Purpose
To evaluate relationships between retinal vessel caliber, AIDS-related factors, and mortality.
Design
Longitudinal, observational cohort study.
Methods
We evaluated data for participants without ocular opportunistic infections at initial examination (baseline) in the Longitudinal Studies of the Ocular Complications of AIDS (1998–2008). Semi-automated evaluation of fundus photographs (1 eye/participant) determined central retinal artery equivalent (CRAE), central retinal vein equivalent (CRVE), and arteriole-to-venule ratio (AVR) at baseline. Multiple linear regression models, using forward selection, identified independent relationships between indices and various host- and disease-related variables.
Results
Included were 1250 participants. Mean follow-up for determination of mortality was 6.1 years. Smaller CRAE was related to increased age ( P < .001) and hypertension ( P < .001); larger CRAE was related to lower hematocrit ( P = .002). Larger CRAE and CRVE were associated with black race ( P < .001). Larger CRVE was related to smoking ( P = .004); smaller CRVE was related to age ( P < .001) and higher mean corpuscular volume ( P = .001). We observed the following relationships with AIDS-associated factors: smaller CRAE and larger CRVE with history of highly active antiretroviral therapy (HAART; P < .001); and larger CRAE with lower CD4+ T lymphocyte count ( P = .04). We did not identify independent relationships with human immunodeficiency virus RNA blood levels. There was a 12% (95% CI, 2%–21%) increase in mortality risk per quartile of decreasing AVR ( P = .02).
Conclusions
Variations in retinal vascular caliber are associated with AIDS-specific factors and are markers for increased mortality risk. Relationships are consistent with the hypothesis that the vasculature is altered by known atherogenic effects of chronic HAART or the prolonged inflammatory state associated with AIDS.
Advances in digital image analysis software have enabled large, population-based studies to measure retinal vessel caliber reliably and reproducibly as a potent biomarker of vascular disease. Morphologic variations of retinal arterioles and venules are likely to reflect variations in cerebral and coronary vessels, with which they are anatomically and physiologically similar. Abnormalities in vessel caliber have been described in many disease states, including hypertension, diabetes mellitus, coronary artery disease, stroke, and renal dysfunction. Variations in the retinal vascular caliber have not been studied among people with Acquired Immunodeficiency Syndrome (AIDS), although this population is known to be at increased risk for cardiovascular morbidity.
In this study, we investigated relationships between vessel caliber indices and demographic, medical, and laboratory characteristics of participants in the Longitudinal Study of the Ocular Complications of AIDS (LSOCA). Based on relationships seen in the setting of other diseases, we hypothesize that narrower arterioles and more dilated venules will be related to systemic morbidity and mortality.
Methods
Patient Population
LSOCA is an NIH-sponsored prospective epidemiologic study of people with AIDS, which began in September 1998. A description of its design and methods and a summary of data for study participants at study enrollment (baseline) have been published previously. Data were collected from study participants every 6 months per protocol. The current study includes data collected through December 31, 2008 for participants without ocular opportunistic infections at baseline.
Data Collection
We collected the following baseline demographic, medical, and laboratory information: age, sex, race/ethnicity (self-reported), hemoglobin, hematocrit, mean corpuscular volume (MCV), platelet count, leukocyte count, and absolute neutrophil count. We also collected the following AIDS-related information at baseline: time since AIDS diagnosis, lymphopenia as AIDS-defining illness, CD4+ T lymphocyte count (baseline and nadir), CD8+ T lymphocyte count, human immunodeficiency virus (HIV) RNA blood level (baseline and peak), use of highly active antiretroviral therapy (HAART) (on HAART at baseline; ever on HAART), and Karnofsky score (a global measure of one’s ability to conduct normal activities). We identified the following potential comorbidities at baseline: history of smoking, hypertension, diabetes mellitus, renal disease, coronary heart disease, peripheral vascular disease, and stroke.
Vessel caliber indices were determined in a semi-automated manner by certified graders at the University of Wisconsin Fundus Photograph Reading Center (IVAN software; Department of Ophthalmology and Visual Science, University of Wisconsin, Madison, Wisconsin, USA) using a standardized protocol, described previously. Briefly, the 6 largest arterioles and venules in a ring-shaped area located between 0.5 and 1.0 disc diameter from the optic disc margin are identified. Computer software measures the caliber of these individual vessels, then combines them into 2 summary variables for the eye: the projected caliber size of the central retinal artery (central retinal artery equivalent [CRAE]), and the projected caliber size of the central retinal vein (central retinal vein equivalent [CRVE]), using formulas derived by Parr and Spears and by Hubbard, with revision by Knudtson. These indices are used to calculate the arteriole-to-venule ratio (AVR), as CRAE/CRVE.
