29 Socket Reconstruction
PREOPERATIVE EVALUATION
A functional anophthalmic socket that permits the wear of a comfortable and mobile ocular prosthesis requires (a) a centrally positioned orbital implant of adequate volume, (b) a conjunctival or mucous membrane-lined socket with adequately deep fornices, (c) a correctly positioned lower eyelid of normal length and tone to support the weight of the prosthesis, and (d) an upper eyelid with good levator function that is properly oriented, in apposition to the prosthesis, and with symmetric height and contour with the opposite side. A lack of or disturbance of one or more of these parameters may lead to the inability to retain a prosthesis and severe disfigurement and psychological distress of the patient.
A thorough history is important in an evaluation of the anophthalmic patient, including determination of the ocular condition leading to enucleation or evisceration, type and size of the orbital implant, previous surgery or therapy (e.g., volume augmentation, eyelid malposition repair, radiation), antecedent periocular trauma and associated orbital fractures, prior sinus disease, and past management of the ocular prosthesis. Examination of the socket should begin with an assessment of the eyelids and periorbital region, with the best-fitting prosthesis properly positioned in the socket. Excessive lower lid laxity and malposition may develop because of its support for the weight of the prosthesis, requiring assessment of its medial and lateral canthal tendon attachments. Measurements should be obtained of the vertical and horizontal palpebral fissures, as well as of the upper eyelid crease and fold and levator function. Notation of the degree of a superior sulcus defect and Hertel exophthalmometry can provide information on orbital volume. The excursion of the ocular prosthesis on vertical and, especially, horizontal eye movements should be noted. A history of previous trauma mandates palpation of the orbital bony rim and evaluation for asymmetry of the malar eminence and lateral and medial canthal positions.
Upon removal of the prosthesis, the socket should be assessed for adequacy of the superior and inferior fornices, presence of symblepharon, integrity of the conjunctival lining, position of the orbital implant, and signs of infection or inflammation. Palpation of the socket cavity can reveal migration of and shape and size characteristics of the orbital implant, as well as space-occupying orbital masses. If the possibility of a significant orbital fracture or recurrent tumor exists, radiographic studies with computerized tomography are indicated.
Finally, careful evaluation of the ocular prosthesis is important and includes searching for protein deposits, scratches, and irregularities, aided by the use of a magnifying loupe or slit-lamp biomicroscope. An excessively large and heavy prosthesis may aggravate lower lid laxity and result in a dropped socket appearance. Close interaction with an ocularist is critical, as modification or replacement of a prosthesis can correct upper lid ptosis, superior sulcus defects, eyelid malpositions, poor motility, and mild socket contraction in selected cases. Nonsurgical management of sockets in children with congenital anophthalmos is also performed by an ocularist using a progressively enlarging series of conformers until maximum socket and eyelid development is reached.
SURGICAL INDICATIONS
Appropriate management of anophthalmic socket disorders initially requires individualized consideration of a patient’s symptoms and goals. Certain clinical conditions responsive to surgical intervention, such as upper lid ptosis or superior sulcus defects, may be considered a minor cosmetic blemish by some patients who prefer no treatment, but a major disfigurement by others who request corrective measures. On the other hand, such problems as implant extrusion and socket contracture require surgical intervention to maintain a functional socket that can retain a prosthesis and prevent secondary complications such as infection or chronic pain. The disorders of the anophthalmic socket are amenable to surgical therapy.
Implant extrusion
Extrusion of orbital implants can occur in the early postoperative period or develop months or years after enucleation or evisceration. Inadequate wound closure, insertion of oversized implants, and rarely, infection may be responsible for early extrusion. Patients usually present within days or weeks of surgery with mucopurulent discharge, conjunctival inflammation, and evidence of a wound dehiscence. Management involves treating infections with appropriate antibiotics, possibly replacing the implant that is too large with one that is smaller in diameter, and performing a meticulous closure of the overlying Tenon’s capsule and conjunctiva.
