Purpose
To determine whether serologic factors correlate with early relapse in cases of IgG4-related orbital inflammation, a lymphoproliferative disorder.
Design
Retrospective cohort study.
Methods
We examined the orbital images and serum data of 30 patients with (n = 24) or without (n = 6) corticosteroid treatment before and after corticosteroid treatment and 6 months after discontinuation of corticosteroid treatment.
Results
Seven patients (23%) (all with corticosteroid treatment) had elevated serum levels of rheumatoid factor. Of the 24 patients with corticosteroid treatment, 24 (100%) showed regression of the lesion, but 8 of the 24 (33%) showed relapse. The serum IgG4 levels before and after corticosteroid treatment were not significantly different between the relapsed and nonrelapsed groups ( P = .5 and P = .2, respectively). However, the incidence of patients who were rheumatoid factor–positive was significantly higher in the relapsed group ( P = .02). The 6 patients without corticosteroid treatment showed minor proliferation or regression of the lesion at the 6-month follow-up.
Conclusions
The serum rheumatoid factor level may correlate with proliferative activity in IgG4-related orbital inflammation.
IgG4-related orbital inflammation, which includes IgG4-related dacryoadenitis and IgG4-related orbital myositis, is an orbital inflammatory disease that is characterized by the infiltration of IgG4-positive lymphoplasmacytic cells into the ocular adnexa. These cells can also infiltrate other tissues, such as the salivary glands and lymph nodes. Patients with IgG4-related orbital inflammation have characteristic serum abnormalities that may include elevated levels of serum IgG4, IgE, soluble interleukin (IL)-2 receptor, and polyclonal hypergammaglobulinemia. In spite of the extensive data on this disease, the pathogenesis of IgG4-related orbital inflammation has not been determined.
IgG4-related orbital inflammation responds well to steroids but often recurs soon after discontinuation of steroid treatment. The risk factors for early relapse of IgG4-related orbital inflammation are unknown. We hypothesized that serum abnormalities may cause early relapse of IgG4-related orbital inflammation and may also account for the enhanced proliferative activity characteristic of IgG4-related orbital inflammation.
The purpose of this study was to determine the serum characteristics of patients with early recurrence of IgG4-related orbital inflammation. Toward this end, we examined orbital images on computed tomographic (CT) scans and the serum characteristics of patients before and after corticosteroid treatment and 6 months after discontinuation of corticosteroid treatment. Findings in patients with early relapse were compared with those in patients without early relapse.
Patients and Methods
Patients
We designed a retrospective cohort study to evaluate the relationship between the serum characteristics of patients with IgG4-related orbital inflammation with or without early relapse. All patients were examined between February 1, 2005 and May 31, 2011. We excluded patients with distinct autoimmune diseases including Sjögren syndrome, lymphoma (especially on marginal zone B-cell lymphoma with IgG4-positive plasma cells), thyroid orbitopathy, vasculitis (eg, Wegener granulomatosis), and granulomatous lesions (eg, xanthogranuloma and sarcoidosis).
Patients were diagnosed with IgG4-related orbital inflammation if the ocular adnexal lesion had dense lymphoplasmacytic infiltrations, an IgG4-to-IgG plasma cell ratio >40%, and >100 IgG4-positive plasma cells in a single high-power field. In addition, a serum IgG4 level of ≥135 mg/dL was used for the diagnosis. Of the 31 patients who met the criteria for IgG4-related orbital inflammation, 1 patient with concurrent autoimmune pancreatitis was excluded because the patient was undergoing treatment for pancreatitis. The study thus included 30 patients.
Clinical Data
We recorded each patient’s age, sex, laterality, duration of symptoms, illnesses (including allergic diseases), and the anatomic location of the lesion. To determine that there was systemic involvement, 23 patients (77%) underwent screening at their initial examinations by whole-body CT scans, gallium-67 imaging, or 18 F- fluorodeoxyglucose positron emission tomography. The remaining 7 patients refused to undergo screening for systemic involvement.
