Purpose
To investigate factors affecting patient response to intravitreal ranibizumab treatment for age-related macular degeneration (AMD).
Design
Retrospective chart review.
Methods
We reviewed medical records of 105 consecutive eyes with AMD treated with intravitreal ranibizumab injections and followed for more than 1 year after treatment. Response to ranibizumab treatment was compared between typical neovascular AMD and polypoidal choroidal vasculopathy (PCV). Furthermore, we investigated associations of age, lesion size, and single nucleotide polymorphisms (SNPs) in CFH and ARMS2 genes with treatment response.
Results
Forty-nine eyes were diagnosed with typical neovascular AMD and 56 eyes with PCV. Serous retinal detachment and retinal edema resolved similarly in both typical neovascular AMD and PCV after treatment. However, visual acuity (VA) significantly improved in eyes with PCV, whereas VA was maintained in typical neovascular AMD. At the third and twelfth months after injection, VA was better in PCV than in typical neovascular AMD ( P = .027 and P = .044, respectively), although there were no differences in baseline VA between the 2 groups. Age and size of greatest linear dimension were significantly associated with visual prognosis in typical neovascular AMD but not in PCV. There was no clear association between 3 SNPs and responsiveness to ranibizumab treatment.
Conclusions
Although exudative changes were equivalent following ranibizumab treatment in both typical neovascular AMD and PCV, there was a significant increase in VA in PCV compared to typical neovascular AMD. Age and greatest linear dimension correlated with visual prognosis only in typical neovascular AMD and not in PCV.
Age-related macular degeneration (AMD) is the leading cause of severe visual impairment in industrialized countries in people over 50 years of age. However, anti–vascular endothelial growth factor (VEGF) treatment, such as bevacizumab or ranibizumab, has dramatically improved visual prognosis in patients suffering from neovascular AMD. After numerous reports of favorable results following anti-VEGF treatment for neovascular AMD, anti-VEGF treatment has been extended to treat eyes with polypoidal choroidal vasculopathy (PCV), a subtype of neovascular AMD. Although some reports show that PCV is refractory to anti-VEGF treatment, recent studies have demonstrated improvements in visual acuity (VA) after anti-VEGF treatment for PCV.
Recently, increasing numbers of studies have compared the characteristics of PCV and typical AMD. Maruko and associates demonstrated that Japanese patients with neovascular AMD could be further characterized into subtypes including PCV (54.7%), typical neovascular AMD (35.3%), retinal angiomatous proliferation (4.5%), and PCV + typical neovascular AMD (5.5%). They included predominantly classic choroidal neovascularization (CNV), minimally classic CNV, and occult with no classic CNV into typical neovascular AMD. It has been reported that there is greater VA improvement in PCV compared to typical neovascular AMD after photodynamic therapy (PDT). Furthermore, we have previously shown that there are significant differences in the genetic associations involved in the development of typical neovascular AMD and PCV. For instance, the ARMS2 gene is more strongly related to typical neovascular AMD development than PCV, whereas there is no significant difference in the association of the CFH gene with typical neovascular AMD or PCV.
In addition to disease development, recent studies have examined genetic associations with various treatments for AMD and PCV; several studies have shown a significant association between ARMS2/HTRA1 and visual outcome in eyes with AMD and PCV after PDT, whereas a definitive association with CFH could not be found. The association of the aforementioned genes with response to ranibizumab treatment is still controversial. In addition to genetic associations, baseline VA, CNV lesion size, and age are important predictors of VA outcomes after ranibizumab treatment for AMD. However, it is not clear if these factors are associated with VA outcomes after ranibizumab treatment for PCV.
In the present study, we aimed to elucidate predictive factors of response to ranibizumab in neovascular AMD. At first, we compared response to ranibizumab treatment between typical neovascular AMD and PCV. Furthermore, we evaluated the correlation of baseline VA, age, and lesion size to VA outcome after ranibizumab treatment in typical neovascular AMD and PCV patients. In addition, we investigated the association of 3 major AMD-susceptibility single nucleotide polymorphisms (SNPs) in the CFH (Y402H, I62V) and ARMS2 (A69S) genes and attempted to correlate their presence with response to ranibizumab treatment.
