Purpose
To propose a simple, noninvasive retro-mode imaging technique employing confocal scanning laser ophthalmoscopy (cSLO) as a novel imaging modality for the detection of alterations in retinal pigment epithelium (RPE) in central serous chorioretinopathy (CSCR).
Design
Retrospective observational case series.
Methods
A total of 48 eyes from 45 patients at various phases of CSCR were included. All patients underwent spectral-domain optical coherence tomography (SD-OCT) using a volume scan protocol, fluorescein angiography (FA), indocyanine green angiography (IA), and retro-mode imaging by cSLO. We investigated retro-mode images to evaluate RPE status by comparing the results with those of SD-OCT, FA, and IA.
Results
Patients had various phases of CSCR: acute (23 eyes), chronic (17 eyes), and resolved (8 eyes). Retro-mode imaging detected subtle alterations in subretinal fluid (SRF) and RPE, which were undetectable with FA or IA. The correspondence between SD-OCT scanning the entire macula and retro-mode imaging differed according to the chronicity of the disease. In cases with acute CSCR, the correspondence with SD-OCT reached 44.44% in eyes with semicircular pigment epithelial detachment (PED), 33.33% in eyes with low to flat PED, and 35.71% in eyes with RPE protrusion. In cases with chronic CSCR, the correspondence with SD-OCT reached 83.33%, 57.14%, and 85.71% in eyes with semicircular PED, low to flat PED, and RPE protrusion, respectively. Correspondence in cases with resolved CSCR was 100% in eyes with either semicircular or low to flat PED and 83.33% in eyes with RPE protrusion.
Conclusions
Simple and noninvasive retro-mode imaging by cSLO provides improved comprehensive topographic information of RPE alterations in CSCR. Our detailed interpretation may be useful for future research of retro-mode imaging in various macular disorders.
Central serous chorioretinopathy (CSCR) is characterized by serous detachment of the neurosensory retina at the macula and causes blurred vision, micropsia, metamorphopsia, and moderate visual loss in young and middle-aged adults. The pathologic sequence of CSCR has been reported based on data obtained using various imaging modalities including fluorescein angiography (FA), indocyanine green angiography (IA), and optical coherence tomography (OCT).
Choroidal vascular hyperpermeability is probably a primary event in CSCR, which manifests as choroidal hyperfluorescence on IA. Subsequent dysfunction of the retinal pigment epithelium (RPE) appears as focal leakage on FA, and shows various patterns on OCT. Eventually, serous detachment of the neurosensory retina develops. However, even with progressing imaging techniques, there are no specific imaging methods capable of depicting all of the clinical features of CSCR; therefore, the exact pathogenesis remains unclear.
The detection and characterization of various RPE abnormalities is important for the initial diagnosis, for the treatment choice, and for the follow-up of patients with CSCR. However, conventional invasive imaging modalities such as FA and IA cannot provide detailed information about the outer retina and RPE alterations. Spectral-domain OCT (SD-OCT) is currently one of the best available imaging tools. Recent studies based on SD-OCT have demonstrated morphologic abnormalities in the RPE in most CSCR cases, such as pigment epithelial detachment (PED) and protruding or irregular RPE, and this method shows different features depending on the chronicity of CSCR. However, SD-OCT has some limitations regarding the detection of whole CSCR-related abnormalities of the outer retina and RPE in the posterior fundus pole. Essentially, SD-OCT provides cross-sectional images of a limited area, rather than comprehensive topographic fundus images. Therefore, specific protocols for scanning the entire macula are still required. Fundus autofluorescence (FAF), another noninvasive imaging modality, provides topographic information about the metabolic activity of the RPE. However, FAF does not provide detailed anatomic images of the RPE. Technically, FAF requires image processing with averaging of single frames to reduce noise and to obtain optimized contrast.