Laboratory Data
Hematologic examinations were performed on peripheral blood collected from all patients at the time of diagnosis and at each of the follow-up visits, with an emphasis on the serum levels of IgG, IgG4, and IgE. To analyze patients with high serum level of IgG4, we divided patients into 2 groups according to serum level: Group 1 included those with IgG4 levels ≥900 mg/dL and Group 2 included those with IgG4 levels ≥135 mg/dL but <900 mg/dL. Blood eosinophilia was diagnosed when the number of eosinophils was ≥600 cells/mm 3 . Patients with serum levels of soluble IL-2 receptor were examined in more detail to determine whether an in vivo activation of the immune system had occurred. We determined the serum levels of antinuclear antibody, anti-SS-A/Ro, and rheumatoid factor (a latex turbidimetric immunoassay) at the time of diagnosis in all patients. When the antibody titer was ≥1:160, the patient was classified as being positive for antinuclear antibody. We also compared the rheumatoid factor levels in patients with IgG4-related orbital inflammation with the levels in age- and sex-matched controls.
Treatments and Outcomes
Corticosteroid treatment was initiated only after informed consent was obtained from the patient. An alternative treatment for patients who were unwilling to receive corticosteroid treatment was 30 Gy radiotherapy. Four patients refused any treatment because of mild signs and symptoms. Two patients refused corticosteroid treatment but received 30 Gy radiotherapy because of diabetes mellitus. Of the remaining 24 patients, 23 patients received corticosteroid treatment and 1 patient received corticosteroid treatment following 30 Gy radiotherapy. Corticosteroid treatment began with oral administration of 30 mg/day prednisolone for 1 or 2 weeks, and this dose was reduced by 5 mg/day at 2-week intervals. Of the 24 patients receiving corticosteroid treatment, 12 patients received a maintenance dose of 5 mg/day for at least 3 months following the tapering of prednisolone.
A regression and a recurrence of IgG4-related orbital inflammation was determined radiologically. We evaluated the response of orbital lesions to medical treatment by comparing axial and coronal CT scans before and after treatment and at 6 months after discontinuation of corticosteroid treatment. To determine the degree of regression of the orbital lesion after corticosteroid treatment, we assumed that the shape of the lesion was an ellipsoid to calculate its approximate volume. We determined the regression factor for the semi-major axis (X), the semi-minor axis (Y), and the major axis (Z) before and after corticosteroid treatment. We defined the regression rate as the XYZ value after corticosteroid treatment divided by the XYZ value before corticosteroid treatment. We also defined the relapse ratio as the XYZ value at 6 months after discontinuation of corticosteroid treatment divided by the XYZ value before corticosteroid treatment. When the regression rate was <40%, it was considered regression ( Figure 1 ); when the rate was ≥40% but <80%, it was considered partial regression; and when the ratio was ≥80%, it was considered poor regression. When the relapse rate was ≥80%, it was considered complete relapse ( Figure 1 ); and when the relapse rate was ≥40% but <80%, it was considered partial relapse. Finally, when the relapse rate was <40%, it was considered minor relapse ( Figure 1 ).
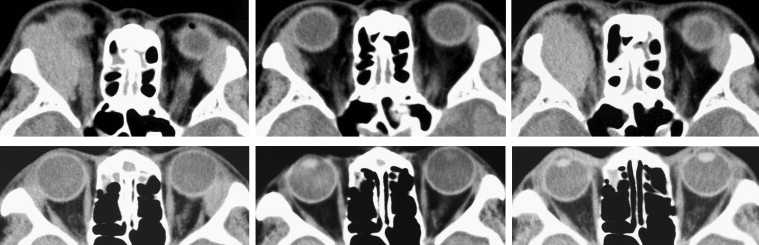
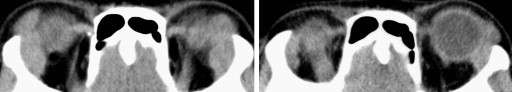
For patients without corticosteroid treatment, we defined the regression as a rate of the XYZ value at the 6-month follow-up divided by the XYZ value before the corticosteroid treatment. When the rate was ≥100%, it was considered proliferation; and when the rate was ≥80% but <100%, it was considered poor regression. When the rate was ≥40% but <80%, it was considered partial regression ( Figure 2 ).
Histopathology, Immunohistochemistry, and Flow Cytometric Analysis
Biopsy specimens of the ocular adnexal mass–forming lesions including orbital soft tissues were collected from all patients. Part of the biopsy specimen was embedded in paraffin for conventional histologic and immunohistochemical analyses, and the remainder was used immediately for flow cytometry analysis. The histopathology and immunohistochemistry methods and procedures were described previously. Four fresh samples from the lesions were analyzed by flow cytometry (FACSCalibur direct dual-parameter flow cytometer; Becton Dickinson and Company, San Jose, California, USA) using fluorescein isothiocyanate– or phycoerythrin-conjugated monoclonal antibodies to CD4, CD5, CD20, and HLA-DR. Biopsy specimens from other organs were collected from 4 patients with systemic involvement.