Methods
We retrospectively reviewed the medical records of 105 eyes from 105 consecutive patients with subfoveal neovascular AMD. All patients were treated with 3 loading intravitreal injections of 0.5 mg ranibizumab (Lucentis; Novartis, Bülach, Switzerland) at 1-month intervals and were followed up for more than 12 months after the initial treatment at Kyoto University Hospital. Before treatment, all patients underwent a complete ophthalmologic examination, including measurement of best-corrected visual acuity (VA), intraocular pressure testing, indirect ophthalmoscopy, slit-lamp biomicroscopy with a contact lens, spectral-domain optical coherence tomography (OCT) (Spectralis HRA+OCT; Heidelberg Engineering, Heidelberg, Germany), and fluorescein and indocyanine green angiography (HRA-2; Heidelberg Engineering). Best-corrected VA was measured using a Landolt chart and converted to a logarithm of the minimal angle of resolution (logMAR) for statistical analysis. The diagnosis of PCV was based on indocyanine green angiography, which revealed a branching vascular network terminating in polypoidal swellings. Typical AMD involved classic CNV, occult CNV, or a combination of both. Greatest linear dimension was also determined by indocyanine green angiography.
Ranibizumab injections were administered in a sterile manner, and prophylactic topical antibiotics were applied regularly for 1 week after the injection. After the 3 loading injections, patients were followed up every month, and retreatments were performed as required when VA declined more than 0.2 logMAR along with signs of exudation on OCT or angiography, when retinal thickness increased greater than 100 μm, or if subretinal fluid, subretinal hemorrhage, or active CNV persisted or developed. Photodynamic therapy (PDT) was administered to some eyes whose retinal edema or subretinal fluid did not decrease after initial ranibizumab treatment; we judged that those eyes were resistant to ranibizumab.
Genotyping was performed in 78 patients. Genomic DNA was prepared from patients’ peripheral blood using a DNA extraction kit (QuickGene-610L; Fujifilm, Minato, Tokyo, Japan). CFH Y402H rs1061170, I62V rs800292, and ARMS2 A69S rs10490924 were genotyped using the Taqman SNP assay with the ABI PRISM 7700 system (Applied Biosystems, Foster City, California, USA).
All values are presented as mean ± standard deviation. Patient age, baseline greatest linear dimension, and visual acuity at baseline, 3 months, and 12 months were compared using unpaired t test between typical neovascular AMD and PCV. Sex ratio, baseline existence rate of retinal edema and serous retinal detachment, and disappearance rate of retinal exudative change during follow-up were compared using χ 2 test between typical neovascular AMD and PCV. Visual acuity change during follow-up was evaluated using a paired t test. Associations of baseline visual acuity, patient age, and greatest linear dimension to visual acuity at 3 months and 12 months and visual acuity change during follow-up were evaluated with the Pearson correlation test. Associations of genotypes to visual acuity at 3 months and 12 months and visual acuity change during follow-up were evaluated with analysis of variance and post hoc comparisons of Scheffe’s procedure, and associations to disappearance of retinal exudative change at 3 months and 12 months were evaluated with χ 2 test for trend. P values of less than .05 were considered statistically significant.
Results
Demographics of the study population are shown in Table 1 . Of the 105 eyes evaluated, 49 had typical neovascular AMD and 56 had PCV. Mean age was not significantly different between the 2 groups. All eyes presented with an exudative change attributable to AMD: retinal edema was seen in 35 of 49 eyes (71.4%) with typical neovascular AMD and in 28 of 56 eyes (50%) with PCV and serous retinal detachment was seen in 34 of 49 eyes (69.4%) with typical neovascular AMD and 41 of 56 eyes (73.2%) with PCV. All exudative features revealed by OCT resolved in 32 of 49 eyes (65.3%) with typical neovascular AMD and 35 of 56 eyes (62.5%) with PCV at the third month, and in 34 of 49 eyes (69.4%) with typical neovascular AMD and 31 of 56 eyes (55.4%) with PCV at the twelfth month. There were no significant differences between typical neovascular AMD and PCV with respect to the effectiveness of ranibizumab to resolve retinal exudative change ( P = .77 and P = .14, respectively).
