Purpose
To describe the indications for secondary enucleations in uveal melanoma and analyze associations and outcomes.
Design
Retrospective interventional case series.
Methods
Data of patients who underwent secondary enucleation for uveal melanoma in the London Ocular Oncology Service, between 2008 and 2014, were retrieved from medical records analyzed. Cox regression model was performed to analyze associations with secondary enucleation and metastases and Kaplan-Meier estimates to assess the probability of metastatic spread and death.
Results
During the study period 515 enucleations were performed for uveal melanoma, 99 (19%) of which were secondary enucleations. Tumors were located at the ciliary body in 21 eyes (21%), juxtapapillary in 31 (31%), and choroid elsewhere in 47 (48%). Primary treatment included Ru 106 plaque radiotherapy, proton beam radiotherapy, and transpupillary thermotherapy in 85, 11, and 3 eyes, respectively. Indications for secondary enucleation were tumor recurrence in 60 (61%), neovascular glaucoma in 21 (21%), and tumor nonresponse in 18 eyes (18%). Twenty patients (20%) were diagnosed with metastasis and 12 out of 20 died of metastatic spread. On multivariate analysis, juxtapapillary tumor location was found to associate with tumor nonresponse ( P = .004) and nonresponding patients with metastatic spread ( P = .04).
Conclusions
Indications for secondary enucleations for uveal melanoma were tumor recurrence, neovascular glaucoma, and tumor nonresponse. This review identified a possible high-risk group (nonresponse), which proved radioresistant to treatment. These tumors were more frequently found in the juxtapapillary location and were associated with metastatic spread.
Uveal melanoma is the most common primary intraocular malignancy in adults, with an incidence of approximately 6 individuals per million population. Three decades ago, the main treatment for uveal melanoma was primary enucleation, aimed at extending survival by removing the eye containing a tumor with metastatic potential. Following the Collaborative Ocular Melanoma Study (COMS) trial, which showed no advantage for enucleation in respect to melanoma-related mortality, treatment now consists mainly of eye-preserving therapies, with plaque brachytherapy currently being the most common. Despite this shift, some patients still require a secondary enucleation after initial conservative treatment, owing to tumor growth or development of neovascular glaucoma (NVG). Reported rates of secondary enucleation for uveal melanoma range widely from 3.9% to 31.2%, according to different studies. It is noteworthy that most of these reports do not focus on secondary enucleation specifically but rather on treatment outcomes in general. The few cohorts that discuss secondary enucleation are limited in size. The objective of this study is to describe a large case series of uveal melanoma patients that underwent secondary enucleation and determine the clinical indications for eye removal and associated tumor risk factors, and to report patient survival.
Methods
All enucleation cases for uveal melanoma performed in the London Ocular Oncology Service from January 1, 2008, through December 31, 2014, were retrieved from the surgical log book. Of these, only patients that underwent secondary enucleation were included in the study and their medical records were retrospectively reviewed. The data retrieved from the charts included sex, age, ocular and systemic medical history, laterality, type and timing of primary and salvage treatments, radioactive treatment dosage calculations, surgical notes, indications for and timing of secondary enucleation, clinical examinations throughout the follow-up including early and late postoperative complications, pathology results, development of metastasis, and death from metastatic disease or other causes. Missing medical data were retrieved by contacting the patient’s general practitioner (performed by I.D.F.). Tumor location was categorized as originating from the ciliary body; the juxtapapillary choroid, defined as tumor edge located within 1.5 mm from the optic disc (hereafter referred to as juxtapapillary); or choroid elsewhere (hereafter referred to as choroid). The study protocol was approved by Moorfields Eye Hospital institutional review board.
Surgical Technique and Patient Management
All enucleations were executed under general anesthesia. After identifying the correct eye, a 360 degree peritomy was performed, 4/0 vicryl sutures were used to secure each rectus muscle, and all extraocular muscles were detached. Eye traction was carried out using silk sutures or artery clamps and the eye was removed with a snare. After meticulous hemostasis was performed, a hydroxyapatite implant was inserted; Tenon and conjunctiva were sutured with 5/0 and 6/0 vicryl sutures, respectively; and a temporary conformer was placed on top. Removed eyes were sent for pathologic evaluation. Tumor cell aspirate for cytologic evaluation was sent only in a minority of selected cases. Patients were given 250 mg oral cephalexin qds (4 times a day of oral tablet) for a week and g (guttae [eye drops]). chloramphenicol qid (4 times a day of eye drops, 1 drop each time) for 28 days. Pathology of the specimen was reviewed and only those with macroscopic extrascleral extension were referred for adjuvant radiotherapy. All patients were referred to the National Artificial Eye Service.
Statistical Analysis
All calculations were performed using Microsoft Excel 2013 software (Microsoft Corporation, Redmond, Washington, USA) and SPSS software version 17.0 (SPSS, Inc, Chicago, Illinois, USA).
