 2008 and March 2010 were screened using the criteria listed below, and vitreous fluid samples were obtained from the 27 patients who were enrolled. The indications for pars plana vitrectomy were: (1) clinically detectable diffuse macular edema or cystoid macular edema persisting for more than 3 months, (2) best-corrected visual acuity worse than 20/40, (3) persistent macular edema despite retinal photocoagulation, or (4) a combination thereof. In addition, the subjects were limited to patients who could fix on the central landmark during optical coherence tomography.
Before surgery, focal laser photocoagulation was carried out for macular edema in 6 eyes (mean, 245 shots; range, 120 to 378 shots) at 3.6 ± 0.8 months (range, 3 to 5 months) before surgery. The Branch Vein Occlusion Study (BVOS) demonstrated the effectiveness of argon laser photocoagulation for BRVO, but it was recommended that this should not be performed within 3 months of onset, during which time spontaneous improvement may occur. Accordingly, all 6 patients who underwent retinal photocoagulation were treated more than 3 months after the onset of BRVO. Absence of posterior vitreous detachment has been reported to contribute to persistent macular edema in patients with retinal vascular occlusion. In addition, Saika and associates reported the effectiveness of vitrectomy combined with surgical posterior vitreous detachment for BRVO patients with macular edema. Accordingly, we performed vitrectomy more than 3 months after the occurrence of BRVO in patients with clinically detectable diffuse macular edema, cystoid macular edema, or persistent macular edema at least 3 months after photocoagulation.
Significant macular edema was defined as retinal thickening of 1 optic disc area or more that involved the fovea. Fifteen of the 42 patients were excluded because of previous ocular surgery or vitreous injection of anti-VEGF agents or triamcinolone acetonide in 11 patients, diabetic retinopathy in 2 patients, and a history of ocular inflammation or vitreoretinal disease in 2 patients. Vitreous fluid samples also were obtained from 22 patients with nonischemic ocular disease (MH) as a control group (MH group). None of the patients in the MH group had proliferative vitreoretinopathy. A diagnosis of hypertension or hyperlipidemia was based on data from the medical records. Hypertension was defined as current treatment with antihypertensive drugs or a blood pressure of more than 140/90 mm Hg. Hyperlipidemia was defined as current treatment with lipid-lowering agents or a total cholesterol level of more than 240 mg/dL.
Fundus Findings
The fundus was examined before surgery by biomicroscopy with a fundus contact lens and by standard fundus color photography. In addition, fluorescein angiography was performed with a Topcon TRC-50EX fundus camera, an image-net system (Tokyo Optical Co. Ltd., Tokyo, Japan), and a preset lens with a slit lamp.
Both preoperative and operative fundus findings were recorded for each subject. A masked grader independently assessed retinal ischemia by examining fluorescein angiograms and measured the ischemic region of the retina with the public domain Scion Image program, as reported previously. On digital fluorescein photographs, the disc area was outlined with a cursor and then measured, and the same was done for the nonperfused area (sites of retinal photocoagulation were excluded when calculating the size of the nonperfused area). Then, the severity of retinal ischemia was calculated as the nonperfused area divided by the disc area.
Optical coherence tomography was performed in each subject within 1 week before vitrectomy using an instrument from Zeiss-Humphrey Ophthalmic Systems (Dublin, California, USA). The fundus was scanned with the measuring beam focused on horizontal and vertical planes crossing the center of the fovea, which was located by examination of the fundus photograph and by each patient’s fixation. Cross-sectional images were collected by a single experienced examiner (H.N.) who continued each examination until highly reproducible scans were obtained. The thickness of the central fovea was defined as the distance between the inner limiting membrane and the retinal pigment epithelium (including any serous retinal detachment) and was measured automatically by computer software. The thickness of the neurosensory retina was defined as the distance between the inner and outer neurosensory retinal surfaces, and the severity of macular edema was graded from the measured retinal thickness. The average preoperative retinal thickness was 607 ± 176 μm, with a range of 321 to 939 μm.