Infrared imaging has been used for the visualization of deeper retinal structures because it can penetrate deeper layers. Previously, confocal scanning laser ophthalmoscopy in dark-field (indirect) mode was used to visualize deep retinal lesions such as drusen and elevated choroidal neovascularizations. Recently, based on the principles of retro-illumination, retro-mode imaging with infrared lasers (the implementation of which has been made possible by the newly introduced F-10 [Nidek, Gamagori, Japan] confocal scanning laser ophthalmoscope [cSLO]) was used to investigate several retinal pathologies including macular edema secondary to polypoidal choroidal vasculopathy, myopic retinoschisis, drusen, and subthreshold laser scars. These studies implied that retro-mode imaging could be useful in the study of deep retinal pathologies and RPE changes. However, to date, there are no reports describing retro-mode imaging of various RPE pathologies associated with CSCR. Accordingly, the aim of this study was to propose noninvasive retro-mode imaging by cSLO as a novel imaging modality for detecting morphologic alterations of the RPE in CSCR. We also sought to compare the findings obtained using this approach with those obtained by FA, IA, and OCT.
Methods
Patients
For this observational case study, we retrospectively enrolled 48 eyes from 45 consecutive patients with CSCR who were examined at Hanyang University Medical Center from March 1, 2010 to September 30, 2010. Patients were classified into 3 groups (acute, chronic, and resolved CSCR) according to the onset of subjective symptoms and the presence of serous detachment. CSCR patients with an onset of subjective symptoms such as visual disturbance, metamorphopsia, chromatopsia, or micropsia within the past 3 months were defined as acute. Patients who had first experienced subjective systems more than 3 months previously were classified as chronic. Resolved CSCR was defined as disease without sensory retinal detachment as determined by SD-OCT. Patients who had other retinal diseases, including uveitis, severe media opacity, and history of retinal surgery, were excluded from the study.
Imaging Studies
All subjects underwent comprehensive ophthalmologic examinations and imaging studies including fundus photography, SD-OCT (3D OCT-2000; Topcon, Tokyo, Japan), retro-mode with infrared laser, FA, and IA (F-10, Nidek, Gamagori, Japan). All imaging studies were performed on subjects with dilated pupils. The fundus camera incorporated in the SD-OCT device was used to obtain color fundus images. SD-OCT images were taken using a volume scan protocol, which generated a cube of data through a 6-mm 2 grid after acquiring a series of 128 B-scans, each of which was composed of 512 A-scans.
Prior to beginning angiographic studies, retro-mode imaging of the posterior poles was performed with a field of view of 40 degrees, an optical resolution of 16 to 20 μm, and image size of up to 1024 × 720 pixels. Two different retro-mode images per eye were obtained via 2 confocal apertures, which were deviated to the right (“DR” mode) or left sides (“DL” mode). These were obtained consecutively. FA and IA were performed simultaneously in the standard manner using F-10 cSLO. All images were obtained by the same well-trained technician, who applied the same protocol to all patients.
Retro-mode Imaging
F-10 is a newly developed cSLO apparatus that has 4 different wavelengths (blue, 490 nm; green, 532 nm; red, 660 nm; infrared, 790 nm) and 8 apertures (5 confocal apertures and 3 apertures with a central stop). Retro-mode imaging uses infrared laser light because of its ability to penetrate deeper layers, and it consists of a modified version of indirect (dark-field) imaging. Instead of a ring aperture of dark-field mode, retro-mode uses only part of the annular aperture. The annular aperture deviated laterally from the light pathway supplied by the confocal aperture, which collects backscattered light from 1 direction and blocks it from other directions, which creates the shadow to 1 side of the abnormal feature, thereby creating pseudo-3-dimensional (3-D) images ( Figure 1 ) . The shadows of lesions appear differently according to the laterality of the annular aperture. Both right-deviated and left-deviated annular apertures (DR and DL) are used.
