Purpose
To evaluate the efficacy of half-dose photodynamic therapy (PDT) targeting only the focal leakage point on fluorescein angiography for acute central serous chorioretinopathy.
Design
Nonrandomized, retrospective, comparative, interventional case series.
Methods
Ten consecutive eyes with acute central serous chorioretinopathy underwent PDT, and later, 11 eyes were observed without treatment. Main outcome measures included achievement of complete resolution of subretinal fluid, change in best-corrected visual acuity, and central retinal sensitivity.
Results
Complete resolution of subretinal fluid was achieved in 80.0% and 18.2% of eyes in the PDT group and the observation group at 1 month ( P = .009), 100% and 27.3% at 3 months ( P = .001), and 90% and 63.6% at 12 months ( P = .311), respectively. At 12 months, 3 eyes (27%) in the observation group had a persistent lesion, whereas no such lesions were observed in the eyes in the PDT group. One eye in each group showed recurrence during the 12-month follow-up period. Visual acuity improved significantly in both groups at each time point, and the differences between groups were not significant. The mean central retinal sensitivity at 3 months was significantly higher in the PDT group compared with the observation group.
Conclusions
Fluorescein angiography-guided half-dose PDT seems to facilitate faster resolution of subretinal fluid in acute central serous chorioretinopathy without, however, convincing long-term anatomic and functional benefits. This protocol may enhance the safety of PDT further.
Central serous chorioretinopathy (CSC) is characterized by serous detachment of the neurosensory retina in the central macula secondary to a focal fluorescein angiographic leak at the level of the retinal pigment epithelium (RPE). However, there is controversy regarding whether the pathologic features of this condition primarily are choroidal or are a disorder of the RPE, and whether they is focal or widespread.
Direct application of a focal laser to the point of RPE leakage results in faster resolution of the detachment. The laser coagulates the diseased RPE cells, inducing scar tissue. The scar then may prevent further subretinal fluid (SRF) accumulation, allowing the surrounding healthy epithelium to pump the fluid back to the choriocapillaris. In contrast, indocyanine green angiography (ICGA) shows multiple choroidal hyperfluorescent areas secondary to choroidal hyperpermeability, supporting the suggestion that the primary pathologic characteristic is choroidal. Subsequently, several studies have demonstrated that photodynamic therapy (PDT) applied to the regions of documented choroidal vascular hyperpermeability on ICGA is effective for the treatment of CSC by inducing local choroidal hypoperfusion. However, conventional PDT for CSC is not without complications and may increase the risk of RPE atrophy, choroidal ischemia, and secondary choroidal neovascularization (CNV). Modification of PDT parameters (half-dose or low-fluence PDT) has been attempted to minimize the risk of complications, while maintaining sufficient photodynamic effects required for treating CSC. Nonetheless, these potential side effects may lead to restriction of the extensive use of PDT for acute CSC, which is usually self-limiting. However, even if eyes with resolved CSC show full recovery of visual acuity, they often show focal areas of reduced retinal function. Furthermore, some cases have multiple recurrent episodes or chronic persistent features, which lead to RPE decompensation and photoreceptor damage, resulting in progressive visual loss.
The histologic features of PDT-treated human eyes demonstrate occlusion of the physiologic choroid within the entire irradiated area. In addition, moderate damage to the overlying RPE was observed. Whether this damage was a direct consequence of the PDT effect or developed secondary to choriocapillary occlusion is uncertain. In the treatment of CSC, transient RPE damage may have a positive effect by inducing subsequent remodeling of RPE cells and recovery from metabolic impairment at the RPE level. With this assumption, the safety of PDT can be promoted by limiting the laser spot size to the focal area of leakage on fluorescein angiography. The present study was performed to evaluate the short-term efficacy of fluorescein angiography-guided PDT with half-dose verteporfin in the treatment of acute CSC in comparison with untreated cases.