Sample Collection
Samples of undiluted vitreous fluid (300 to 500 μL) were collected at the start of vitrectomy by aspiration into a 1-mL syringe attached to the vitreous cutter before commencing the intravitreal infusion of balanced salt solution. The vitreous samples were transferred immediately into sterile tubes and were frozen rapidly at −80° C. Blood samples were collected simultaneously and were centrifuged at 3000 g for 5 minutes to obtain plasma, after which aliquots were stored at −80° C until assay.
Measurement of Soluble Vascular Endothelial Growth Factor Receptor-2, Vascular Endothelial Growth Factor, and Soluble Intercellular Adhesion Molecule 1
Levels of sICAM-1 and VEGF were measured in vitreous fluid samples from the same eye as well as in plasma with enzyme-linked immunosorbent assay kits for human VEGF (R&D Systems, Minneapolis, Minnesota, USA) and human sICAM-1 (Bender Med Systems, Burlingame, California, USA). The VEGF kit detected 2 of the 4 VEGF isoforms, which were VEGF 121 and VEGF 165 . Vitreous fluid levels of sVEGFR-2 also were determined by enzyme-linked immunosorbent assay according to the manufacturer’s instructions (Mitsubishi Chemical Medience Corporation, Tokyo, Japan). A volume of 100 μL of assay diluent RD 1W (R&D Systems) and 100 μL of either the standard control or a 5-fold diluted vitreous fluid sample were added to each well of the enzyme-linked immunosorbent assay plate, which was incubated for 2 hours on a shaker at room temperature and then was washed. Then, a horseradish peroxidase-labeled polyclonal antibody for sVEGFR-2 was added to each well, followed by incubation for 2 hours with shaking at room temperature and washing of the plate. Next, the substrate solution (200 μL), color reagent A (hydrogen peroxide; R&D Systems) and color reagent B (chromogen; R&D Systems) were mixed together in equal volumes and were added to each well, after which the plate was incubated for 30 minutes at room temperature in darkness. Finally, 50 μL of stop solution (2N sulfuric acid; R&D Systems) was added, and the absorbance was determined at 450 nm (with correction at 540 or 570 nm) by using a microplate reader. The levels of these factors in the vitreous fluid samples and plasma were within the detection ranges of the assays, with the minimum detectable concentration being 78.1 pg/mL for sVEGFR-2 (intra-assay coefficient of variation [CV], 5.4%; inter-assay CV, 6.8%), 15.6 pg/mL for VEGF (intra-assay CV, 5.6%; inter-assay CV, 6.8%), and 3.3 ng/mL for sICAM-1 (intra-assay CV, 5.5%; inter-assay CV, 7.6%).
Statistical Analysis
All analyses were performed with SAS software version 9.1 (SAS Institute, Cary, North Carolina, USA). Data are presented as the mean ± standard deviation or as the median with the interquartile range or frequency. The Student t test was used to compare normally distributed unpaired continuous variables between the 2 groups, and the Mann–Whitney U test was used for variables with a skewed distribution. The paired t test or Wilcoxon signed-rank test was used to compare paired continuous variables. To examine the relation between sVEGFR-2, VEGF, or sICAM-1 and the severity of macular edema, Spearman rank-order correlation coefficients were calculated. Two-tailed P values of less than .05 were considered to indicate a statistically significant difference.
Results
The Table summarizes the demographic and clinical features of the 2 groups. The mean age ± standard deviation of the BRVO group (13 men and 14 women) was 70.6 ± 7.1 years, whereas that of the MH group (11 men and 11 women) was 67.8 ± 8.6 years. The mean duration ± standard deviation of BRVO was 4.8 ± 2.1 months (range, 3 to 10 months). Fifteen of the 27 BRVO patients (56%) had hypertension and 10 patients (37%) had hyperlipidemia. Clinical and demographic data (including age and gender) did not show any significant differences between the BRVO and MH groups, apart from a significant difference of the systolic blood pressure ( P < .001).
| BRVO Group | Macular Hole Group | P Value | |
|---|---|---|---|
| No. a | 27 | 22 | |
| Gender (male/female) | 13/14 | 11/11 | .897 |
| Age (y) | 70.6 ± 7.1 b | 67.8 ± 8.6 b | .218 |
| Blood pressure (mm Hg) | |||
| Systolic | 136 ± 15 b | 122 ± 12 b | <.001 |
| Diastolic | 82 ± 11 b | 77 ± 10 b | .106 |
| Hypertension | 15 | 4 | .008 |
| Hyperlipidemia | 10 | 5 | .280 |
| Duration of BRVO (mos) | 4.8 ± 2.1 b | — | — |
a Number of patients with data.