| Typical Neovascular AMD | PCV | P | |
|---|---|---|---|
| Number of eyes | 49 | 56 | |
| Age (years) | 75.9 ± 8.8 | 74.2 ± 8.6 | NS |
| Sex (male/female) | 34/15 | 41/15 | NS |
| Baseline retinal exudative change | |||
| GLD (μm) | 4490.4 ± 273.9 | 3988.1 ± 322.6 | NS |
| Retinal edema | 71.2% | 50.0% | .025 |
| SRD | 69.3% | 73.2% | NS |
| Disappearance of retinal exudative change | |||
| 3 months | 65.3% | 62.5% | NS |
| 12 months | 69.4% | 55.3% | NS |
| Visual acuity (logMAR) | |||
| Baseline | 0.56 ± 0.42 | 0.48 ± 0.41 | NS |
| 3 months | 0.57 ± 0.48 | 0.38 ± 0.37 a | .027 |
| 12 months | 0.60 ± 0.53 | 0.40 ± 0.47 b | .044 |
a P < .01 compared with baseline.
Although there were no differences in baseline VA between typical neovascular AMD and PCV ( P = .29), VAs were better in PCV than in AMD at the third and twelfth months ( P = .027 and P = .044, respectively). So we compared VA change between typical neovascular AMD and PCV. In eyes with PCV, after the first treatment, VA significantly improved at the third month ( P = .002) and at the twelfth month ( P = .028), whereas in typical neovascular AMD, VA was stable at the third month ( P = .79) and at the twelfth month ( P = .23).
Twelve eyes with typical neovascular AMD and 15 eyes with PCV had been previously treated with anti-VEGF therapy, and 2 eyes with typical neovascular AMD and 7 eyes with PCV had been previously treated with PDT. After the 3 loading injections, an average of 1.37 ± 1.52 and 1.70 ± 1.88 injections were added to the treatment of patients with typical neovascular AMD and PCV, respectively, during the 1-year follow-up period. There was no significant difference in the frequency of additional treatments between the 2 groups ( P = .33). Seven of 49 eyes (14.3%) with typical neovascular AMD and 8 of 56 eyes (14.3%) with PCV were treated with PDT after the initial treatment because ranibizumab treatment did not decrease the retinal edema or subretinal fluid. The number of eyes resistant to ranibizumab treatment was not significantly different between patients with typical neovascular AMD or PCV ( P > .99). In the 7 typical neovascular AMD eyes and the 8 PCV eyes treated with PDT, the average VA did not significantly change at the third month ( P = .96 and P = .27, respectively) and the twelfth month ( P = .55 and P = .60, respectively). VA declined more than 0.2 logMAR in 1 or 2 eyes with typical neovascular AMD and PCV at the third month and twelfth month, while VA remained unchanged in most eyes.
Since the MARINA and ANCHOR studies have shown that important predictors of VA outcomes in AMD after ranibizumab treatment are baseline VA, CNV lesion size, and patient age, we evaluated the correlations of baseline VA, CNV lesion size, and patient age to the visual prognosis. The Pearson correlation test revealed significant correlation between baseline VA and the VA at the third month in both the typical neovascular AMD and PCV groups ( P < .0001). Better baseline VA resulted in better VA outcome after ranibizumab treatment. Similarly, VA at twelfth month was significantly correlated with baseline VA ( P < .0001).
