Purpose
To analyze risk factors for postoperative recurrence of fibrovascular tissue in eyes with aggressive posterior retinopathy of prematurity (AP ROP) treated with early vitreous surgery.
Design
Retrospective, consecutive, observational case series.
Methods
Thirty-one patients (50 eyes) with AP ROP who underwent early vitreous surgery between March 2005 and April 2008 participated. Eyes with stage 4A or 4B disease in which fibrovascular tissue was not attached to the vitreous base were included; those in which fibrovascular tissue was attached extensively to vitreous base or those without dense photocoagulation to the nonvascularized retina were excluded. Eligible eyes were divided into 2 groups based on postoperative recurrence or no recurrence of fibrovascular tissue. Data on gender, gestational age, birth weight, Apgar score, intubation duration, severe systemic complications, preoperative ROP stage, zone, fibrovascular tissue and vitreous base adhesion, clock hours of fibrovascular tissue, postmenstrual age at the initial application of dense photocoagulation, dense photocoagulation to both vascularized and nonvascularized retina, postmenstrual age at vitrectomy, and intraoperative hemorrhage were collected and analyzed.
Results
Fifty eyes of 31 patients underwent early vitrectomy. Seven (14%) eyes were excluded and 43 eyes (86%) were included. Eight (18%) of 43 eyes had a recurrence of fibrovascular tissue. Both univariate and multivariate analysis indicated application of dense photocoagulation to both the vascularized and nonvascularized retina was a significant factor in the decreased recurrence of fibrovascular tissue ( P = .002 and P = .008, respectively).
Conclusions
Application of preoperative dense photocoagulation to vascularized and nonvascularized retina may be important for lowering the recurrence of fibrovascular tissue in eyes with AP ROP.
Aggressive posterior retinopathy of prematurity (AP ROP) is characterized by posterior retinopathy usually in zone I, substantial dilatation and tortuosity of the vessels at the posterior pole, a flat network of neovascularization on the retinal surface, circumferential fibrovascular tissue extending for 12 clock hours, and rapid progression to stage 5 without the classic course that includes stages 1 through 3. Previous randomized trials have reported that application of photocoagulation to the nonvascularized retina prevents retinal detachment in classic ROP, but that photocoagulation often cannot prevent progression in eyes with AP ROP.
Because the visual prognosis is poor after vitrectomy performed when AP ROP progresses to stage 5, several early surgical interventions have been tried to prevent stage 5 retinal detachment. Scleral buckling that reduces the traction between the fibrovascular tissue and the retina is ineffective for AP ROP that is characterized by fibrovascular tissue nearly 360 degrees circumferentially in the posterior retina. Lens-sparing vitrectomy usually is successful if performed for tractional retinal detachment in eyes with classic ROP, but the surgery is unsuccessful for AP ROP. Although it is controversial whether lensectomy is necessary during vitrectomy to treat retinal detachment in eyes with AP ROP, wide-field vitrectomy with lensectomy may be required to treat retinal detachment in eyes with AP ROP, which always is characterized by high disease activity despite the disadvantages of lens removal.
Eyes with AP ROP that have undergone early vitrectomy often have a retinal reattachment; however, among these eyes, we recently identified eyes in which proliferation of fibrovascular tissue recurred after surgery. Because a very low birth weight, young gestational age, or long-term high oxygen therapy are risk factors for severe ROP, we analyzed the correlation between recurrent fibrovascular tissue proliferation after early vitrectomy and the associated risk factors.
Methods
The medical records of all eyes that underwent early vitrectomy with lensectomy for stage 4 ROP associated with AP ROP were reviewed retrospectively at the Department of Ophthalmology, National Center for Child Health and Development, Tokyo, Japan, from March 2005 through April 2008. All eyes with AP ROP were diagnosed at the referring hospital or our institution based on the description published in 2005. Eyes with stage 4A or stage 4B ROP in which fibrovascular tissue did not reach the vitreous base were included; eyes with fibrovascular tissue attached extensively to the vitreous base or eyes without dense and early application of photocoagulation even to the nonvascularized retina were excluded, because those eyes usually have a stage 5 retinal detachment, and rigorous evaluation of recurrence of fibrovascular tissue is almost impossible. The follow-up period after vitrectomy exceeded 6 months in all eyes in the analysis. All surgeries were performed by 1 surgeon (N.A.).