Late extrusions are usually caused by cicatricial contraction of orbital tissues or the breakdown of tissues covering the implant owing to prolonged pressure by the prosthesis. Such orbital implants as wire mesh implants, Allen implants, and exposed integrated implants were notorious for late extrusions because of their configuration and relation with the central socket conjunctival surface and, thus, their use has been abandoned. Porous implants have largely replaced these older styled implants given their potential to become vascularized and integrated with the surrounding orbital tissues. Delayed extrusions, which may be partial or complete, might be manifested by irritative symptoms, discharge, and the inability to retain an ocular prosthesis. Small fistulas between the implant and conjunctival lining are typical of partial extrusions, and these fistulas will gradually enlarge, with associated thinning of the adjacent tissues covering the implant. When identified at an early stage, partial extrusions can be effectively managed with a patch graft that uses donor sclera, autogenous fascia lata, or temporalis fascia. Complete extrusion of an orbital implant, however, requires the introduction of a secondary implant if there is adequate mucous membrane present, or a dermis-fat graft in patients for whom there is a deficiency of conjunctiva.
Implant migration
Migration of the orbital implant from its central location to a peripheral position in the anophthalmic socket may result in difficult prosthetic fitting and wear, poor motility, foreshortened fornices, and eventual implant extrusion. Contractile scar tissue formation, gravitational forces, and differential forces of extraocular muscles when imbricated over the implant are some of the etiologic factors leading to a malpositioned implant. This condition is best corrected by removal of the migrated implant, which is then replaced with either a secondary implant or a dermis-fat graft more centrally positioned in the orbit.
Enophthalmos and superior sulcus deformity (orbital volume deficiency)
A reduction in the volume of orbital contents always results following enucleation or evisceration at least partly because of the replacement of the globe with an orbital implant smaller in diameter. Other factors attributed to this deficit include coexistent orbital fractures, lack of an orbital implant, redistribution of the orbital soft tissues, radiation-induced fat atrophy, and contractile fibrosis of connective tissue elements. One or more of these factors lead to anophthalmic enophthalmos and superior sulcus defects. Gravitational effects and loss of support of the superior transverse ligament and superior rectus–levator muscular complex are also thought to play a role in the development of the sulcus deformity.
Surgical correction of these clinical entities may include one or more volume augmentation procedures. If an orbital implant is absent, the preferred method of volume replacement is the insertion of a spherical implant or dermis-fat graft. In a socket in which the primary alloplastic implant is too small (i.e., <16-mm diameter), exchange with as large a secondary implant as the socket will allow is recommended. When the original implant is of satisfactory size and is centrally located, or when the foregoing procedures have been performed with inadequate volume augmentation, placement of a subperiosteal implant along the orbital floor may be corrective. Numerous materials for such implants have been utilized, including autogenous or lyophilized bone, Teflon or glass beads, Proplast, and bone cement; however, my preferences are prefabricated wedge-shaped implants made of polymethyl methacrylate resin or high-density polyethylene, or room temperature vulcanizing (RTV) silicone implants. Not only do these implants provide additional intraorbital volume, but they also yield a mechanical displacement of the orbital implant and soft tissues in an anterior and superior direction that helps to further correct the enophthalmos and superior sulcus defects. Recent reports of the use of non–animal-based synthetic fillers (stabilized hyaluronic acid and calcium hydroxylapatite) for anophthalmic orbital volume augmentation have shown promising results, but the long-term results of such products have not been established.
Upper eyelid ptosis
Ptosis of the upper eyelid associated with anophthalmos is a common problem. Similar to the pathomechanical alterations causing a deepened superior sulcus, ptosis usually results from stretching and laxity of the orbital tissues caused by gravity, enophthalmos, disturbance in the suspension of Whitnall’s ligament, and excessive anterior advancement of the superior rectus muscle over the implant. Foreshortening of the superior fornix by closing Tenon’s capsule and conjunctiva under excessive tension, or direct damage to the levator muscle during enucleation surgery, might also contribute to the development of ptosis. If the upper lid malposition cannot be improved by altering the prosthesis, then surgical correction is indicated. External levator surgery is performed when levator function is fair to good (>6 mm of excursion). When there is poor or no levator function, frontalis suspension may be considered. These surgical techniques are described elsewhere in this text.