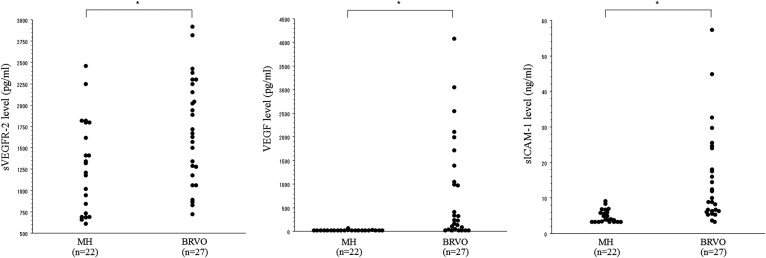
The vitreous fluid concentration of sVEGFR-2 was significantly higher in the BRVO group (median, 1670 pg/mL; interquartile range [IQR], 1205 to 2225 pg/mL) than in the MH group (median, 1265 pg/mL; IQR, 731 to 1800 pg/mL; P = .017; Figure 1 , Left). The vitreous fluid level of VEGF also was significantly higher in the BRVO group (median, 237 pg/mL; IQR, 42.2 to 1305 pg/mL) than in the MH group (median, 15.6 pg/mL; IQR, 15.6 to 15.6 pg/mL; P < .001; Figure 1 , Middle), as was the case for the vitreous level of sICAM-1 (median, 10.1 ng/mL; IQR, 6.3 to 22.5 ng/mL; vs median, 4.1 ng/mL; IQR, 3.3 to 6.0 ng/mL; P < .001; Figure 1 , Right).
There was a strong positive correlation between the vitreous fluid levels of sVEGFR-2 and sICAM-1 in the BRVO group (ρ = 0.70; P < .001). There also was a significant correlation between the vitreous fluid levels of VEGF and sICAM-1 in the BRVO group (ρ = 0.45; P = .018). However, no correlation of sVEGFR-2 with VEGF was detected in the BRVO group (ρ = 0.14; P = .490) or in the MH group (ρ = 0.02; P = .942). There was also no correlation of sVEGFR-2 with sICAM-1 in the MH group (ρ = 0.12; P = .572) or of VEGF with sICAM-1 in the MH group (ρ = 0.21; P = .337; Figure 2 ).
Vitreous fluid levels of VEGF and sICAM-1 were correlated significantly with the nonperfused area of the retina in the BRVO group (ρ = 0.86; P < .001; and ρ = 0.49; P = .012, respectively; Figure 3 , Middle and Right), but the vitreous level of sVEGFR-2 was not (ρ = 0.15; P = .468; Figure 3 , Left). In addition, the vitreous fluid levels of sVEGFR-2, VEGF, and sICAM-1 all were correlated significantly with the severity of macular edema in the BRVO group (ρ = 0.42; P = .034; ρ = 0.56; P = .004; and ρ = 0.54; P = .006, respectively; Figure 4 ). There were no significant differences in sVEGFR-2, VEGF, or sICAM-1 levels between the 6 eyes that received preoperative retinal photocoagulation and the 21 eyes without it (data not shown; P = .179, P = .321, and P = .930, respectively). The vitreous level of sICAM-1 was not correlated significantly with either the extent or the timing of retinal photocoagulation in the 6 treated patients (ρ = 0.70; P = .117; and ρ = 0.10; P = .823, respectively). The vitreous level of VEGF also showed no significant correlation with the extent or timing of photocoagulation (ρ = 0.66; P = .150; and ρ = 0.33; P = .534, respectively). Furthermore, the vitreous sVEGFR-2 level showed no significant correlation with the extent or timing of photocoagulation (ρ = 0.48; P = .277; and ρ = 0.43; P = .334, respectively).