Methods
We conducted a retrospective review of the medical records of 21 eyes in 20 consecutive patients diagnosed with acute CSC between June 2011 and December 2011 at the Seoul St. Mary’s Hospital of the Catholic University of Korea in South Korea. Inclusion criteria were as follows: (1) visual symptom for 3 months or fewer, (2) single active focal RPE leakage in fluorescein angiography consistent with acute CSC, and (3) SRF involving the fovea, with or without RPE detachment on optical coherence tomography (OCT). Patients with a history of previous treatment, such as laser photocoagulation, PDT, or intravitreal anti–vascular endothelial growth factor injection, were excluded. Patients who had evidence of chronic CSC, CNV, polypoidal choroidal vasculopathy, or any other maculopathy causing SRF accumulation also were excluded. Eyes were divided into 2 groups: the PDT group, in which patients underwent fluorescein angiography-guided half-dose PDT, and the observation group, in which patients did not receive any treatment for CSC. Ten consecutive eyes were included in the PDT group and 11 eyes were included in the observation group. This nonrandomized, retrospective, comparative, interventional case series was approved by the Institutional Review Board of the Catholic Medical Center. Informed consent was obtained from all patients before PDT.
PDT was performed with a half-dose of verteporfin (3 mg/m 2 ; Visudyne [Novartis AG, Bern, Switzerland]). Verteporfin was administrated over 8 minutes, followed by laser delivery at 689 nm at 10 minutes from the start of infusion. A total light energy of 50 J/cm 2 over 83 seconds was delivered to the location of focal leakage identified by fluorescein angiography using a direct contact lens (Volk TransEquator lens, ×1.44 laser spot magnification; Volk Optical, Mentor, Ohio, USA). The spot size was determined by a single physician (W.K.L.) based on the following policy: the smallest size enough to cover the point of active leakage on early-phase fluorescein angiography. After treatment, all patients were instructed to avoid exposure to strong light for 3 days.
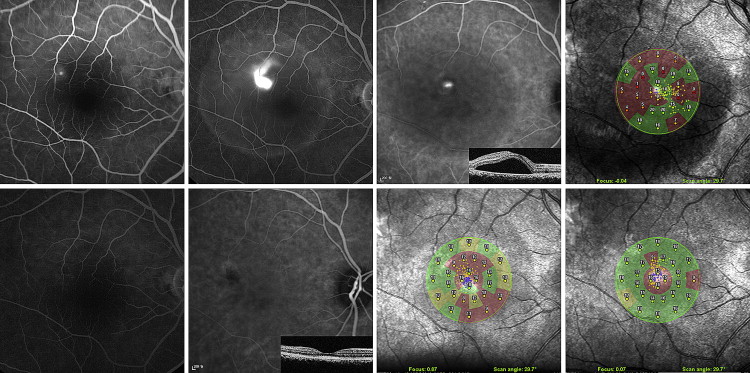
A comprehensive ocular examination, including Snellen best-corrected visual acuity (BCVA) test and dilated fundus examination with slit-lamp biomicroscopy, was performed at baseline and at each follow-up visit. All patients were observed at 1 and 3 months and then were followed up at 1- to 6-month intervals according to OCT findings. OCT (Cirrus OCT [Carl Zeiss Meditec, Dublin, California, USA]) was performed at each visit to determine the presence of SRF and central macular thickness (CMT). Fluorescein angiography and ICGA (Heidelberg Retina Angiograph [Heidelberg Engineering, Heidelberg, Germany]) were performed at baseline and then on an as-needed basis. To evaluate objective visual function, microperimetry (OCT and scanning laser ophthalmoscopy [OPKO/Ophthalmic Technologies, Toronto, Canada]) was performed to determine retinal sensitivity at baseline and at the 1- and 3-month follow-ups. The parameters investigated in this study included Polar 3 test pattern and Goldmann III size stimulus with a duration of 200 ms. The Polar 3 pattern is a circular grid composed of 28 points covering the 12 degrees of the central area. The mean 12-degree central sensitivity was evaluated.