Lower eyelid laxity and inadequate inferior fornix
The lower eyelid provides much of the support of the orbital implant and bears all of the weight of the ocular prosthesis. Over a period, gravitational forces and the weight of the prosthesis loosen the supporting elements of the lower lid. Laxity of the eyelid is accentuated as the orbital tissues migrate inferiorly and the prosthesis is enlarged in an attempt to improve the resultant superior sulcus deformity or upper lid ptosis. Sagging of the lower eyelid contributes to the disfiguring dropped socket appearance and, eventually, may lead to the inability to maintain a properly positioned prosthesis. Horizontal lid shortening and tightening with a lateral tarsal strip procedure is effective in correcting the lower lid laxity and is described in detail in a previous chapter.
Dehiscence of the lower lid retractors, development of scar tissue in the cul-de-sac, and anteroinferior migration of the orbital implant may result in foreshortening of the inferior fornix. Particularly when an inadequate fornix is combined with lower lid laxity, retention of the prosthesis may be compromised. Re-formation of the inferior fornix is achieved with refixation and stenting of the fornix and lower lid tightening when there is sufficient socket conjunctiva. In patients in whom the fornix is deficient of conjunctiva, a hard palate mucosal graft or a free mucous membrane graft that is stented into position is required.
Socket contracture
Acquired contraction of the socket represents the most serious disorder of anophthalmos and the most difficult to manage. Precipitating factors include chronic infection or inflammation, trauma, radiation therapy, surgery, and migrating or extruding implants. Once the etiologic factors leading to socket contracture are identified and eliminated, surgical and prosthetic management can begin. The spectrum of socket contracture has been divided into mild, moderate, and severe forms.
Mild socket contracture refers to cicatricial entropion of the upper or lower eyelids caused by fibrotic contracture of the posterior lamella. The entropic lid margin and eyelashes result in irritative symptoms and ocular discharge. A transverse blepharotomy and marginal rotation procedure, which is discussed elsewhere in the text, effectively repositions the eyelid and lashes to a normal position.
Contracture of the superior or inferior fornices leading to the inability to retain an ocular prosthesis is categorized as moderate socket contracture. Its development may occur in the early or late postoperative period and is associated with chronic discharge and irritation, poor prosthetic motility, and enophthalmos. Foreshortening of the inferior fornix with inadequate conjunctiva is the most common form of this condition. Reconstructive surgery involves the re-formation of a fornix and placement of a partial- or full-thickness mucous membrane graft, usually obtained from the lip or buccal mucosa, which is held in position with a Silastic (polymeric silicone) stent secured to the inferior orbital rim periosteum or a conformer. Alternatively, hard palate grafts may be implanted to expand the socket mucosal surface and are more resistant to postreconstruction contracture. If the superior fornix contracts, changes in the upper eyelid position, crease, fold, and levator function may develop, as well as extrusion of the prosthesis. Reformation of the superior fornix is achieved in the same manner as that for the inferior fornix except that a conformer is preferable to a Silastic stent to maintain the configuration of the mucous membrane graft, as it is less likely to damage the levator muscle. Moderate socket contracture is difficult to correct, may require multiple surgical procedures, and often results in an unsatisfying outcome.
The severe form of socket contracture typically manifests marked vertical forniceal contracture, horizontal shortening of the palpebral fissure, and in certain patients, obliteration of the lid margins. Active cicatricial processes and multiple previously failed surgeries may be contraindications to attempt reconstruction. Partial- or full-thickness mucous membrane grafts are stented into position with specially designed conformers that often require fixation to the orbital rims to prevent extrusion. Four to six months later, the conformer is removed, and an immediate prosthetic fitting is undertaken. The goal of creating a socket cavity that can retain an ocular prosthesis is often not achieved, however, and, even when successful, the final appearance is often only fair with an immobile prosthesis and eyelids.
