Purpose
To investigate risk factors associated with visual field progression in the Low-pressure Glaucoma Treatment Study, a prospective trial designed to compare the effects of the alpha2-adrenergic agonist brimonidine tartrate 0.2% to the beta-adrenergic antagonist timolol maleate 0.5% on visual function in low-pressure glaucoma.
Design
Prospective cohort study.
Methods
Low-pressure Glaucoma Treatment Study patients with ≥5 visual field tests during follow-up were included. Progression was determined using pointwise linear regression analysis, defined as the same 3 or more visual field locations with a slope more negative than −1.0 dB/year at P < 5%, on 3 consecutive tests. Ocular and systemic risk factors were analyzed using Cox proportional hazards model and further tested for independence in a multivariate model.
Results
A total of 253 eyes of 127 subjects (mean age, 64.7 ± 10.9 years; mean follow-up, 40.6 ± 12 months) were analyzed. Eyes randomized to timolol progressed faster than those randomized to brimonidine (mean rates of progression, −0.38 ± 0.9 vs 0.02 ± 0.7 dB/y, P < .01). In the final multivariate model adjusting for all tested covariates, older age (hazard ratio [HR] = 1.41/decade older, 95% confidence interval [CI] = 1.05 to 1.90, P = .022), use of systemic antihypertensives (HR = 2.53, 95% CI = 1.32 to 4.87, P = .005), and mean ocular perfusion pressure (HR = 1.21/mm Hg lower, 95% CI = 1.12 to 1.31, P < .001) were associated with progression whereas randomization to brimonidine revealed a protective effect (HR = 0.26, 95% CI = 0.12 to 0.55, P < .001).
Conclusions
While randomization to brimonidine 0.2% was protective compared to timolol 0.5%, lower mean ocular perfusion pressure increased the risk for reaching a progression outcome in the Low-pressure Glaucoma Treatment Study. This suggests that the beneficial effect of randomization to the brimonidine arm was independent of possible differences in ocular perfusion pressures between the 2 treatment arms. The current results and large number of drop-outs in the brimonidine 0.2% arm suggest that more research is necessary before altering clinical practice paradigms.
Glaucoma is a progressive disorder characterized by structural and functional abnormalities of the optic nerve. Even though intraocular pressure (IOP) is the most important modifiable risk factor for disease onset and progression, glaucoma can exist even among individuals for whom IOP measurements are within the statistically defined “normal range.” Although an artificial construct, low-pressure (normal-tension, normal-pressure, or low-tension) glaucoma is a widely used term to classify the disease in patients with glaucomatous optic neuropathy with or without visual field loss whose pressures are within the 95th percentile of the normal distribution of IOP measurements in the healthy population (IOP <22 mm Hg using Goldmann applanation tonometry).
The Low-pressure Glaucoma Treatment Study is a multicenter, double-masked, prospective, randomized clinical trial that aimed to investigate visual field outcomes in low-pressure glaucoma patients treated with either a topical beta-adrenergic antagonist (timolol maleate 0.5%) or alpha2-adrenergic agonist (brimonidine tartrate 0.2%). The results of this trial revealed that subjects randomized to topical brimonidine 0.2% had better preservation of visual function than those receiving timolol 0.5% despite similar IOP levels. It is unclear, however, whether this outcome was attributable to different mechanisms of drug action or whether other risk factors, including IOP, also played a significant role. In the present study, we investigated baseline and intercurrent risk factors for visual field progression among participants enrolled in the Low-pressure Glaucoma Treatment Study.
Patients and Methods
The methodology of the Low-pressure Glaucoma Treatment Study, including baseline characteristics and study design, has been described in detail elsewhere. In brief, the Low-pressure Glaucoma Treatment Study Group was a multicenter, prospective clinical trial in which patients were randomized to treatment with topical brimonidine tartrate 0.2% vs timolol maleate 0.5%. The institutional review boards at all 13 participating centers approved the study protocol and informed consent was obtained.