A series of univariate Cox proportional hazard regressions were used to assess the degree of relationship of all of the variables to indications for secondary enucleation and to development of metastasis. A subsequent multivariate model was performed using only variables found significant ( P < .05) in the univariate analysis. Kaplan-Meier survival estimates of the probability of death and of developing metastasis were performed as a function of time from initial treatment.
Results
In the 7-year study period, 515 patients had their eye removed for uveal melanoma; 99 (19%), or approximately one fifth of the total number, had a secondary enucleation performed. Table 1 summarizes the study patients’ demographic and clinical features at presentation. Tumors were located at the ciliary body in 21 patients (21%) and choroid in 47 (48%) and were juxtapapillary in 31 (31%). Nine patients (9%) were diagnosed with a diffuse melanoma and 11 (11%) were initially diagnosed with a nevus that transformed into melanoma during follow-up. Primary treatments included Ru 106 plaque brachytherapy in 85 patients (86%), proton beam radiotherapy in 11 patients (11%), and transpupillary thermotherapy (TTT) in 3 patients (3%).
| Features | Number N = 99 | Percentage |
|---|---|---|
| Age (y) | ||
| Median (mean, range) | 60 (60, 24–89) | |
| Sex | ||
| Male | 61 | 62 |
| Female | 38 | 38 |
| Laterality | ||
| Right | 49 | 49 |
| Left | 50 | 51 |
| LogMAR | ||
| Median (mean, range) | 0.3 (0.66, −0.08–2.7) | |
| Tumor location | ||
| Ciliary body | 21 | 21 |
| Choroid | 47 | 48 |
| Juxtapapillary | 31 | 31 |
| Tumor size (mm) | ||
| Median (mean, range) | ||
| Height | 4.6 (4.4, 1.0–9.0) | |
| Base diameter | 10.8 (10.2, 4.0–16.9) | |
| Primary treatment | ||
| Ru 106 plaque radiotherapy | 85 | 87 |
| Proton beam radiotherapy | 11 | 11 |
| Transpupillary thermotherapy | 3 | 3 |
Table 2 depicts the subdivision of primary treatment type and tumor location. Tumors originating from all 3 anatomic locations were mainly treated with plaque brachytherapy (100%, 95.7%, and 61.3% for ciliary body, choroid, and juxtapapillary tumors, respectively), whereas only one third of the juxtapapillary tumors were treated with proton beam radiotherapy as primary treatment. For patients treated with Ru 106 plaque brachytherapy the median treatment time was 69 hours (mean 82, range 36–180), median radiation dose to the tumor base was 366 Gy (mean 368, range 128–1205) and to the tumor apex was 100 Gy (mean 105, range 80–120). Proton beam radiotherapy was performed at the Clatterbridge Centre for Oncology and included administration of 4 daily fractions on consecutive days, with a total proton dose of 53.1 Gy, having an assumed relative biological effect of 1.1. In order to reach tumor control after primary treatment, 35 patients (35%) received additional salvage TTT, 2 (2%) received additional plaque brachytherapy salvage treatment, and 1 (1%) received a combination of TTT and plaque brachytherapy. The median number of TTT salvage treatments was 1 (mean 0.9, range 0–4). Salvage treatment, whether in the form of TTT or plaque brachytherapy, postponed the need for secondary enucleation by a median interval of 22 months (mean 30.7, range 1–125).
| Primary Treatment | Ciliary Body | Choroid | Juxtapapillary | All |
|---|---|---|---|---|
| Plaque brachytherapy | 21 | 45 | 19 a | 85 (86%) |
| Proton beam radiotherapy | 0 | 1 | 10 | 11 (11%) |
| TTT | 0 | 1 | 2 | 3 (3%) |
| 21 (21%) | 47 (48%) | 31 (31%) | 99 (100%) |
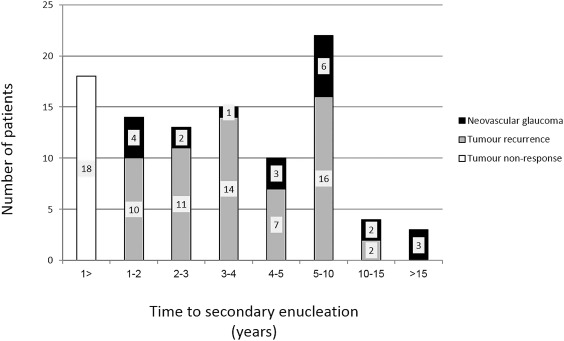
Indications for secondary enucleation were tumor recurrence in 60 patients (61%), neovascular glaucoma (NVG) in 21 (21%), and tumor nonresponse in 18 (18%). Tumor nonresponse was defined as growth of uveal melanoma within the first year of primary treatment. The median time from primary treatment to secondary enucleation was 38 months (mean 55.5, range 5–456). The bar chart ( Figure 1 ) shows the time line from primary treatment to secondary enucleation and how this is related to the clinical indication for secondary enucleation. Juxtapapillary tumor location accounted for 6 (33%) of the tumor nonresponse, 17 (28%) of tumor recurrence, and 8 (38%) of NVG cases ( Table 3 ). When assessing the hazard ratio (HR) using the Cox regression model, multivariate analysis indicated that juxtapapillary tumor location was associated with tumor nonresponse (HR 0.09 [95% confidence interval (CI), 0.02–0.46], P = .004) and tumor height at presentation with tumor recurrence (HR 1.87 [95% CI, 1.03–3.38], P = .04). No clinical parameters were found to associate with the development of NVG indicating secondary enucleation.