TIMING AND PREFERENTIAL ORDER OF SURGICAL PROCEDURES
Surgical management of anophthalmic socket disorders may require multiple staged operations for a functional and cosmetically acceptable result. Most reconstructive procedures should be delayed at least 4 to 6 months following the initial enucleation or evisceration to allow the orbital tissues to heal and scar tissue to mature. If the fornices are foreshortened or the socket is contracted, preventing the retention of a prosthesis, re-formation of an adequately deep socket, with or without an oral mucous membrane graft, is the first priority. Correction of orbital implant malposition and size or a disturbance of the integrity of overlying tissues is the next goal. Volume replacement procedures may be considered to correct superior sulcus defects and enophthalmos. Severe lower lid laxity can be corrected at the time of volume augmentation or later. Finally, ptosis surgery of the upper eyelid may be undertaken following the revision or replacement of the ocular prosthesis after the completion of any of the foregoing procedures.
SURGICAL ANATOMY OF THE ANOPHTHALMIC SOCKET
The anophthalmic socket comprises a complex unit of orbital fat and connective tissue, extraocular muscles, vessels, nerves, Tenon’s capsule, and a conjunctival lining. The pathomechanical changes that develop following the surgical removal of the eye are largely dependent on the type of primary surgery and positioning of the orbital implant within the orbit. For example, if an evisceration has been performed, the orbital tissues are not disturbed, and the conjunctiva and Tenon’s capsule maintain their anatomic configuration, minimizing the potential for development of socket contracture and implant migration. On the other hand, when enucleation has been undertaken, major alterations in the structural and functional elements of the orbit develop. A deficit in orbital volume, loss of support of the superior rectus–levator muscular complex, retraction of the extraocular muscles, scarring and contracture of the orbital fibrous connective tissue septa, redistribution of the orbital fat, and stretching of the lower lid supporting tendons by the weight of the prosthesis all contribute to the clinical disorders of the anophthalmic socket. Recognition of these anatomic changes is essential to establish the proper management and surgical approaches for these patients.
Patch graft technique for extruding orbital implants
Repair of partially extruding implants with a patch graft can be performed under local or general anesthesia. Temporalis fascia is the preferred patch graft material because it is easily accessible; the scar at the site of graft harvesting remains hidden by the hairline, and it will result in minimal reaction and shrinkage and maximize viability because it is autogenous. The donor site is located just above the anterior aspect of the helix of the ear and superior to the palpable superficial temporal artery that should be avoided. A disposable razor can be used to trim the hair in the proposed surgical field and lidocaine (Xylocaine) 2% with 1:100,000 epinephrine is injected subcutaneously. The socket conjunctiva in the area of dehiscence is also infiltrated with the same local anesthetic solution to minimize bleeding. An alternative to temporalis fascia for patching material is preserved sclera that, if selected, should be placed in antibiotic solution at the beginning of surgery.
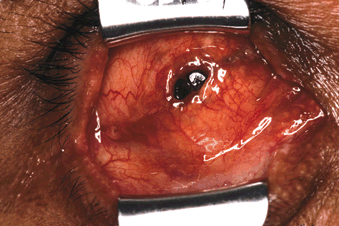
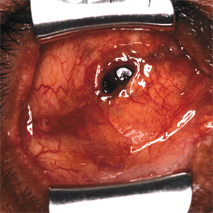
Figure 29-1. A lid speculum is inserted to expose the central socket cavity and the site of implant exposure. Dehiscence of the conjunctiva and Tenon’s capsule is seen in the socket.