Inclusion and Exclusion Criteria
Study patients had previously diagnosed low-pressure glaucoma that fulfilled the following eligibility criteria: all known untreated IOP ≤21 mm Hg; open iridocorneal angles; at least 2 reproducible visual fields with glaucomatous defects in 1 or both eyes on automated perimetry (Humphrey Field Analyzer; Carl Zeiss Meditec, Inc, Dublin, California, USA), with the location of the field defect being consistent with the photographic appearance of the optic nerve head; and age ≥30 years. To determine eligibility based on IOP, all patients receiving IOP-lowering treatment underwent a 4-week washout without therapy. Baseline IOP (measured with a calibrated Goldmann applanation tonometer) had to be ≤21 mm Hg in both eyes with <5 mm Hg difference between the eyes on an office diurnal curve (8:00 AM, 10:00 AM, 12:00 PM, 4:00 PM) assessed prior to randomization.
Ocular exclusion criteria included the following: a history of IOP >21 mm Hg in the patient record, best-corrected visual acuity worse than 20/40 in either eye, a history of angle closure or an occludable angle by gonioscopy, prior glaucoma incisional surgery, inflammatory eye disease, prior ocular trauma, diabetic retinopathy or other diseases capable of causing visual field loss or optic nerve deterioration, extensive glaucomatous visual field damage with a mean deviation worse than −16 decibels (dB), or a clinically determined threat to central fixation in either eye. Systemic exclusion criteria included a resting pulse <50 beats/minute; severe or uncontrolled cardiovascular, renal, or pulmonary disease that would preclude safe administration of a topical beta-adrenergic antagonist; and a prior myocardial infarction or stroke. Continuation of systemic medications that could affect IOP was allowed as long as the doses remained constant throughout the trial.
Randomization, Treatment, and Masking
Patients were randomly assigned to receive monotherapy with either brimonidine tartrate 0.2% containing benzalkonium chloride (Alphagan; Allergan, Inc, Irvine, California, USA) or timolol maleate 0.5% containing benzalkonium chloride (Timoptic; Merck & Co Inc, West Point, Pennsylvania, USA) twice daily in both eyes, including the morning before each visit. To allow for higher patient attrition in the brimonidine group attributable to an expected rate of adverse events of approximately 20%, randomization and delivery of medications (provided by Allergan, Inc, Irvine, California, USA) to the sites were stratified in blocks of 7 (4 to brimonidine and 3 to timolol). The randomization list was maintained and masked study medications were provided in new 10-mL white bottles labeled with the assigned randomization number directly to the clinical centers by an independent pharmacy (Fountain Valley Cancer Center Pharmacy, Fountain Valley, California, USA). Ocular treatment other than the study medication was not permitted. Investigators, patients, and the visual field reading and coordinating centers were all masked to patient assignment.
Endpoints requiring discontinuation from the study included: treated IOP >21 mm Hg that was repeated within 1 month, safety concern as judged by the treating physician, symptomatic ocular allergic adverse events (hyperemia, pruritus, stinging, and/or conjunctival folliculosis) requiring medication cessation, retinal events that could alter visual acuity or visual field (eg, age-related macular degeneration), the occurrence of systemic (eg, respiratory or cardiovascular) adverse events that prevented the administration of topical timolol, nonocular intolerable events associated with topical brimonidine (eg, xerostomia, fatigue, drowsiness), or if the patient moved or declined continued participation. Collection of data from discontinued patients ceased at their final study visit. Data up to this point were included in the analysis, but discontinued patients were no longer followed as part of the study.
Study Visits
Patients were examined at 1 and 4 months after initiation of treatment. Subsequent visits were at 4-month (±2 weeks) intervals. Pre- and post-randomization morning visits recorded the following: ocular and systemic history, blood pressure, pulse, corrected visual acuity, IOP, slit-lamp examination, and optic disc evaluation for cup-to-disc ratio and the presence or absence of disc hemorrhage. Gonioscopy and stereoscopic optic disc photographs were performed annually. Full-threshold standard achromatic perimetry (Humphrey 24-2) visual field was performed at 4-month intervals according to protocol guidelines.