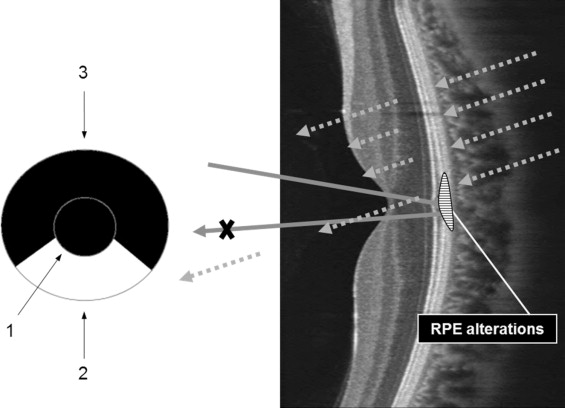
One masked reader (Y.U.S.) reviewed and interpreted each retro-mode image independent of the SD-OCT images. Another masked reader (B.R.L.) compared the findings of all retro-mode images with those obtained by FA, IA, and SD-OCT. Every single B-scan image obtained by a volume scan protocol of SD-OCT was reviewed to detect RPE lesions. In addition, we described the findings of various RPE morphologic alterations on retro-mode imaging according to the chronicity of CSCR.
Results
A total of 35 men and 10 women ranging in age from 33 to 68 years (mean age 50.88 ± 9.51 years) were enrolled in our retrospective study. The sequential involvement of both eyes was demonstrated in 3 patients; thus, 48 eyes were included. Patients had various phases of CSCR at the initial visit: acute CSCR (23 eyes, 47.9%), chronic CSCR (17 eyes, 35.4%), and resolved CSCR (8 eyes, 16.7%). Typical FA leakage patterns (smokestacks or inkblots) were observed in 35 eyes (72.9%), and nonspecific hyperfluorescent patterns without active leakage were observed in 13 eyes (27.1%). In IA, choroidal vascular hyperpermeability appearing as hyperfluorescent patterns was observed in 42 eyes (87.5%). SD-OCT revealed the presence of subretinal fluid (SRF) in 40 eyes (83.3%), semicircular PED in 18 eyes (37.5%), low to flat PED in 11 eyes (22.92%), RPE protrusion in 25 eyes (52.08%), and subretinal precipitations in 18 eyes (37.5%). The leakage patterns provided by angiographic studies did not show detailed RPE status like SD-OCT ( Supplemental Table , available at AJO.com ). In contrast, retro-mode imaging detected subtle SRF and RPE alterations, which were invisible on FA or IA.
The overall correspondence between the findings of retro-mode imaging and SD-OCT reached 97.9% in patients with SRF, 64.7% in patients with semicircular pigment PED, 54.55% in patients with low to flat PED, 51.85% in patients with RPE protrusion, and 38.89% in patients with precipitations. This correspondence differed according to the chronicity of the disease ( Table 1 ). In cases with acute CSCR, the correspondence between the 2 methods was 44.44% in eyes with semicircular PED, 33.33% in eyes with low to flat PED, and 35.71% in eyes with RPE protrusion. In cases with chronic CSCR, correspondence was 83.33% in eyes with semicircular PED, 57.14% in eyes with low to flat PED, and 85.71% in eyes with RPE protrusion. In cases with resolved CSCR, correspondence was 100% in eyes with either semicircular PED or low to flat PED and 83.33% in eyes with RPE protrusion. The correspondence was also influenced by the amount of SRF. Retro-mode imaging detected RPE abnormalities in more cases with shallow or no SRF than in cases with bullous SRF ( Table 2 ).
| Acute CSCR | Chronic CSCR | Resolved CSCR | ||||
|---|---|---|---|---|---|---|
| Number of Eyes a | Correspondence (%) b | Number of Eyes a | Correspondence (%) b | Number of Eyes a | Correspondence (%) b | |
| Semicircular PED | 3 | 44.44 | 5 | 83.33 | 3 | 100.00 |
| Low to flat PED | 1 | 33.33 | 4 | 57.14 | 1 | 100.00 |
| RPE protrusion | 5 | 35.71 | 6 | 85.71 | 1 | 83.33 |
| Subretinal precipitations | 2 | 28.57 | 5 | 45.45 | 0 | 0.00 |
a The number of eyes detected in the retro-mode imaging.