We categorized study participants who died during follow-up on the basis of immediate and contributing causes of death, using available death certificates; specifically, we identified whether death was associated with diseases characterized by vasculopathy (renal disease, cardiovascular disease, stroke), liver disease, AIDS-related opportunistic infections or malignancies, other AIDS-related disorders, or trauma, as described in a previous LSOCA publication about this cohort.
Definitions
For purposes of this study, HAART was defined as the concurrent use of 3 or more antiretroviral drugs. Study definitions for the following conditions can be found in a previous publication about this cohort: diabetes mellitus, hypertension, cardiovascular disease, stroke, peripheral vascular disease, and renal disease.
Data Analysis and Statistical Techniques
Unless otherwise noted, the unit of analysis was the eye, and 1 eye per study participant was evaluated. Wong and associates have demonstrated a strong correlation between the eyes of an individual, for both CRAE and CRVE, and concluded that measurements from 1 eye accurately reflect a person’s systemic microvascular status. If vessel caliber indices could be determined for both eyes, the eye with better photographic quality was selected as the study eye. Values for each vessel caliber index were grouped by quartiles and modeled ordinally. With regard to CRAE and CRVE, the first quartile contained narrower arterioles and venules, respectively, while the fourth quartile contained wider arterioles and venules. With regard to AVR, the first quartile included relatively narrower arterioles vs venules, while the fourth quartile included relatively wider arterioles vs venules.
Ordered logistic regression was used to assess the cross-sectional relationship between baseline factors (demographic, medical, laboratory, visual function) and vessel caliber index quartiles as the response variable. A forward selection model with P value entry criterion of .05 was used to create adjusted models, using the following covariates: age, black race, hematocrit, MCV, history of HAART (previous use, use at baseline, or both), time since diagnosis of AIDS, and history of smoking. Factors for which there were statistically significant associations on adjusted models were chosen as covariates in subsequent adjusted models.
Vessel caliber indices were used as predictors in 1) cross-sectional analyses using logistic regression of selected comorbidities at baseline and linear regression of Karnofsky score at baseline; and 2) longitudinal analyses using Cox regression of incident death during follow-up. The Fisher exact test was used to examine the relationship between baseline vessel caliber indices and causes of, or factors contributing to, death.
Because diabetes mellitus can cause vascular disease similar to that seen in people with AIDS, we performed subgroup analyses, looking for significant ( P < .01) interactions between diabetes mellitus and relationships that involve vessel caliber. Similar subgroup analyses were performed to look for interactions with hypertension.
P values were 2-sided and were not adjusted for multiple comparisons. Statistical analyses were performed using SAS (SAS/STAT User’s Guide, Version 9.2, 2010; SAS Institute, Cary, North Carolina, USA) and Stata (Stata Statistical Software: Release 11, 2009; StataCorp LP, College Station, Texas, USA) statistical packages.
Results
As of December 31, 2008, 2221 individuals had enrolled in LSOCA, 1712 of whom had no ocular opportunistic infections. The demographic, medical, laboratory, and ophthalmic examination data for this subpopulation are described in a previous publication. Median age of the cohort was 43 years (range 38–48 years), and 34% of participants self-reported race/ethnicity as being black. Participants were significantly more likely to be excluded if they were older (mean age 44 ± 10 years for those excluded vs 43 ± 8 years for those included, P = .03) or black (34% of black participants were excluded vs 25% of nonblack participants, P = .0003). These differences were attributed to the quality of the fundus photographs; participants who were older or black were more likely to have problems with dilation, resulting in photographs of lower quality that could not be used to determine vascular caliber indices.
Median time since AIDS diagnosis was 4.2 years (range 1.6–7.1 years), and median CD4+ T lymphocyte count at baseline was 192 cells/μL (range 81–350 cells/μL). HAART had been used before or at baseline in 90% (981/1093) of participants; 85% (1056/1249) of participants were using HAART at baseline. Among the 1712 eligible participants, 1250 eyes had vessel caliber measurements at baseline. The Figure illustrates the normal distribution of the vessel caliber indices.