Main outcome measures included the number of eyes achieving complete resolution of SRF, BCVA change, and CMT change at 1, 3, and 12 months after PDT. The recurrence rate was evaluated at 12 months. We also assessed central retinal sensitivity at 1 and 3 months.
For statistical analysis, Snellen visual acuity was converted to the logarithm of the minimal angle of resolution (logMAR) units. A repeated-measures analysis of variance (ANOVA) was used to assess the time course of changes in each parameter over time. If significant differences were observed, the Bonferroni post hoc test for multiple comparisons was performed to find time points showing a significant difference from the baseline value. The clinical parameters at baseline and at 1, 3, and 12 months were compared using the unpaired t test for continuous variables and the χ 2 test for categorical variables. SPSS for Windows software version 19 (SPSS, Inc., Chicago, Illinois, USA) was used for statistical analyses. In all analyses, P < .05 was taken to indicate statistical significance.
Results
Baseline Characteristics
A total of 21 eyes from 20 consecutive patients with acute CSC were included in this study. Thirteen patients were male and 7 were female. The mean age was 42.9 years (range, 31 to 61 years). The mean duration of symptoms was 4.9 ± 3.4 weeks (range, 1 to 12 weeks). The mean number of previous episodes was 0.62 ± 0.67 (range, 0 to 2). On baseline fluorescein angiography, all eyes showed a single focal active leakage point. Sixteen eyes showed ink-blot appearance and 5 had a smokestack appearance. Twelve eyes showed RPE detachment on baseline OCT. Ten eyes (47.6%) were treated with fluorescein angiography-guided half-dose PDT and 11 eyes (52.4%) were followed up without treatment. The size of the laser spot in all eyes of the PDT group was 1100 μm, as determined using a Volk TransEquator lens (the smallest spot size). The mean logMAR BCVA at baseline was 0.27 ± 0.15 (Snellen equivalent, 20/37; range, 20/63 to 20/20) in the PDT group and 0.19 ± 0.12 (Snellen equivalent, 20/31; range, 20/50 to 20/20) in the observation group. The mean CMT was 501 ± 113 μm (range, 329 to 668 μm) in the PDT group and 438 ± 110 μm (range, 272 to 633 μm) in the observation group. The mean 12-degree retinal sensitivity was 11.4 ± 1.2 dB (range, 10.1 to 12.7 dB) in the PDT group and 11.6 ± 1.2 dB (range, 10.0 to 13.3 dB) in the observation group. All patients were followed up for at least 12 months. Table 1 summarizes the baseline characteristics of the patients in the 2 groups. There were no significant differences in age, sex, duration of symptoms, number of previous episodes, fluorescein angiography findings, OCT features, visual acuity, CMT, or retinal sensitivity between the 2 groups.