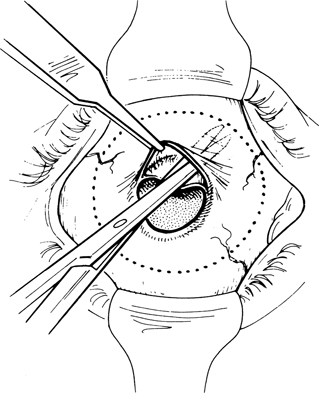
Figure 29-2. The edges of the fistula are freshened and the conjunctiva is undermined from the adjacent scar tissue and Tenon’s capsule with scissors.
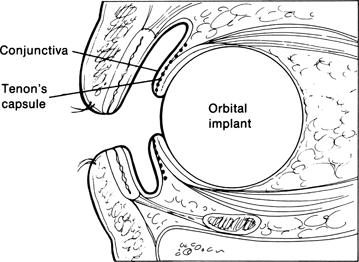
Figure 29-3. The plane of dissection is depicted by the dotted line in this cross-sectional view of the socket. The size of the patch graft necessary to cover the exposed orbital implant and overlap the edges of Tenon’s capsule by at least 2 to 3 mm is measured.

Figure 29-4. Harvesting of the fascia involves making a 4-cm incision with a Bard-Parker size 15 blade in the temporal scalp. The superficial temporal artery should first be identified by feeling for its pulsations, and the incision is made posterior to this landmark to avoid cutting the vessel. Sharp dissection with scissors through the subcutaneous tissue is performed until the temporalis fascia is exposed.
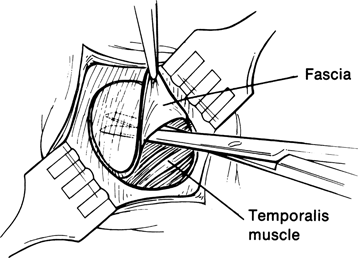
Figure 29-5. With the edges of the scalp incision retracted, a Bard-Parker size 15 blade is used to outline the fascial graft to be harvested. Blunt and sharp scissor dissection is used to separate the fascia from the underlying temporalis muscle, and the graft is set aside in saline-soaked gauze. Hemostasis is maintained with a bipolar cautery. The fascial defect is not repaired. The scalp incision is closed with deep interrupted 4–0 Vicryl sutures, and skin closure is achieved with interrupted 6–0 nylon sutures.
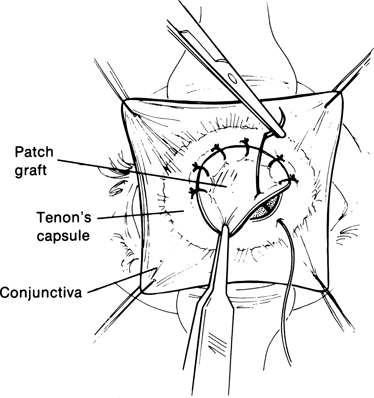
Figure 29-6. The temporalis fascia graft (or preserved scleral graft) is trimmed to size and placed into the defect in the socket between Tenon’s capsule and conjunctiva. The graft is oversized so that its borders overlap the edges of the defect when it is secured into place with multiple interrupted 6–0 Vicryl sutures passed between the edges of Tenon’s capsule and the overlapping edges of the graft.
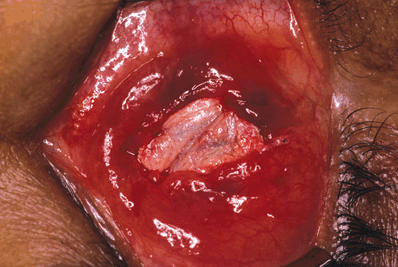
Figure 29-7. The temporalis fascia patch graft is shown sutured into position to cover the exposed orbital implant.
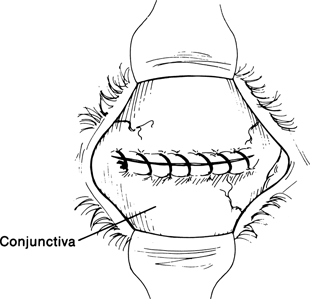
Figure 29-8. The conjunctival edges are then sutured together with a running 6–0 plain gut suture over the graft to provide its blood supply.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