b Correspondence between spectral-domain optical coherence tomography and retro-mode imaging.
| OCT Findings | |||||
|---|---|---|---|---|---|
| SRF | Semicircular PED | RPE Protrusion | Low to Flat PED | Subretinal Precipitations | |
| Correspondence between OCT and retro-mode imaging | Bullous | 54.5% | 30.77% | 33.33% | 22.22% |
| Shallow | 75.0% | 61.00% | 60.00% | 55.56% | |
| No SRF | 100.0% | 83.33% | 100.00% | — | |
| Total | 64.7% | 51.85% | 54.55% | 38.89% | |
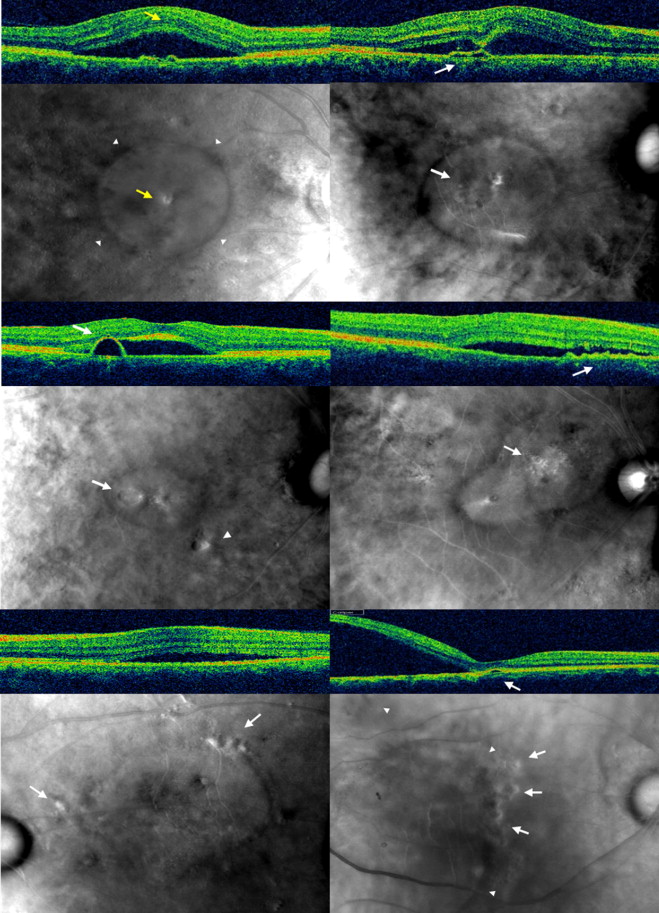
Retro-mode imaging detected the presence of SRF and revealed relatively clear demarcation of the SRF in all cases, with the exception of 2 eyes with very limited SRF. RPE alterations that were relatively small compared to the height of the SRF, if located within the SRF, were not easily visualized using retro-mode imaging ( Figure 2 , Top left). However, RPE alterations located at the margin of the SRF or outside the SRF were easily detected even in cases with bullous SRF ( Figure 2 , Bottom right). Detailed clinical characteristics of enrolled patients were shown in the Supplemental Table (available at AJO.com ).