| Variable | PDT Group (n = 10) | Observation Group (n = 11) | P Value |
|---|---|---|---|
| Age (y) | 42.20 ± 4.83 | 43.45 ± 9.22 | .698 a |
| Gender (male/female) | 6/4 | 8/3 | .659 b |
| Duration of symptoms (wks) | 5.10 ± 3.67 | 4.73 ± 3.35 | .810 a |
| No. of previous episodes | 0.50 ± 0.53 | 0.73 ± 0.79 | .451 a |
| Fluorescein angiography finding | |||
| Leaking appearance (Ink-blot/smokestack) | 7/3 | 9/2 | .635 b |
| Distance from foveal center to leaking point (μm) | 1105 ± 453 | 1080 ± 525 | .909 a |
| OCT features | |||
| SRF + PED/SRF | 5/5 | 7/4 | .670 b |
| BCVA (logMAR) | 0.27 ± 0.15 | 0.19 ± 0.12 | .198 a |
| Central macular thickness (μm) | 501 ± 113 | 438 ± 110 | .212 a |
| 12-degree retinal sensitivity (dB) | 11.4 ± 1.2 | 11.6 ± 1.2 | .693 a |
| PDT laser spot size (μm) | 1100 |
Anatomic Results
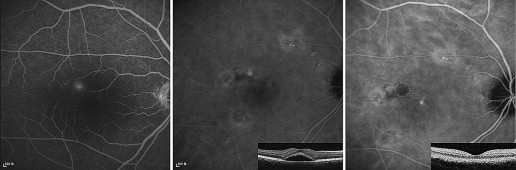
Table 2 summarizes the anatomic and functional results in the 2 groups. After fluorescein angiography-guided PDT, complete resolution of SRF on OCT was achieved in 8 (80.0%) of 10 eyes at 1 month, in all 10 eyes (100%) at 3 months, and in 9 (90%) of 10 eyes at 12 months. In these eyes, posttreatment fluorescein angiography showed no dye leakage ( Figures 1 and 2 ). In the observation group, complete resolution was achieved in 2 (18.2%) of 11 eyes at 1 month, in 3 (27.3%) of 11 eyes at 3 months, and in 7 (63.6%) of 11 eyes at 12 months. The differences between the groups were statistically significant at both 1 month ( P = .009) and 3 months ( P = .001), but not at 12 months ( P = .311). One eye in the PDT group showed SRF because of recurrence between the 9- and 12-month follow-ups (recurrence rate, 1/10 [10%]; Figure 3 ). Among 4 eyes showing SRF in the observation group, 3 eyes had persistent disease and 1 eye had recurrence after spontaneous resolution (recurrence rate, 1/7 [14%]).
| Variable | PDT Group (n = 10) | Observation Group (n = 11) | P Value |
|---|---|---|---|
| Complete resolution of SRF, no. (%) | |||
| 1 Month | 8 (80.0) | 2 (18.2) | .009 a |
| 3 Months | 10 (100.0) | 3 (27.3) | .001 a |
| 12 Months | 9 (90.0) | 7 (63.6) | .311 a |
| Central macular thickness, μm | |||
| Baseline | 501 ± 113 | 438 ± 110 | .212 b |
| 1 Month ( P value c ) | 238 ± 78 (<.001 c ) | 340 ± 86 (.024 c ) | .011 b |
| 3 Months ( P value c ) | 244 ± 108 (<.001 c ) | 340 ± 97 (.336 c ) | .047 b |
| 12 Months ( P value c ) | 231 ± 58 (<.001 c ) | 274 ± 64 (.001 c ) | .178 b |
| P value d | <.001 d | <.001 d | |
| BCVA (logMAR) | |||
| Baseline | 0.27 ± 0.15 | 0.19 ± 0.12 | .198 b |
| 1 Months ( P value c ) | 0.09 ± 0.13 (.001 c ) | 0.12 ± 0.12 (.023 c ) | .605 b |
| 3 Months ( P value c ) | 0.06 ± 0.11 (.001 c ) | 0.10 ± 0.10 (.382 c ) | .388 b |
| 12 Months ( P value c ) | 0.07 ± 0.12 (.006 c ) | 0.12 ± 0.10 (.911 c ) | .315 b |
| P value d | <.001 d | .090 d | |
| 12-degree retinal sensitivity (dB) | |||
| Baseline | 11.4 ± 1.2 | 11.6 ± 1.2 | .693 b |
| 1 Month ( P value c ) | 14.2 ± 1.3 (<.001 c ) | 13.2 ± 1.8 (.023 c ) | .286 b |
| 3 Months ( P value c ) | 16.2 ± 1.3 (.001 c ) | 13.5 ± 2.4 (.034 c ) | .043 b |
| P value d | <.001 d | .007 d |
c Bonferroni post hoc test for multiple comparison between baseline and posttreatment data.