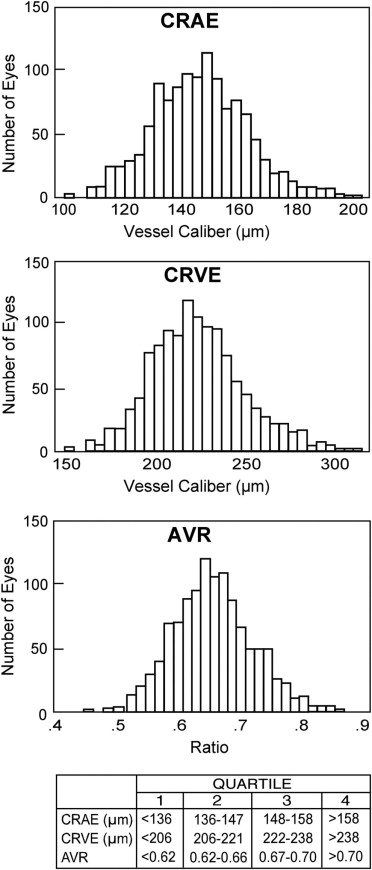
Table 1 shows relationships between vessel caliber indices and demographic, medical, and selected laboratory factors at baseline. Table 2 shows the relationships between vessel caliber indices and AIDS-specific factors. Crude (univariate) analyses demonstrated that at least 1 vessel caliber index was statistically associated with the following factors: age, sex, black race, weight, history of smoking, hemoglobin, hematocrit, MCV, time since AIDS diagnosis, lymphocytopenia, HIV RNA blood level, and history of HAART. On multivariate analyses, at least 1 vessel caliber index remained independently associated with the following factors: age, black race, hematocrit, MCV, history of smoking, time since AIDS diagnosis, and history of HAART. There was also an independent association between increased CRAE and decreased CD4+ T lymphocyte count. There were no significant relationships between any vessel caliber index and cotton-wool spots (all P values ≥ .54, data not shown).
| Quartile | P Value | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | Crude | Adjusted a | |
| Age (median, years) | ||||||
| CRAE | 45 | 44 | 42 | 41 | <.001 | <.001 |
| CRVE | 45 | 43 | 42 | 42 | <.001 | <.001 |
| AVR | 44 | 43 | 43 | 42 | <.001 | <.001 |
| Sex (percent, female) | ||||||
| CRAE | 13 | 17 | 19 | 26 | <.001 | – |
| CRVE | 12 | 19 | 23 | 23 | <.001 | – |
| AVR | 20 | 15 | 18 | 22 | .28 | – |
| Race/ethnicity (percent, black) | ||||||
| CRAE | 24 | 30 | 38 | 44 | <.001 | <.001 |
| CRVE | 21 | 28 | 38 | 49 | <.001 | <.001 |
| AVR | 41 | 35 | 29 | 32 | .008 | .009 |
| Weight (median, kg) | ||||||
| CRAE | 77 | 74 | 75 | 74 | .02 | – |
| CRVE | 75 | 75 | 74 | 74 | .04 | – |
| AVR | 75 | 75 | 75 | 74 | .67 | – |
| Smoking history (percent, ever smoked cigarettes) | ||||||
| CRAE | 58 | 57 | 56 | 69 | .02 | – |
| CRVE | 49 | 59 | 66 | 65 | <.001 | .004 |
| AVR | 62 | 62 | 58 | 57 | .14 | – |
| Hematology (medians) | ||||||
| Hemoglobin (g/dL) | ||||||
| CRAE | 14.1 | 14.0 | 13.6 | 13.2 | <.001 | – |
| CRVE | 13.9 | 13.8 | 13.7 | 13.5 | .002 | – |
| AVR | 14.0 | 13.8 | 13.8 | 13.5 | .03 | – |
| Hematocrit (percentage) | ||||||
| CRAE | 41.4 | 41.2 | 40.0 | 38.8 | <.001 | .002 |
| CRVE | 41.0 | 40.8 | 40.1 | 39.5 | .005 | – |
| AVR | 41.2 | 40.9 | 40.0 | 39.6 | .008 | .001 |
| Mean corpuscular volume (fL) | ||||||
| CRAE | 99.8 | 98.6 | 96.8 | 97.1 | .005 | – |
| CRVE | 101.5 | 98.6 | 96.5 | 96.0 | <.001 | .001 |
| AVR | 97.0 | 98.4 | 97.8 | 100.1 | .009 | .04 |
| Platelet count (× 1000 cells/μL) | ||||||
| CRAE | 214 | 215 | 210 | 214 | .73 | – |
| CRVE | 212 | 213 | 222 | 211 | .34 | – |
| AVR | 220 | 214 | 209 | 215 | .42 | – |
| Leukocyte count (× 1000 cells/μL) | ||||||
| CRAE | 5.0 | 4.7 | 4.6 | 4.4 | .11 | – |
| CRVE | 4.6 | 4.9 | 4.8 | 4.5 | .85 | – |
| AVR | 4.8 | 4.7 | 4.8 | 4.4 | .24 | – |
| Absolute neutrophil count (× 1000 cells/μL) | ||||||
| CRAE | 2.6 | 2.4 | 2.3 | 2.2 | .32 | – |
| CRVE | 2.3 | 2.5 | 2.5 | 2.2 | .87 | – |
| AVR | 2.5 | 2.4 | 2.4 | 2.2 | .83 | – |
a Variables selected from 3 separate ordered logistic regression models, regressing quartiles of CRAE, CRVE, and AVR on a candidate list of all baseline characteristics (n = 1181 complete cases), using forward-selection entry criterion P < .05. Significant variables for regression of CRAE are age, black race, hematocrit, HAART (ever used), and CD4+ T lymphocyte count; for regression of CRVE, they are age, black race, history of smoking, mean corpuscular volume, HAART (ever used and current use); for regression of AVR, they are age, black race, hematocrit, mean corpuscular volume, and current use of HAART.