Description of Subretinal Pathologies Using Retro-mode Imaging
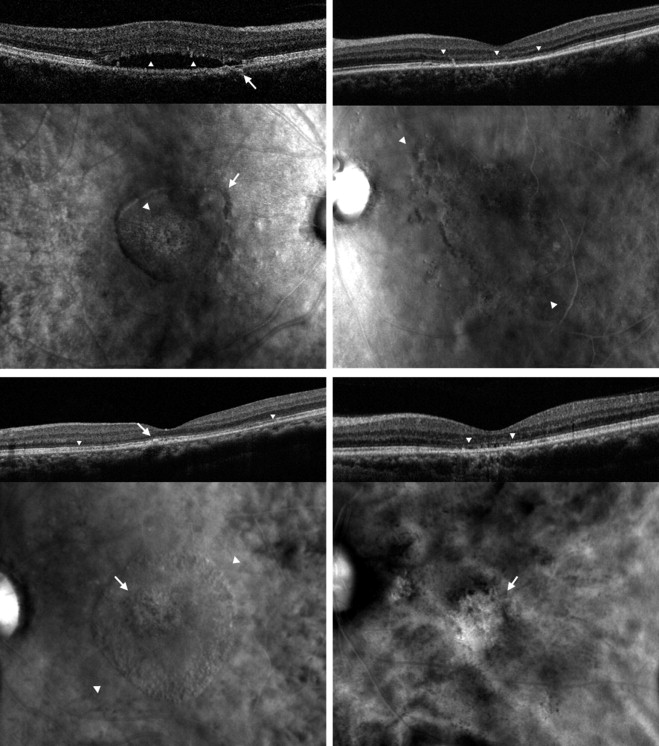
Retro-mode imaging identified SRF as a well-defined, lowly to moderately convex, circular to ovoid, translucent prominence with a dark shadow ( Figure 2 , Top left). Semicircular PED was identified as a well-defined, lowly to moderately convex, circular to slightly irregular, translucent prominence with a dark shadow ( Figure 2 , Middle left). Low to flat PED and RPE protrusion were identified as a well- to ill-defined, flat to lowly convex, slightly irregular, translucent to opaque prominence with a dark shadow ( Figure 2 , Top right and Middle right respectively). Subretinal precipitations appeared as multiple small, relatively hyperreflective dots ( Figure 3 , Top left). In cases of resolved CSCR, disrupted outer photoreceptors appeared as a well-defined, ragged, platy surface area. ( Figure 3 , Top right, Bottom).
Results
A total of 35 men and 10 women ranging in age from 33 to 68 years (mean age 50.88 ± 9.51 years) were enrolled in our retrospective study. The sequential involvement of both eyes was demonstrated in 3 patients; thus, 48 eyes were included. Patients had various phases of CSCR at the initial visit: acute CSCR (23 eyes, 47.9%), chronic CSCR (17 eyes, 35.4%), and resolved CSCR (8 eyes, 16.7%). Typical FA leakage patterns (smokestacks or inkblots) were observed in 35 eyes (72.9%), and nonspecific hyperfluorescent patterns without active leakage were observed in 13 eyes (27.1%). In IA, choroidal vascular hyperpermeability appearing as hyperfluorescent patterns was observed in 42 eyes (87.5%). SD-OCT revealed the presence of subretinal fluid (SRF) in 40 eyes (83.3%), semicircular PED in 18 eyes (37.5%), low to flat PED in 11 eyes (22.92%), RPE protrusion in 25 eyes (52.08%), and subretinal precipitations in 18 eyes (37.5%). The leakage patterns provided by angiographic studies did not show detailed RPE status like SD-OCT ( Supplemental Table , available at AJO.com ). In contrast, retro-mode imaging detected subtle SRF and RPE alterations, which were invisible on FA or IA.
The overall correspondence between the findings of retro-mode imaging and SD-OCT reached 97.9% in patients with SRF, 64.7% in patients with semicircular pigment PED, 54.55% in patients with low to flat PED, 51.85% in patients with RPE protrusion, and 38.89% in patients with precipitations. This correspondence differed according to the chronicity of the disease ( Table 1 ). In cases with acute CSCR, the correspondence between the 2 methods was 44.44% in eyes with semicircular PED, 33.33% in eyes with low to flat PED, and 35.71% in eyes with RPE protrusion. In cases with chronic CSCR, correspondence was 83.33% in eyes with semicircular PED, 57.14% in eyes with low to flat PED, and 85.71% in eyes with RPE protrusion. In cases with resolved CSCR, correspondence was 100% in eyes with either semicircular PED or low to flat PED and 83.33% in eyes with RPE protrusion. The correspondence was also influenced by the amount of SRF. Retro-mode imaging detected RPE abnormalities in more cases with shallow or no SRF than in cases with bullous SRF ( Table 2 ).