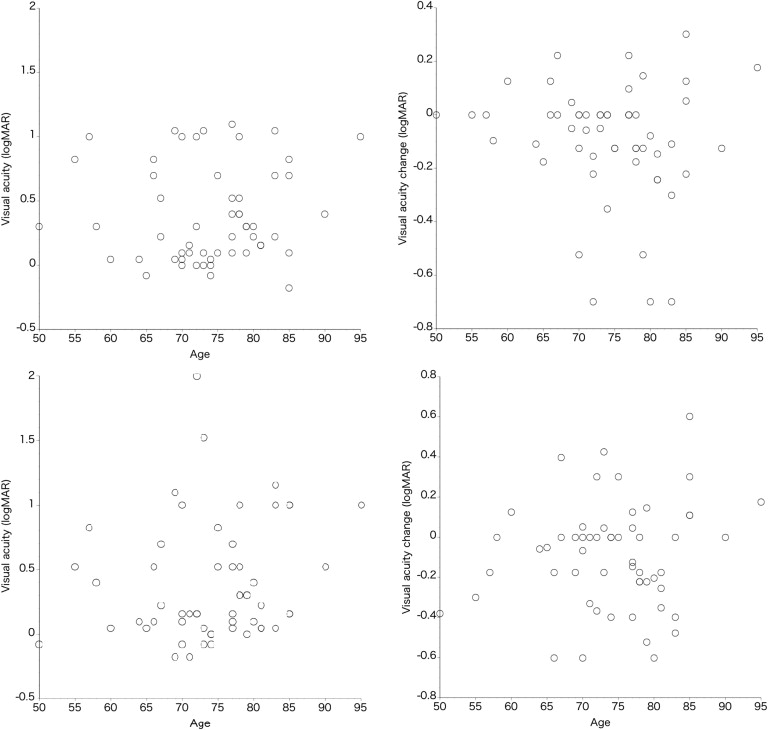
In patients with typical neovascular AMD, patient age was significantly associated with VA change at the third month, VA at the twelfth month, and VA change at the twelfth month ( Figure 1 , P = .040, P = .020, and P = .0014, respectively). In contrast, the age of patients with PCV was not associated with VA or VA change at the third and twelfth months ( Figure 2 , P > .22).
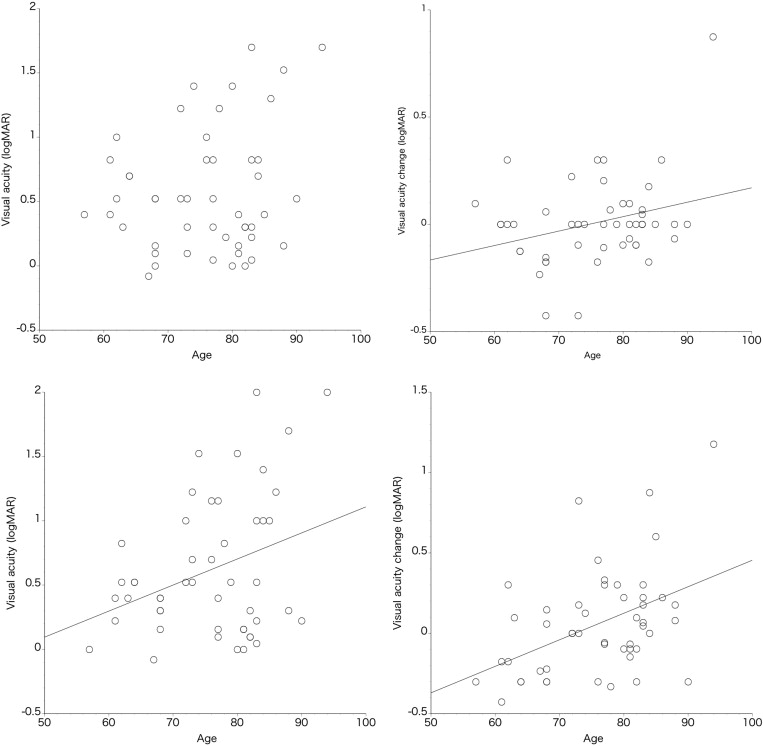
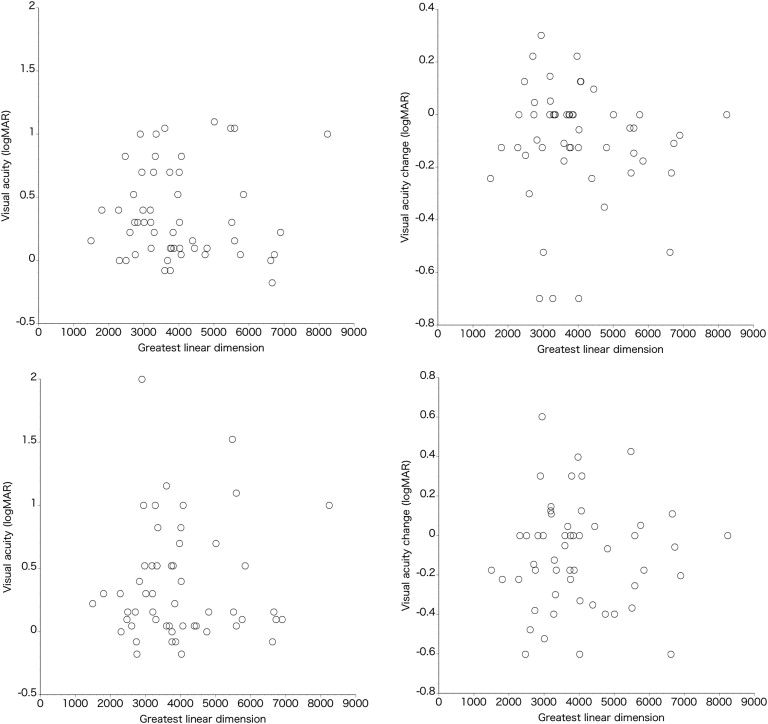
The association of greatest linear dimension with VA or VA change showed a trend similar to that of the aforementioned age association. In patients with typical neovascular AMD, the greatest linear dimension showed a significant association with VA at the third month and twelfth month, and VA change at the twelfth month ( Figure 3 ; P = .015, P = .0004, and P = .0021, respectively). In contrast, greatest linear dimension in patients with PCV was not associated with VA or VA change at either the third or twelfth month ( Figure 4 , P > .63).
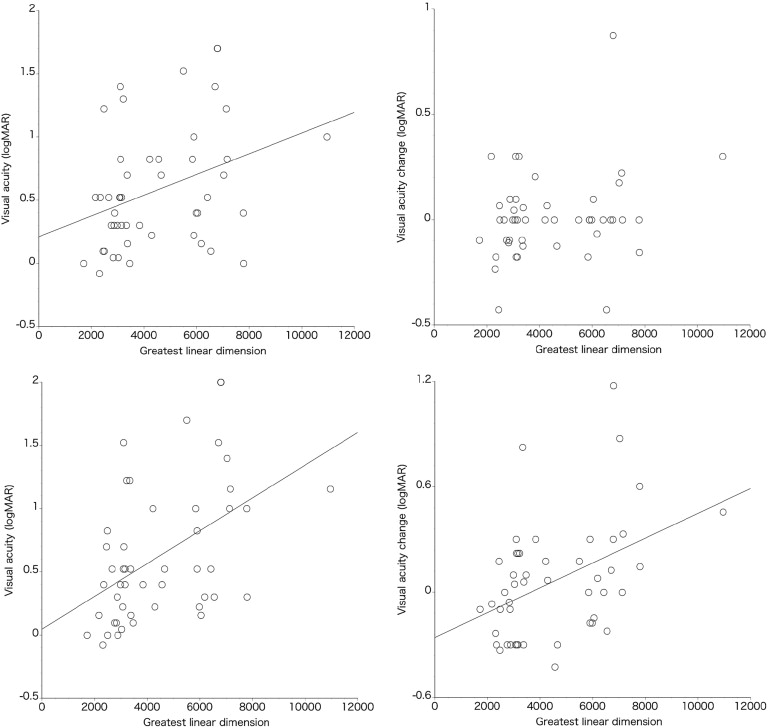
Finally, we evaluated the association of genetic polymorphisms with the treatment response in AMD. For this analysis, typical neovascular AMD and PCV were first combined for evaluation and then separated for further evaluation. The associations of SNPs (CFH Y402H, I62V, and ARMS2 A69S) with VA and/or VA change are shown in Table 2 . There was no significant association between these SNPs and baseline VA, VA or VA change at the third month, and VA or VA change at the twelfth month, except for the association between CFH I62V and VA change at the third month; eyes with a GA genotype had better VA change than those with a GG genotype ( P = .0009). After Bonferroni’s correction, the association of I62V with VA change at the third months became weaker ( P = .027). Furthermore, there was not an association trend such as GG > GA > AA or GG < GA < AA. In the OCT findings, the 3 SNPs were not significantly associated with the resolution of retinal exudative change at either the third or twelfth month ( Table 3 ). When patients with typical neovascular AMD and PCV were evaluated separately, the 3 SNPs did not show any association with VA, VA change, or the resolution rate of retinal exudation.