| Indication for Secondary Enucleation | Ciliary Body | Choroid | Juxtapapillary | All |
|---|---|---|---|---|
| Tumor nonresponse a | 4 | 8 | 6 | 18 (18%) |
| Tumor recurrence | 14 | 29 | 17 | 60 (61%) |
| Neovascular glaucoma | 3 | 10 | 8 | 21 (21%) |
| 21 (21%) | 47 (48%) | 31 (31%) | 99 (100%) |
a Defined as tumor growth within the first year of primary treatment.
Pathology evaluation revealed that tumors were of spindle cell type in 60 patients (61%), epithelioid in 12 (12%), mixed cell in 23 (23%), and signet cell/balloon cell in 1 (1%), and in 3 (3%) the tumor was necrotic, precluding tissue diagnosis. Extraocular extension was detected in 8 patients (8.1%), 2 of whom, with incomplete excision from macroscopic extraocular extension, received additional orbital radiotherapy (50 Gy in 20 daily fractions).
Of the patients that received salvage TTT and/or secondary radiotherapy, the indications for secondary enucleation were tumor recurrence in 28 (74%), NVG in 8 (21%), and tumor nonresponse in 2 (2%). Of the 36 patients that received additional salvage TTT, in 4 tumors extraocular extension was diagnosed in pathology evaluation, of which only in 1 (of a total of 2 patients receiving orbital radiotherapy) further orbital radiotherapy was indicated.
There were no significant socket-related complications, no cases of implant extrusion, and no tumor recurrences. The 2 patients who received orbital radiotherapy had no posttreatment complications. All patients fitted with a permanent prosthesis were satisfied with their cosmetic appearance.
The median follow-up time from initial treatment was 58 months (mean 80, range 9–460). The median time to developing metastasis from uveal melanoma diagnosis was 35 months (mean 40.8, range 6–99). There was no loss to follow-up. Only 2 of the patients receiving salvage treatment were diagnosed with distant metastasis. Systemic spread was detected in 20 patients (20%), 6 of whom had metastases identified at the time of secondary enucleation. Twelve of 20 patients died from metastatic disease; the median survival in the metastatic group was 9 months (mean 10.8, range 1–36). All deaths were from metastatic spread and none from other nonmelanoma-related causes.
Univariate analysis revealed that age at presentation, sex, tumor location, number of TTT treatments, and indication for enucleation were associated with metastatic spread ( P ≤ .04). On multivariate analysis, however, only female sex ( P = .03) and tumor nonresponse ( P = .04) remained significant factors ( Table 4 ). Adding pathology results to the analysis did not change its output. Using Kaplan-Meier estimates, the tumor nonresponse group was found to be at significantly higher risk to developing metastasis (5-year cumulative metastasis-free patient survival rate was 26% ± 20%) as compared to the recurrence group (5-year cumulative metastasis-free patient survival rate was 83% ± 5%, P < .001; Figure 2 ). The 5- and 10-year cumulative metastasis-free patient survival rate for the whole cohort was 80% ± 5% and 70% ± 6%, respectively, and cumulative survival rate was 86% ± 4% and 81% ± 6%, respectively ( Figure 3 ).
| Univariate (HR [95% CI]) | P Value | Multivariate (HR [95% CI]) | P Value | |
|---|---|---|---|---|
| Age at presentation | 1.04 [1.00–1.08] | .04 | 1.03 [0.98–1.09] | .26 |
| Sex | 0.23 [0.09–0.6] | .003 | 0.17 [0.03–0.87] | .03 |
| Tumor location | 3.81 [1.56–9.31] | .003 | 1.26 [0.22–7.25] | .79 |
| Number of TTT treatments | 0.28 [0.89–0.89] | .03 | 0.37 [0.09–1.55] | .17 |
| Indication for enucleation b | 0.16 [0.06–0.41] | <.0001 | 0.15 [0.03–0.89] | .04 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