Outcome Measures
The main outcome measure was visual field progression in 1 eye as determined by pointwise linear regression. Visual field progression analysis was performed using Progressor software (Medisoft, Inc, Leeds, UK). Visual field analyses were performed by an independent reading center (Devers Eye Institute, Legacy Health System, Portland, Oregon, USA) masked to the treatment assignment. Linear regression of the sensitivity (in dB) was performed at each test location to obtain the rate of change at that location, based on all fields up to and including the last examination. Default Progressor criteria were used to define a significant negative slope (at least −1.0 dB/year for inner points and −2.0 dB/year for edge points) at the P < 5% level. Edge points for the Humphrey 24-2 field included the 2 outer nasal locations, 1 above and 1 below the horizontal. Criteria for visual field progression required confirmation at the next 2 examinations of a significant negative slope at the same 3 or more test locations. For eyes not reaching a progression endpoint, we used the time to last follow-up visit (either the final scheduled visit or the censored one). Progressing visual field locations were not required to be contiguous. Because of these criteria, progression could not be determined until 5 visual field tests had been collected (the 16-month visit). Consequently, for the purposes of this manuscript, only those eyes with enough visual field tests to determine a progression outcome (yes/no) were included in the analysis (1 at baseline plus 4 tests following randomization).
We examined clinical characteristics that predicted the development of visual field progression. The following demographic and ocular parameters were investigated: age at baseline, sex, family history of glaucoma, central corneal thickness (CCT), cup-to-disc ratio (estimated during slit-lamp examination), refractive error spherical equivalent (SE), and lens status. Pre-randomization data collected and investigated for risk assessment were: IOP mean, peak, and fluctuation during the diurnal curve; baseline pulse; systolic (SBP) and diastolic blood pressure (DBP); presence of systemic comorbidities (migraine, Raynaud phenomenon, systemic hypertension, and diabetes mellitus); and use of systemic medications (categorized as systemic antihypertensives, systemic beta-blockers, and antidiabetic agents). Post-randomization variables consisted of series length (ie, follow-up time), detection of at least 1 optic disc hemorrhage on stereophotographs any time during follow-up, and mean, peak, and fluctuation of IOP and blood pressure during follow-up. For numerical variables, the mean was calculated by averaging all values recorded during the follow-up period. Fluctuation was defined as the standard deviation (SD) of all measurements in the same interval. Mean ocular perfusion pressure (MOPP) was estimated by the equation MOPP = 2/3 × [DBP + 1/3 × (SBP – DBP)] – IOP. Data on systemic comorbidities and medications were obtained from participants’ self-reports.
Statistical Analysis
Descriptive statistics are presented with frequency tables and graphs, whereas estimates of center and dispersion are described as mean and SD, respectively. Cox proportional hazards model was used to investigate the risk of progression (progression: yes or no, based on the progression criteria described above) based on follow-up time (progression endpoint or data censoring for progressing and nonprogressing eyes, respectively). Variables with P < .10 in the univariate model were entered in a multivariate model. Generalized estimating equations were used to control for inter-eye relationships. Since one of the assumptions of regression analyses is that the predictors should not be strongly and significantly correlated with one another (ie, no colinearity), if pairs of predictor variables had moderate to strong significant correlation, the variable with the most significant P value in the univariate analysis was entered in the multivariate model. Cox proportional hazards multivariate model was performed using a backward elimination approach based on likelihood ratios. Variables in the saturated multivariate model with P > .10 were subsequently removed and variables were entered in the model if P < .05. Alpha level was set at 5% (2-sided) and computerized statistical analyses were performed using SPSS for Windows (version 16.1; IBM SPSS Statistics Inc, Armonk, New York, USA).
