Purpose
To determine the rate of recurrence and associated risk factors after the use of mitomycin C (MMC), interferon alpha-2b, or both for management of noninvasive ocular surface squamous neoplasia (OSSN).
Design
Retrospective, noncomparative, interventional case series.
Methods
Clinical practice setting of 135 patients treated consecutively with topical MMC (0.4 mg/mL), interferon alpha-2b (1 million units/mL), or both for OSSN observed for clinical recurrence.
Results
Clinical recurrences were diagnosed in 19 (14.1%) of 135 eyes after topical treatment. The mean time to recurrence was 17.2 months (range, 4 to 61 months), with 14 eyes (73.7%) recurring within a 2-year period. There was no greater risk of recurrence identified for variables including lesion size, lesion location, gender, age, treatment type, or treatment duration. Post hoc log-rank pairwise comparisons revealed that lesions initially treated using surgery alone had significantly reduced time to recurrence (21.1 ± 5.6 months) compared with previous topical treatment with MMC (with or without surgery; 29.6 ± 4.7 months; P = .04) and primary OSSN (23.2 ± 1.8 months; P = .09).
Conclusions
Topical MMC and interferon alpha-2b are an effective treatment method for a wide range of noninvasive OSSNs. Topical therapy avoids the morbidity of excisional surgery with equivalent or reduced recurrence rates and should be considered as primary therapy.
Ocular surface squamous neoplasia (OSSN) encompasses a spectrum of lesions arising from the squamous cells of the conjunctiva, cornea, or both. Excessive exposure to ultraviolet B radiation is the major etiologic factor; however, other causes, such as human papillomavirus types 16 and 18 and HIV seropositivity, also have been implicated. OSSN is described as a relatively low-grade malignancy because invasive disease is uncommon and tends to be preceded by dysplasia and carcinoma in situ. However, a lack of appropriate and effective treatment for OSSN can result in malignant change followed by local invasion and, rarely, metastasis. Surgical excision with adequate margins and adjunctive cryotherapy is a well-established treatment for OSSN, although this is an invasive option with numerous disadvantages.
Tabin and associates described high recurrence rates after surgical excision (33% at 10 years despite histologically clear surgical margins). Evidence suggests that the microscopic changes associated with OSSN extend beyond the macroscopic margin; thus, surgical excision to achieve clear margins is difficult. Techniques to examine tumor margins and conserve normal tissue such as Moh micrographic surgery have been adapted for oculoplastic tumor work; however, frozen sections are challenging to perform on small fragile corneal or conjunctival specimens. Additionally, because of the multifocal nature of OSSN, surgical excision results in wide collateral damage to adjacent areas of normal epithelium, including the potential for limbal stem cell deficiency and visually disturbing corneal scarring.
A recent study by Galor and associates examined the rates of recurrence after surgical excision using various techniques and found them to be 10% at 1 year and 21% at 5 years. The authors identified tarsal involvement and positive pathologic margins as the strongest predictors of clinical recurrence after surgical excision of OSSN lesions. High-grade lesions, large lesions, and a previous patient history of OSSN were associated with an increased risk of tumor recurrence. The mean time to clinical recurrence after surgical excision was 2.5 years.
The purpose of this study was to identify predictive factors of OSSN recurrence after topical treatment of noninvasive OSSN using a large, retrospective case series. The study examined the rates of recurrence of OSSN after the use of topical treatment for both primary therapy and for the treatment of recurrences using mitomycin C (MMC), interferon alpha-2b (IFN), or both. Identification of risks for recurrence potentially may allow for tailored management plans, with the aim of improving treatment success.
Methods
In this retrospective, noncomparative, interventional case series study, clinical data of 143 eyes of 135 patients diagnosed with OSSN were reviewed. Approval to conduct this study was obtained prospectively from the Royal Australian and New Zealand College of Ophthalmologists Human Research Ethics Committee (study no. 29.11). The National Health and Medical Research Council guidelines were followed, and it adhered to the tenets of the Declaration of Helsinki.
Database information allowed identification of all prescriptions for MMC or IFN dispensed consecutively by 2 oncology pharmacies: the Royal Brisbane and Women’s Hospital, a tertiary referral hospital in Brisbane, Queensland, Australia, and Custom Care Compounding Pharmacy in Dural, New South Wales, Australia. Eligible patients were those whose first prescription was dispensed between December 2001 and December 2012. Of these patients, those who were treated by a single ophthalmologist (G.A.L.) were chosen as candidates for inclusion in the study. This was to ensure uniformity in treatment regimen and clinical examination techniques. OSSN was diagnosed principally by clinical examination, but in some equivocal cases, by histology. All cases were primary or recurrent noninvasive lesions. Invasive lesions were excluded on the basis of tethering to the underlying sclera on indirect palpation of the lesion through the lid. For those patients with recurrent disease, previous treatment methods were recorded. Treatment with MMC or IFN continued until either clinical resolution was achieved or the treatment was deemed to have failed. Treatment failure was defined as lack of response after 3 to 4 cycles of treatment with MMC, failure to achieve complete clinical resolution after up to 6 months of treatment with IFN, or both. Patients with fewer than 6 months of follow-up after clinical resolution were excluded from this study.