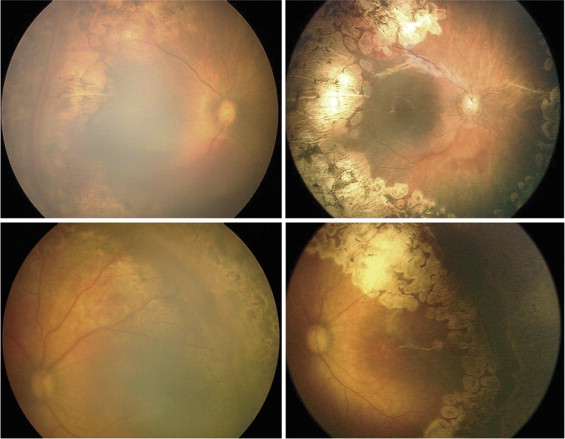
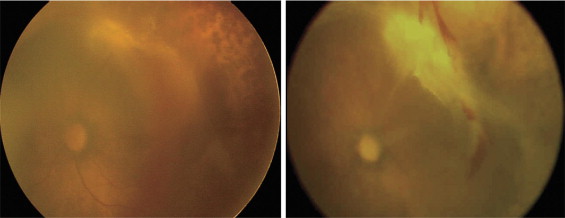
Data collected from each case record included gender, gestational age, birth weight, the Apgar score at 5 minutes, the duration of intubation, and the presence of severe systemic complications (i.e., hydrocephalus, patent ductus arteriosus, or necrotizing enterocolitis requiring surgery). We included the duration of intubation as an indicator of the degree of oxygen exposure. ROP findings included the preoperative ROP stage, zone, fibrovascular tissue and vitreous base adhesion, clock hours of fibrovascular tissue, postmenstrual age at the initial application of photocoagulation, photocoagulation to both vascularized and nonvascularized retina ( Figure 1 ) or only to nonvascularized retina ( Figure 2 ), postmenstrual age at vitrectomy, and intraoperative hemorrhage. Although the area of photocoagulation generally is limited to the nonvascularized retina, because the importance of photocoagulation to the vascularized retina has been suggested in eyes with severe ROP, we included eyes treated with photocoagulation applied to both the vascularized and nonvascularized retina. ROP findings were recorded by detailed retinal drawings and RetCam (Massie Research Laboratories, Inc, Pleasanton, California, USA). The eyes that fulfilled the inclusion criteria were divided into 2 groups based on the recurrence or absence of recurrence of fibrovascular tissue after surgery. The factors listed previously were compared between the 2 groups.
Statistical analyses were performed using statistical software (StatLab, SPSS for Windows, version 16.0; SPSS, Inc, Chicago, Illinois, USA). Univariate analyses to determine the association between risk factors and recurrence after early vitrectomy were performed using the Mann–Whitney U test, the t test, and the Fisher exact test as appropriate. A multivariate logistic regression model was constructed with recurrence as the dependent variable and with the factors that differed at the significance level of P < .2 in univariate analyses, including the gestational age, preoperative ROP stage, fibrovascular tissue and vitreous base adhesion, the postmenstrual age at the initial application of photocoagulation, and photocoagulation to both the vascularized and nonvascularized retina as independent variables. P < .01 was considered significant.
Results
A total of 50 eyes of 31 patients (19 girls, 12 boys) underwent early vitrectomy. Among them, 5 (10%) eyes that had not received sufficient photocoagulation even to the nonvascularized retina and 2 (4%) eyes that had extensive fibrovascular tissue adhesion to the vitreous base were excluded. Forty-three eyes (86%) of 29 patients were included. Of the 29 patients, 19 were girls and 10 were boys, with a mean gestational age of 25.2 ± 2.3 weeks and a mean follow-up of 23.8 ± 10.7 months.
Eight (18.0%) of 43 eyes had a recurrence of fibrovascular tissue after surgery ( Figure 2 ), and the others did not have a recurrence and the retina reattached ( Figure 1 ). The recurrences, which began 2 to 8 weeks after surgery and progressed gradually, were characterized by proliferation that developed mainly toward the vascularized posterior retina probably via the residual vitreous framework, where fibrous strands often formed between the disc and the fibrovascular tissue with an irregular tractional retinal detachment ( Figure 2 ). Three of the 8 eyes underwent a second vitrectomy 1 to 2 months after surgery because of severe recurrent fibrovascular tissue with a total retinal detachment. Because recurrent fibrovascular tissue adhered strongly to the retina where the tissue was not removed completely, retinal reattachment was obtained in only 1 eye. Four of 8 eyes had a recurrence of fibrovascular tissue that was less severe with a partial retinal attachment after primary surgery, and 1 eye had early progression to phthisis bulbi, and a second vitrectomy was not performed.