Results
One hundred ninety-three patients were assessed for eligibility in the Low-pressure Glaucoma Treatment Study, of which 178 were randomized to treatment with either timolol or brimonidine. The characteristics of these patients are described in detail elsewhere. Of those, 253 eyes of 127 subjects (mean age, 64.7 ± 10.9 years; women, 58%; European ancestry, 71%) had at least 5 visual field tests and met the inclusion and exclusion criteria for this study. The following results, therefore, refer to this subset of eyes with the minimum number of fields required for PLR trend analysis. Table 1 shows the clinical characteristics of this population.
| Parameter | Timolol (n = 69) | Brimonidine (n = 58) | P Value |
|---|---|---|---|
| Age (years) | 65.2 ± 10.8 | 63.3 ± 11.1 | .18 b |
| Sex (male/female) | 26/43 | 27/31 | .36 c |
| Follow-up time (months) | 41.1 ± 11.6 | 40.4 ± 12.4 | .68 b |
| Positive family history for glaucoma (%) | 25 (36) | 17 (29) | .45 c |
| Use of systemic antihyptertensives (%) | 32 (46) | 24 (41) | .59 c |
| Use of systemic beta-blockers (%) | 11 (16) | 6 (10) | .43 c |
| Treatment for diabetes mellitus (%) | 6 (8) | 11 (18) | .11 c |
| Positive history of migraine (%) | 3 (4) | 6 (10) | .29 c |
| Positive history of Raynaud phenomenon (%) | 10 (14) | 5 (8) | .41 c |
| Spherical equivalent (diopters) | −0.64 ± 2.6 | −0.55 ± 2.2 | .78 b |
| Lens status (per eye) (pseudophakic eyes, %) | 10 (7) | 16 (13) | .08 c |
| At least 1 disc hemorrhage detected (%) | 12 (8) | 6 (5) | .33 c |
| Number of eyes with recurrent disc hemorrhages | 10 (7) | 3 (2) | .15 c |
| Cup-to-disc ratio | 0.72 ± 0.1 | 0.67 ± 0.1 | .03 b |
| Central corneal thickness (μm) | 547.8 ± 38.6 | 540.6 ± 29.0 | .10 b |
| Untreated diurnal curve IOP (mm Hg) | |||
| Mean | 15.2 ± 2.5 | 16.2 ± 1.9 | <.01 b |
| Peak | 16.7 ± 2.7 | 17.7 ± 2.0 | .04 b |
| Fluctuation | 1.28 ± 0.7 | 1.37 ± 0.6 | .30 b |
| Treated follow-up IOP (mm Hg) | |||
| Mean | 13.9 ± 2.3 | 14.1 ± 1.9 | .38 b |
| Peak | 16.4 ± 2.6 | 17.0 ± 2.3 | .03 b |
| Fluctuation | 1.59 ± 0.5 | 1.86 ± 0.6 | <.01 b |
| Mean % IOP reduction from baseline (%) | 8 ± 10 | 12 ± 10 | <.01 b |
| Baseline blood pressure (mm Hg) | |||
| Systolic | 132.3 ± 17.0 | 128.9 ± 18.2 | .13 b |
| Diastolic | 76.9 ± 9.2 | 74.7 ± 10.7 | .08 b |
| Follow-up blood pressure (mm Hg) | |||
| Mean systolic | 128.7 ± 13.1 | 129.4 ± 14.0 | .68 b |
| Mean diastolic | 75.8 ± 6.9 | 74.5 ± 7.9 | .14 b |
| Fluctuation systolic | 10.9 ± 6.1 | 10.9 ± 5.6 | .95 b |
| Fluctuation diastolic | 6.4 ± 3.8 | 6.2 ± 3.3 | .55 b |
| Mean ocular perfusion pressure (mm Hg) | |||
| Average follow-up | 48.4 ± 5.5 | 47.7 ± 5.8 | .32 b |
| Fluctuation follow-up | 3.7 ± 2.7 | 3.2 ± 2.4 | .19 b |
| Heart rate (beats/min) | |||
| Follow-up mean | 66.0 ± 6.7 | 70.9 ± 10.1 | <.01 b |
| Follow-up fluctuation | 5.68 ± 3.3 | 6.26 ± 3.5 | .18 b |
a All data are presented as mean ± standard deviation unless otherwise specified.