Statistical Analyses
To determine the characteristics of patients with high serum IgG4 levels, we compared the sex, laterality distribution, duration of symptoms, association of allergic disease, and incidence of blood eosinophilia between the high IgG4 group (serum IgG4 level ≥900 mg/dL) and low IgG4 group (serum IgG4 level <900 mg/dL) using the χ 2 test or Fisher exact test ( Table 1 ). Patient age, laboratory data, and duration of symptoms were compared using the Mann-Whitney U test ( Table 1 ). We also compared the incidence of systemic involvement of IgG4-related lesions between the 2 groups by the Mann-Whitney U test (eg, salivary gland involvement, 1; and salivary gland and lymph nodes involvement, 2).
| High IgG4 Group (n = 8) | Low IgG4 group (n = 22) | Total | P Value | |
|---|---|---|---|---|
| Serum IgG4 Level ≥900 mg/dL | Serum IgG4 Level <900 mg/dL | |||
| Age (y), mean (SD) | 63 (12) | 57 (16) | 58 (15) | .4 |
| Sex, male, n (male-to-female ratio) | 6 (6:2) | 12 (12:10) | 18 (18:12) | .4 |
| Laterality, bilateral, n (%) | 7 (88) | 14 (64) | 21 (70) | .4 |
| Mean duration of symptoms, months (range) | 33 (3-84) | 16 (0.3-72) | 21 (25) | .07 |
| Association of allergic diseases, n | 5 | 7 | 12 | .2 |
| Serum IgG level, mg/dL a (SD) | 4401 (1529) | 1819 (328) | 2508 (1416) | N.A. |
| Serum IgG4 level, mg/dL a (SD) | 1836 (588) | 482 (186) | 812 (656) | N.A. |
| Serum IgE level, IU/mL a (SD) | 1707 (1605) | 1285 (1724) | 1398 (1703) | .4 |
| Blood eosinophilia, n | 3 | 0 | 3 | .01 |
| Soluble IL-2 receptor, U/mL a | 1366 (512) | 560 (197) | 768 (481) | .001 |
| Rheumatoid factor before CST (>30 IU/mL), n | 3 | 4 | 7 | .3 |
| Systemic involvement per person, mean (SD) | 1.6 (0.5) | 0.7 (0.7) | 0.9 (0.8) | .005 |
a Normal range: IgG, 870-1700 mg/dL; IgG4, 5-105 mg/dL; IgE, <170 IU/mL; soluble IL-2 receptor, 135-483 U/mL.
To determine the characteristics of patients with early relapse of the lesions, we compared the sex, laterality distribution, duration of symptoms, association of allergic disease, incidence of rheumatoid factor, and maintenance corticosteroid treatment between the relapsed and nonrelapsed groups using the χ 2 test or Fisher exact test ( Table 2 ). Patient age, duration of symptoms, and serum levels of IgG, IgG4, IgE, and soluble IL-2 receptor were compared between the 2 groups using the Mann-Whitney U test ( Table 2 ).
| Relapse | P Value | ||
|---|---|---|---|
| (Relapsed: n = 8) | (Nonrelapsed: n = 16) | ||
| Age, mean (SD) | 54 (18) | 59 (15) | .4 |
| Sex, male (n) | 4 | 10 | .7 |
| Laterality, bilateral (n) | 7 | 10 | .4 |
| Duration of symptoms (mo), mean (SD) | 17 (19) | 23 (26) | 1.0 |
| Association of allergic diseases, n | 5 | 6 | .7 |
| IgG level before CST, mg/dL, a mean (SD) | 2554 (1015) | 2644 (1713) | .2 |
| IgG level after CST, mg/dL, a mean (SD) | 1407 (424) | 1370 (571) | .7 |
| IgG4 level before CST, mg/dL, a mean (SD) | 789 (356) | 849 (770) | .5 |
| IgG4 level after CST, mg/dL, a mean (SD) | 376 (200) | 295 (221) | .2 |
| IgE level before CST, IU/mL, a mean (SD) | 1269 (1516) | 1442 (1962) | .7 |
| IgE level after CST, IU/mL a mean (SD) | 512 (601) | 795 (1009) | .5 |
| Rheumatoid factor before CST (>30 IU/mL), n | 5 | 2 | .02 |
| Soluble IL-2 receptor, U/mL a | 733 (350) | 752 (446) | .8 |
| Maintenance CST, n | 5 | 7 | .7 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