| VA | VA Change | ||||
|---|---|---|---|---|---|
| Baseline | 3 Months | 12 Months | 3 Months | 12 Months | |
| Y402H-CC (n = 3, 4.0%) | 0.52 ± 0.40 | 0.47 ± 0.42 | 0.51 ± 0.50 | −0.04 ± 0.24 | −0.01 ± 0.33 |
| Y402H-CT (n = 19, 25.3%) | 0.50 ± 0.39 | 0.49 ± 0.42 | 0.52 ± 0.46 | −0.02 ± 0.12 | 0.03 ± 0.28 |
| Y402H-TT(n = 53, 70.7%) | 0.89 ± 0.77 | 0.93 ± 0.80 | 0.94 ± 1.09 | 0.04 ± 0.12 | 0.03 ± 0.33 |
| P | NS | NS | NS | NS | NS |
| I62V-GG (n = 47, 65.3%) | 0.49 ± 0.39 | 0.51 ± 0.46 | 0.52 ± 0.54 | 0.23 ± 0.19 | 0.05 ± 0.34 |
| I62V-GA (n = 21, 29.1%) | 0.58 ± 0.42 | 0.40 ± 0.37 | 0.49 ± 0.40 | −0.18 ± 0.24 | −0.09 ± 0.28 |
| I62V-AA (n = 4, 5.6%) | 0.53 ± 0.27 | 0.48 ± 0.17 | 0.43 ± 0.34 | −0.05 ± 0.12 | −0.10 ± 0.18 |
| P | NS | NS | NS | .0009 | NS |
| A69S-TT (n = 33, 44.0%) | 0.58 ± 0.45 | 0.56 ± 0.50 | 0.61 ± 0.57 | −0.01 ± 0.25 | 0.03 ± 0.33 |
| A69S-TG (n = 26, 34.7%) | 0.41 ± 0.34 | 0.38 ± 0.35 | 0.39 ± 0.41 | −0.03 ± 0.19 | −0.01 ± 0.27 |
| A69S-GG (n = 16, 21.3%) | 0.62 ± 0.40 | 0.55 ± 0.42 | 0.58 ± 0.53 | −0.07 ± 0.20 | −0.03 ± 0.36 |
| P | NS | NS | NS | NS | NS |
| Retinal Exudative Change | ||||
|---|---|---|---|---|
| 3 Months | 12 Months | |||
| Resolved | Remained | Resolved | Remained | |
| Y402H-CC (n = 3, 4.0%) | 2 | 1 | 3 | 0 |
| Y402H-CT (n = 19, 25.3%) | 14 | 5 | 10 | 9 |
| Y402H-TT (n = 53, 70.7%) | 35 | 18 | 34 | 18 |
| P | NS | NS | NS | NS |
| I62V-GG (n = 47, 65.3%) | 30 | 17 | 30 | 16 |
| I62V-GA (n = 21, 29.1%) | 16 | 5 | 12 | 9 |
| I62V-AA (n = 4, 5.6%) | 3 | 1 | 2 | 2 |
| P | NS | NS | NS | NS |
| A69S-TT (n = 33, 44.0%) | 23 | 10 | 18 | 14 |
| A69S-TG (n = 26, 34.7%) | 21 | 5 | 20 | 9 |
| A69S-GG (n = 16, 21.3%) | 7 | 9 | 9 | 7 |
| P | NS | NS | NS | NS |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