Of the 127 participants, 69 (54%) were randomized to timolol and 58 (46%) to brimonidine ( P = .20). Forty-eight eyes (48/253; 19%) of 40 patients (40/127; 31%) met the predefined PLR progression criteria (31 patients randomized to timolol; 9 patients randomized to brimonidine, P < .01, Fisher exact test). Patients were followed for a mean of 40.6 ± 12 months. As expected, the rate of mean deviation (MD) change (dB/y) was significantly faster in eyes that met our progression criteria than in those that did not (−0.87 ± 0.7 vs −0.04 ± 0.8 dB/y, P < .01). Figures 1 and 2 show the distribution of MD rates of change between progressing and nonprogressing eyes, as well as between the timolol (−0.38 ± 0.9 dB/y) and brimonidine (0.02 ± 0.7 dB/y) groups, respectively ( P < .01).
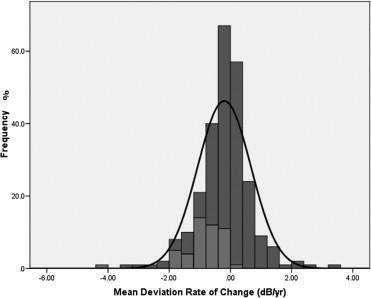
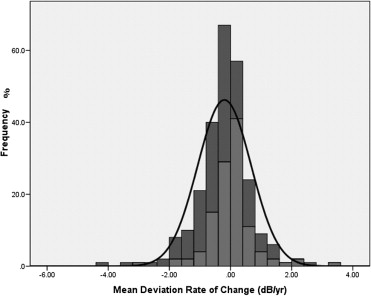
In this subset of participants with at least 5 visual field tests, eyes on brimonidine had significantly higher diurnal mean and peak IOP measurements prior to treatment randomization (respectively, 16.2 ± 1.9 vs 15.2 ± 2.5 mm Hg, P < .01; and 17.7 ± 2.0 vs 16.7 ± 2.7 mm Hg, P = .04) even though treated mean pressures during follow-up were similar between the 2 groups (14.1 ± 1.9 vs 13.9 ± 2.3 mm Hg, P = .38). However, eyes on brimonidine had statistically greater IOP fluctuation (1.86 ± 0.6 vs 1.59 ± 0.5 mm Hg, P < .01) and higher peaks (17.0 ± 2.3 vs 16.4 ± 2.6 mm Hg, P = .03) during follow-up.
Univariate analysis ( Tables 2 and 3 ) revealed the following variables to be associated with visual field progression at P < .25: age, lens status, use of systemic antihypertensives, use of systemic beta-adrenergic antagonists, disc hemorrhage, cup-to-disc ratio, CCT, mean SBP during follow-up, mean DBP during follow-up, SBP fluctuation, MOPP during follow-up, ocular perfusion pressure fluctuation, baseline heart rate, and randomization.