The data and statistics are summarized in the Table . There were more boys in the recurrence group than in the nonrecurrence group, but the difference was not significant ( P = .042). There were no significant differences in the other baseline characteristics between the 2 groups. Before surgery, the incidences of both stage 4B and fibrovascular tissue adhesion to the vitreous base were slightly higher in the recurrence group, but these did not reach significance ( P = .067 and P = .037, respectively). Photocoagulation was applied more often to both vascularized and nonvascularized retina in the group in which there was no recurrence of fibrovascular tissue compared with the group in which there was recurrence (74.3% vs 12.5%); the difference between the two was significant ( P = .002). There was no difference in the ROP findings between the 2 groups. Multivariate analyses using a stepwise logistic regression model also showed that only photocoagulation to both the vascularized and nonvascularized retina (odds ratio, 0.049; 95% confidence interval, 0.005 to 0.459; P = .008) was associated with postoperative recurrence of fibrovascular tissue, and the other factors were not significantly associated with recurrence.
| Total (n = 43) | Recurrence after Early Vitreous Surgery | P Value (2-Tailed) | ||
|---|---|---|---|---|
| Yes (n = 8; 19%) | No (n = 35; 81%) | |||
| Baseline characteristics | ||||
| No. eyes/patients | 43/29 | 8/6 | 35/25 | |
| Male, no (%) | 13 (30.2) | 5 (62.5) | 8 (22.9) | .042 a |
| Gestational age (wks), mean ± SD | 25.1 ± 2.3 | 24.8 ± 2.0 | 25.2 ± 2.4 | .703 b |
| Birth weight (g) mean ± SD | 808.7 ± 369.6 | 789.5 ± 302.2 | 813.0 ± 387.0 | .873 b |
| Apgar score at 5 min, median (range) | 6 (1 to 10) | 6 (2 to 10) | 5 (1 to 10) | .311 c |
| Intubation duration (wks), mean ± SD | 57.0 ± 40.6 | 60.9 ± 49.2 | 56.1 ± 39.2 | .767 b |
| Severe systemic complication, no. (%) | 12 (27.9) | 2 (25.0) | 10 (28.6) | >.99 a |
| Follow-up (mos), mean ± SD | 23.8 ± 10.7 | 22.6 ± 7.8 | 24.1 ± 11.3 | >.99 b |
| ROP findings | ||||
| Zone, no. (%) | .404 a | |||
| 1 | 29 (67.4) | 4 (50.0) | 25 (71.4) | |
| 2 | 14 (32.6) | 4 (50.0) | 10 (28.6) | |
| Stage, no. (%) | .067 a | |||
| 4A | 37 (86.0) | 5 (62.5) | 32 (91.4) | |
| 4B | 6 (14.0) | 3 (37.5) | 3 (8.6) | |
| Fibrovascular tissue and vitreous base adhesion, no. (%) | 5 (11.6) | 3 (37.5) | 2 (5.7) | .037 a |
| Clock hours of fibrovascular tissue, median (range) | 5 (2 to 12) | 9 (2 to 12) | 5 (2 to 12) | .344 c |
| PMA (wks) at initial PHC, mean ± SD | 32.9 ± 1.8 | 32.1 ± 0.8 | 33.1 ± 1.9 | .180 b |
| Interval between initial PHC and virectomy (wks), mean ± SD | 6.9 ± 3.2 | 7.0 ± 2.6 | 6.3 ± 3.3 | .892 b |
| PMA (wks) at vitrectomy, mean ± SD | 39.7 ± 3.1 | 39.1 ± 2.9 | 39.2 ± 3.9 | .536 b |
| Intraoperative hemorrhage, no. (%) | 13 (30.0) | 3 (37.5) | 10 (28.6) | >.99 a |
| PHC to both vascularized and nonvascularized retina, no. (%) | 27 (62.8) | 1 (12.5) | 26 (74.3) | .002 a |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