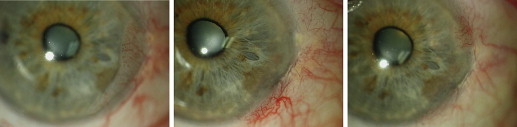
All patients underwent a comprehensive ophthalmic examination at the commencement of treatment and at all follow-up visits, including uncorrected and corrected visual acuity, intraocular pressure measurement, and slit-lamp examination with photography. The diagnosis of OSSN was made by a single ophthalmologist experienced in examining ocular tumors (G.A.L.). In the primary cases, this was performed clinically, relying on the characteristic features of OSSN: gelatinous, papilliform, or leukoplakic lesion with characteristic tufted, superficial, so-called corkscrew vessels. This avoided more invasive incisional biopsy; however, in cases where the clinical diagnosis was uncertain, a scraping or biopsy sample was obtained for histologic diagnosis. Impression cytologic analysis was not available during the course of the study. The location and dimensions of the lesion were recorded at the commencement of treatment, with appropriate measurements obtained using the slit lamp. For purposes of data analysis, the lesions were grouped according to the maximal basal diameter (in millimeters).
The MMC and IFN eye drops used in this study were prepared extemporaneously by the oncology pharmacy of the Royal Brisbane and Women’s Hospital, Brisbane, Australia (2001 through 2009) and the Custom Care Compounding Pharmacy, Sydney, Australia (2010 through 2012) under cytotoxic laminar flow conditions. The MMC (Kyowa Kirin, Berkshire, United Kingdom) was prepared at a concentration of 0.04% by reconstituting 2 mg powder with 5 mL of 0.9% saline, filtered and stored in a sterile plastic bottle. The IFN was prepared at a concentration of 1 million IU/mL by diluting 0.67 mL of 18M IU/1.2 mL injectable recombinant IFN (Intron A; Merck & Co., Inc., Whitehouse Station, New Jersey, USA) with 0.9% saline up to 10 mL, filtered and stored in 3 sterile plastic bottles. All side effects reported by the patients or observed during clinical examination were documented. Patients were administered a regimen of MMC 0.04% 4 times daily for 1 week, followed by 3 weeks off treatment. This treatment cycle was repeated until an end point was reached. If necessary, patients were prescribed lubricant eye drops and dexamethasone 1% 4 times daily to resolve minor redness or irritation. Patients were administered IFN 1 million IU/mL 4 times daily if previous treatment with MMC had failed or if they were unable to tolerate MMC as a result of side effects. Again, this treatment was continued until an end point was reached. Patients who failed to respond to topical treatment underwent surgical excision.
At each follow-up visit, ophthalmic examination and data recording occurred as per the initial consultation. After commencement of treatment, reviews were undertaken every 1 to 2 months until clinical resolution was observed. After achieving clinical resolution, patients were reviewed at 3- to 4-month intervals for the first year, then every 6 months for 1 year, and thereafter annually. Episodes of recurrent disease, as indicated by evidence of active disease on slit-lamp examination or biopsy, were recorded and treated using either further topical MMC, IFN, surgical excision, or a combination thereof.
Statistical Analysis
Univariate and multivariate Cox proportional hazard analyses were used to examine potential factors associated with OSSN recurrence. In cases of bilateral OSSN (n = 8), only the right eye data were included to ensure statistical independence. Factors included in the analyses were patient age and gender, history of previous OSSN treatment (none, MMC interventions, non-MMC interventions), lesion size (<2 mm, 2 to 6 mm, >6 mm), lesion location (conjunctiva, limbus, cornea), treatment type (MMC, IFN), and treatment duration (MMC, number of cycles; IFN, number of months). For the multivariate analysis, a forward stepwise approach was used ( P < .05 for inclusion and P > .10 for exclusion in the model). Kaplan-Meier analysis was used to examine time to OSSN recurrence, and Kaplan-Meier survival curves were compared using the log-rank test. All statistical analyses were performed using SPSS software version 21.0 (IBM Corp, Armonk, New York, USA).
Results
A total of 168 patients who had received treatment with MMC, IFN, or both for OSSN were identified from the 2 pharmacy databases. Of these, 143 eyes of 135 patients (105 men and 30 women) were eligible for inclusion in this study. The reasons for exclusion included follow-up of fewer than 6 months (n = 15), intolerance to either topical medication (n = 6), and for adjunctive treatment in the immediate postoperative period after inadequate excision margin on histologic analysis (n = 12). At commencement of treatment, the mean patient age was 69 years (range, 22 to 91 years). Of the 143 eyes included, 37 (25.9%) were recurrent lesions occurring after previous topical or surgical treatment by other ophthalmologists. All of these lesions as well as another 19 (13.3%) eyes were confirmed by histopathologic examination.
