Purpose
To evaluate whether retro-backplate retroprosthetic membrane is correlated with risk of melt in patients with a type I Boston Keratoprosthesis (KPro).
Design
Retrospective, observational case series.
Methods
Study of 50 eyes of 47 patients with type I Boston KPro and postoperative anterior segment optical coherence tomography (AS OCT) imaging performed at the University of Illinois at Chicago. Main outcome measures were presence of retro-backplate membrane and development of melt requiring explantation. For eyes with melt, membrane thickness was measured using the AS OCT images obtained at the last visit before melt occurred. For eyes without melt, the last available AS OCT images were used for measurement.
Results
AS OCT evidence of a retro-backplate membrane was observed in 100% of eyes that melted and in 34.1% of eyes that did not ( P = .0034; risk ratio, 2.9; 95% confidence interval, 1.9 to 4.4). Retro-backplate membrane thickness in the melt group was 278 μm versus 193 μm in the nonmelt group ( P = .025).
Conclusions
The retro-backplate portion of a retroprosthetic membrane is to be differentiated from the retro-optic portion seen at the slit lamp. The retro-backplate membrane as shown by AS OCT imaging is correlated with an increased risk of sterile keratolysis, possibly because of impedance of nutritional support from the aqueous humor. Further studies are needed to better standardize the AS OCT measurements of retro-backplate membranes as well as to identify early interventions to prevent progression of thin membranes once identified on AS OCT.
Despite recent advances in the field of corneal transplantation, options for restoring meaningful vision in patients with corneal disease and poor prognosis for traditional keratoplasty remain limited. The introduction of the type I Boston Keratoprosthesis (KPro; J .G. Machine Company Inc, Woburn, Massachusetts, USA) by Dohlman and associates in 1974 was a major advancement in the management of these challenging patients. Although early studies showed greatly improved visual outcomes for some patients, major complications were reported in a number of cases. Among these, stromal melting (sterile keratolysis) of the carrier corneal button was a complication that frequently resulted in Boston KPro failure and explantation. Initial studies at that time reported an incidence of melt of up to 51% in patients using the original solid backplate keratoprosthesis model. Since then, the type I Boston KPro implant has undergone modifications in an effort to address the significant rates of corneal melt that initially were observed.
The improved durability of the most recent iteration of the Boston KPro is the result of both changes to the device itself as well as modifications to the clinical management of patients following implantation. The success of these changes has helped to demonstrate the importance of multiple factors that play a role in the pathogenesis of melt. The first major breakthrough in improving Boston KPro retention was the introduction of bandage soft contact lenses, which drastically decreased the incidence of corneal surface desiccation and also improved Boston KPro retention. Further success was obtained with the addition of holes to the Boston KPro backplate, which demonstrated the importance of adequate nourishment to the carrier cornea from host aqueous humor. Studies showed a reduction in the rate of melt from 51% to 10% with the addition of 8 backplate holes to the initial solid backplate design. By increasing the surface area for aqueous diffusion to the carrier cornea, 8-hole—and now 16-hole—backplate models have improved the viability of the Boston KPro implant significantly.
However, even with the most recent Boston KPro design, melts still complicate between 1.4% and 17% of Boston KPro implants. Data from recent studies performed at our institution suggest that the development of a retroprosthetic membrane may predispose patients to corneal melt (unpublished data). One possible explanation for this is that in the presence of a retroprosthetic membrane, significant occlusion of backplate holes may occur, which effectively creates a biologically impermeable backplate by preventing diffusion of aqueous into the carrier cornea. This therefore may predispose patients to melt in a similar fashion to what was seen with older solid backplate Boston KPro designs.
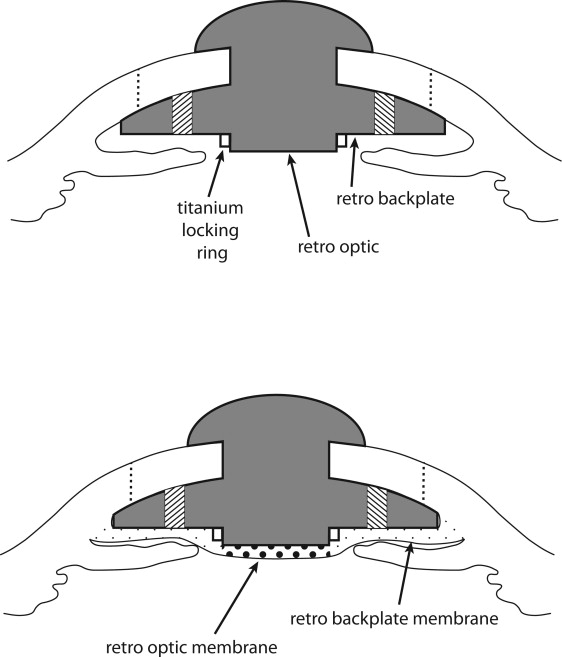
Both histologic and gross evidence from explanted Boston KPros with retroprosthetic membranes suggest that these membranes extend from the peripheral donor cornea and cross the posterior face of the Boston KPro backplate (potentially covering the holes) before becoming clinically evident behind the optic. Thus, some degree of membrane development is likely present in the retro-backplate area before frank development of a retro-optic membrane ( Figure 1 ). Historically, this retro-backplate portion of the retroprosthetic membrane has been underappreciated because only the retro-optic portion results in obscuration of the visual axis in Boston KPro patients. Furthermore, although retro-optic membranes are both evident at the slit lamp and amenable to treatment with the neodymium:yttrium–aluminum–garnet laser, retro-backplate membranes are difficult both to appreciate and to treat. However, because retro-backplate membranes may play a more important role in the pathogenesis of melt by occluding backplate holes and preventing diffusion of vital nutrients from the aqueous humor into the donor cornea, further study is warranted.