| Parameter | HR | 95% Confidence Interval | P Value |
|---|---|---|---|
| Randomization (brimonidine) | 0.29 | 0.14 to 0.58 | .001 |
| Age (per decade older) | 1.21 | 0.90 to 1.62 | .078 |
| Sex (female) | 1.48 | 0.82 to 2.69 | .192 |
| Positive family history for glaucoma | 1.04 | 0.57 to 1.90 | .882 |
| Use of systemic antihypertensives | 1.65 | 0.63 to 2.91 | .082 |
| Use of systemic beta-blockers | 1.71 | 0.85 to 3.43 | .131 |
| Treatment for diabetes mellitus | 0.79 | 0.31 to 2.02 | .629 |
| Positive history of migraine | 0.33 | 0.04 to 2.46 | .284 |
| Positive history of Raynaud phenomenon | 0.83 | 0.30 to 2.32 | .732 |
| Spherical equivalent (per diopter more positive) | 1.00 | 0.89 to 1.11 | .964 |
| Lens status (pseudophakic) | 0.41 | 0.10 to 1.70 | .222 |
| Disc hemorrhage detection | 2.14 | 0.90 to 5.08 | .082 |
| Cup-to-disc ratio (per 0.1-unit increase) | 1.16 | 0.95 to 1.41 | .127 |
| Central corneal thickness (per μm thicker) | 1.00 | 0.99 to 1.01 | .212 |
| Heart rate (beats/min, per unit higher) | |||
| Follow-up mean | 0.98 | 0.95 to 1.02 | .552 |
| Follow-up fluctuation | 0.95 | 0.86 to 1.04 | .333 |
| Parameter | Progression | Stable | HR | 95% Confidence Interval | P Value |
|---|---|---|---|---|---|
| Untreated baseline diurnal IOP (per mm Hg higher) | |||||
| Mean | 15.8 ± 2.6 | 15.6 ± 2.2 | 1.05 | 0.92 to 1.19 | .437 |
| Peak | 17.4 ± 2.3 | 17.7 ± 2.8 | 1.00 | 0.96 to 1.04 | .903 |
| Fluctuation | 1.38 ± 0.8 | 1.31 ± 0.6 | 1.15 | 0.79 to 1.67 | .462 |
| Treated follow-up IOP (per mm Hg higher) | |||||
| Mean | 13.9 ± 2.0 | 14.0 ± 2.2 | 1.00 | 0.88 to 1.13 | .926 |
| Peak | 16.6 ± 2.2 | 16.7 ± 2.6 | 1.00 | 0.90 to 1.11 | .991 |
| Fluctuation | 1.38 ± 0.8 | 1.31 ± 0.6 | 1.06 | 0.67 to 1.70 | .780 |
| Mean % IOP reduction from baseline (per 0.1% greater) | 11 ± 10 | 9 ± 10 | 1.08 | 0.91 to 1.29 | .343 |
| Baseline blood pressure (per mm Hg lower) | |||||
| Systolic | 130.9 ± 18.3 | 130.7 ± 17.5 | 0.99 | 0.98 to 1.01 | .740 |
| Diastolic | 75.9 ± 9.3 | 76.0 ± 10.1 | 0.99 | 0.96 to 1.02 | .508 |
| Follow-up blood pressure | |||||
| Mean systolic (per mm Hg lower) | 125.5 ± 11.8 | 129.8 ± 13.8 | 1.02 | 1.01 to 1.05 | .016 |
| Mean diastolic (per mm Hg lower) | 73.1 ± 5.3 | 75.7 ± 7.7 | 1.08 | 1.03 to 1.13 | <.001 |
| Fluctuation systolic (per mm Hg higher) | 11.7 ± 6.2 | 10.7 ± 5.8 | 1.03 | 0.98 to 1.08 | .228 |
| Fluctuation diastolic (per mm Hg higher) | 6.9 ± 3.5 | 6.2 ± 3.6 | 1.04 | 0.96 to 1.13 | .286 |
| Mean ocular perfusion pressure | |||||
| Baseline (per mm Hg higher) | 47.1 ± 7.1 | 47.4 ± 8.0 | 0.98 | 0.94 to 1.01 | .31 |
| Average follow-up (per mm Hg lower) | 46.4 ± 4.6 | 48.4 ± 5.8 | 1.10 | 1.04 to 1.17 | .001 |
| Fluctuation follow-up (per mm Hg higher) | 4.0 ± 2.5 | 3.4 ± 2.6 | 1.08 | 0.96 to 1.22 | .196 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