The 143 eyes included in the study achieved initial clinical resolution by using only topical agents. Of the 129 eyes treated with MMC, clinical resolution occurred in 102 (79.1%), with a mean treatment of 3.3 cycles (range, 2 to 6 cycles; Table 1 ). Fewer cycles were required if the patient had undergone previous topical treatment, surgery, or both. The 27 eyes that did not resolve fully with MMC were given topical IFN and subsequently achieved clinical resolution with a mean treatment time of 3.9 months (range, 1 to 8 months). IFN was used as the initial topical treatment in 19 eyes in which the patient either had a history of prior failed treatment with MMC (n = 12) or a poor ocular surface before commencement of treatment (n = 7). Of these 19 eyes given IFN as their initial treatment, clinical resolution was achieved with a mean time of 5 months (range, 2 to 8 months).
| Primary Treatment | Previous MMC | Previous Surgery | Previous Surgery + MMC | |
|---|---|---|---|---|
| MMC only (no. of cycles; n = 102) | 3.3 | 2.5 | 2.7 | 3.0 |
| MMC + IFN (cycles/mos; n = 27) | 3.0/3.9 | 2.0/5.5 | 2.6/4.3 | Nil |
| IFN only (mos; n = 19) | 5.0 | 4.4 | 6.0 | 3.8 |
Adverse effects occurred in 76 (58.9%) patients using MMC and in 14 (30.4%) patients using IFN. The most common side effects reported were conjunctival hyperemia or irritation (MMC, n = 63 [48.8%]; IFN, n = 13 [28.2%]), followed by localized allergic or toxic reactions (defined as papillary conjunctivitis, lid swelling, or both; MMC, n = 12 [9.3%]) and punctal stenosis (MMC, n = 7 [5.4%]). There was a single case of corneal erosion in each of the MMC and IFN groups. Patients occasionally had more than 1 adverse effect. There were no cases of limbal stem cell deficiency identified. Most side effects were mild and were tolerated by patients. Side effects significant enough to result in cessation of treatment before clinical resolution occurred in 12 (8.4%) of 143 eyes; all were with MMC. However, 11 of these subsequently were treated successfully with topical IFN.
During the follow-up period, clinical recurrences were diagnosed in 19 (14.1%) of the 135 patients after topical treatment ( Figure 1 ). Of these 19 patients, 12 patients (12.5%) had no prior treatment (n = 96) and 7 patients (18.0%) previously were treated either topically or surgically (n = 39). Fourteen recurrences (15.1%) were in patients treated only with MMC (n = 93), 4 recurrences (20.0%) were in patients treated with both MMC and IFN (n = 20), and only 1 recurrence (4.6%) was in patients treated only with INF (n = 22). The mean time to recurrence was 17.2 months (range, 4 to 61 months), with 14 patients (73.7%) recurring within a 2-year period. Table 2 summarizes the results of the univariate Cox proportional hazard analysis. There was no significantly greater risk of recurrence associated with lesion size or location, gender, age, treatment duration, or treatment type according to univariate Cox proportional hazard analysis ( P > .05). The only factor potentially associated with an increased risk of OSSN recurrence was previous treatment method. Lesions initially treated with surgical excision alone were associated with a greater risk of recurrence (hazard ratio, 2.61; 95% confidence interval, 0.92 to 7.43) that bordered on statistical significance ( P = .07).
| Factor | HR | 95% CI | P Value a |
|---|---|---|---|
| Age (y) | 1.00 | 0.94 to 1.04 | .70 |
| Gender | |||
| Male (n = 105) | |||
| Female (n = 30) | 1.54 | 0.58 to 4.08 | .38 |
| Previous treatment | |||
| None (n = 96) | |||
| MMC (n = 21) | 0.60 | 0.13 to 2.70 | .51 |
| Surgery alone (n = 18) | 2.61 | 0.92 to 7.43 | .07 |
| Lesion size (mm) | |||
| <2 (n = 16) | |||
| 2 to 6 (n = 60) | 0.40 | 0.11 to 1.48 | .40 |
| >6 (n = 59) | 0.94 | 0.29 to 2.99 | .94 |
| Lesion location | |||
| Conjunctiva (n = 6) | |||
| Limbus (n = 98) | 0.49 | 0.11 to 2.24 | .36 |
| Cornea (n = 31) | 0.82 | 0.17 to 4.12 | .81 |
| Treatment | |||
| MMC (n = 93) | |||
| IFN (n = 22) | 0.27 | 0.04 to 2.02 | .20 |
| MMC and IFN (n = 20) | 1.33 | 0.44 to 4.03 | .62 |
| Treatment duration | |||
| MMC (cycles) | 0.96 | 0.60 to 1.52 | .85 |
| IFN (mos) | 0.46 | 0.05 to 4.33 | .50 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