With the advent of anterior segment optical coherence tomography (AS OCT), improved characterization of retro-backplate membranes now is possible. The purpose of this study was to evaluate whether the presence and relative thickness of retro-backplate membranes on AS OCT is correlated with risk of melt in patients with a type I Boston KPro.
Methods
After approval from the University of Illinois Institutional Review Board to perform a retrospective review in accordance with Health Insurance Portability and Accountability Act regulations, we identified all patients who underwent Boston KPro implantation at the Illinois Eye and Ear Infirmary from January 2006 through January 2012 with at least 1 AS OCT (Visante Omni; Carl Zeiss Meditec, Dublin, California, USA) study occurring after implantation.
Analysis of Anterior Segment Optical Coherence Tomography Images
For each eye included in the study, demographic data and details of the clinical course, including the indication for Boston KPro implantation, presence of a clinical retro-optic membrane at the slit lamp, and history of Boston KPro melt requiring explantation, were evaluated. Time from Boston KPro implantation to (1) last available AS OCT imaging and (2) final follow-up or Boston KPro melt also was recorded.
In patients with a documented history of Boston KPro melt requiring explantation, the last available AS OCT images obtained before the onset of melt were analyzed further for OCT evidence of a retro-backplate membrane. In patients with no history of melt, the last available AS OCT before final follow-up was analyzed for OCT evidence of a retro-backplate membrane. All images were analyzed by 2 independent reviewers (J.H.H., N.A.).
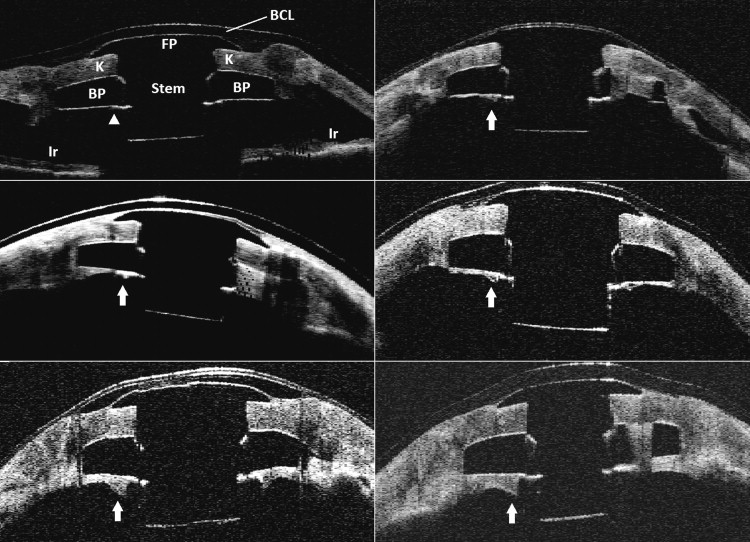
True backplate retroprosthetic membranes were distinguished from artifact at the backplate-aqueous interface or the backplate-titanium locking ring interface by the presence of: (1) a characteristic posterior concavity to the membrane with thickening adjacent to the Boston KPro stem, (2) continuity of the membrane with the donor cornea (which previously was demonstrated on histologic analysis), and (3) discrete shadowing across the membrane at the outer margin of the titanium locking ring ( Figure 2 ).
Analysis of Retro-Backplate Membrane Thickness
Midoptic AS OCT scans at the 180-degree axis were chosen as the standard sequence for comparison because of the large number of patients who had only 180-degree midoptic cut scans and the high frequency of lid shadowing artifacts in many of the vertical and oblique cuts in our study population.
In all eyes with OCT evidence of a retro-backplate membrane, the thickness of the membrane was measured further using 1 of 2 methods. For patients in whom AS OCT images were obtained using standard anterior segment single or quadruple protocols, internal calipers within the Visante operating system were used to measure thickness. For 3 patients, only raw images were available. In these cases, retro-backplate membrane thickness was measured using the caliper functions in ImageJ (National Institutes of Health, Bethesda, Maryland, USA). The known thickness of the backplate at its inner margin adjacent to the Boston KPro stem (0.8 mm) was used as an internal control for determining the final thickness of the retro-backplate membrane.
In all cases, measurements of the retro-backplate membrane were obtained midway between the peripheral edge of the backplate and the edge of the titanium locking ring. All measurements were made on mid-optic scans at the 180-degree axis, obtained from the AS OCT study. When asymmetry in thickness was noted between the retro-backplate membrane at 3 o’clock and the retro-backplate membrane at 9 o’clock, the thicker measurement was recorded preferentially.
Exclusion Criteria
Patients were excluded if: (1) a patient’s AS OCT studies were outdated by more than 15 months relative to date of final follow-up or eventual donor cornea melt, (2) the patients had fewer than 90 days of postoperative follow-up, (3) 180-degree midoptic cuts were not included in the AS OCT studies obtained, or (4) the AS OCT images were deemed poor quality by either of 2 independent reviewers (J.H.H., N.A.). Poor-quality studies were defined as cases in which the posterior surface of the Boston KPro backplate could not be visualized because of inadequate depth of the imaging, motion artifact, or shadowing artifact (ie, from eyelids) or cases in which the retro-backplate membrane could not be distinguished reliably from iris adhesions because of poor image resolution.
Statistical Analysis
A 2-tailed Fisher exact test was used to evaluate whether the presence of a retro-backplate membrane was associated statistically with the development of subsequent Boston KPro melt. A 2-tailed, unpaired t test then was used to evaluate whether the retro-backplate membrane thickness in eyes with documented Boston KPro melt differed significantly from eyes without melt.
Results
In total, 55 eyes of 52 patients met the criteria for inclusion in the study. All patients underwent implantation of a type I Boston KPro with a polymethyl methacrylate backplate. Three eyes were excluded subsequently for inadequate AS OCT quality or depth of imaging, and 2 eyes were excluded for inadequate follow-up. Of the remaining 50 eyes of 47 patients, 12.0% (6/50) were found to have clinical melt that required surgical explantation of the Boston KPro (melt group), whereas 88.0% (44/50) were found to have stable retention of their implants out to final follow-up (nonmelt group). All of the melt patients (6/6) had 7.0-mm diameter backplates, whereas 76.2% of the documented nonmelt patients had 7.0-mm diameter backplates (23.8% had 8.5-mm backplates).
In the melt group, mean time from Boston KPro placement to explantation because of melt was 16.0 months (range, 6.7 to 34.7 months). The mean time from the last available AS OCT study to explantation because of melt was 3.7 months (range, 0.0 to 13.3 months). In the nonmelt group, mean time to final follow-up was 22.0 months (range, 3.0 to 60.0 months). Mean time from the last available AS OCT to final follow-up was 2.6 months (range, 0.0 to 14.4 months). Patient demographics for both groups are shown in Table 1 .
| Melt (n = 6) | Nonmelt (n = 44) | |
|---|---|---|
| Mean age (range), y | 51.2 (3 to 85) | 56.8 (26 to 85) |
| Gender (% male) | 83.3 | 63.0 |
| Indication for KPro | ||
| Chemical injury | 1 | 9 |
| Corneal dystrophies | 0 | 5 |
| Glaucoma | 0 | 5 |
| Failed PKP | 0 | 5 |
| Bullous keratopathy | 0 | 4 |
| Stevens-Johnson syndrome | 0 | 4 |
| Aniridia/LSCD | 3 | 4 |
| Infectious keratitis | 0 | 4 |
| Graft-versus-host disease | 0 | 1 |
| Keratoconus | 0 | 1 |
| Chronic neurotrophic ulcer | 0 | 1 |
| Trauma | 0 | 1 |
| Peters anomaly | 1 | 0 |
| Allergic keratoconjunctivitis | 1 | 0 |
| Mean time to final follow-up or melt (range), mos | 16.0 (6.7 to 34.7) | 22.0 (3.0 to 60.0) |
| Mean time from AS OCT to final follow-up or melt (range), mos | 3.7 (0.0 to 13.3) | 2.6 (0.0 to 14.4) |
Evidence of Retro-Backplate Membranes on Anterior Segment Optical Coherence Tomography
Based on the last available OCT images obtained before melt, 100% of eyes (6/6) in the melt group were found to have AS OCT evidence of a retro-backplate membrane before melt and explantation. Furthermore, at the time of explantation, all eyes in the melt group showed gross evidence of retro-backplate membrane during surgery after removal of the Boston KPro device ( Figure 3 ). In contrast, only 34.1% (15/44) of eyes in the nonmelt group were found to have AS OCT evidence of retro-backplate membrane at or before the final follow-up. This difference was found to be statistically significant ( P = .0034, 2-tailed Fisher exact test). The risk ratio for developing corneal melt in eyes with AS OCT evidence of a retro-backplate membrane further was found to be 2.9 (95% confidence interval, 1.9 to 4.4) compared with eyes with no retro-backplate membrane on AS OCT ( Table 2 ).