| Quartile | P Value | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | Crude | Adjusted a | |
| AIDS history | ||||||
| Time since AIDS diagnosis (median, years) | ||||||
| CRAE | 4.5 | 4.4 | 4.4 | 3.4 | .15 | – |
| CRVE | 4.6 | 4.5 | 3.9 | 3.6 | .05 | – |
| AVR | 4.1 | 4.5 | 4.0 | 4.4 | .49 | – |
| Lymphocytopenia as AIDS-defining illness (percentage) | ||||||
| CRAE | 65 | 67 | 68 | 60 | .16 | – |
| CRVE | 68 | 66 | 66 | 60 | .03 | – |
| AVR | 66 | 60 | 65 | 68 | .28 | – |
| Immunology and virology | ||||||
| CD4+ T lymphocyte count (median, cells/μL) | ||||||
| CRAE | 201 | 194 | 200 | 172 | .73 | .04 |
| CRVE | 207 | 197 | 184 | 182 | .13 | – |
| AVR | 191 | 197 | 190 | 186 | .59 | – |
| Nadir CD4+ T lymphocyte count (median, cells/μL) | ||||||
| CRAE | 42 | 38 | 44 | 42 | .50 | – |
| CRVE | 45 | 41 | 41 | 42 | .21 | – |
| AVR | 39 | 41 | 46 | 42 | .33 | – |
| CD8+ T lymphocyte count (median, cells/μL) | ||||||
| CRAE | 784 | 773 | 798 | 747 | .09 | – |
| CRVE | 756 | 753 | 808 | 766 | .78 | – |
| AVR | 786 | 767 | 787 | 751 | .14 | |
| Peak HIV RNA blood level (median, log copies/mL) | ||||||
| CRAE | 5.3 | 5.3 | 5.3 | 5.3 | .50 | – |
| CRVE | 5.3 | 5.3 | 5.3 | 5.4 | .37 | – |
| AVR | 5.3 | 5.4 | 5.2 | 5.3 | .22 | – |
| HIV RNA blood level (median, log copies/mL) | ||||||
| CRAE | 2.6 | 2.7 | 2.6 | 3.2 | <.001 | – |
| CRVE | 2.6 | 2.6 | 2.9 | 3.5 | <.001 | – |
| AVR | 2.9 | 2.6 | 2.8 | 2.7 | .37 | – |
| Antiretroviral treatment history (percent, individuals) | ||||||
| Ever on HAART | ||||||
| CRAE | 93 | 94 | 87 | 85 | <.001 | <.001 |
| CRVE | 94 | 92 | 90 | 83 | <.001 | <.001 |
| AVR | 89 | 91 | 88 | 90 | .82 | – |
| Current HAART | ||||||
| CRAE | 89 | 88 | 82 | 80 | <.001 | – |
| CRVE | 91 | 87 | 83 | 77 | <.001 | .01 |
| AVR | 79 | 85 | 88 | 86 | .01 | .004 |
a Variables selected from 3 separate ordered logistic regression models, regressing quartiles of CRAE, CRVE, and AVR on a candidate list of all baseline characteristics (n = 1181 complete cases), using forward selection entry criterion P < .05. Significant variables for regression of CRAE are age, black race, hematocrit, HAART (ever used), and CD4+ T lymphocyte count; for regression of CRVE, they are age, black race, history of smoking, mean corpuscular volume, HAART (ever used and current use); for regression of AVR, they are age, black race, hematocrit, mean corpuscular volume, and current use of HAART.
Table 3 shows the relationships between vessel caliber indices and the presence at baseline of selected systemic diseases that are characterized by vasculopathy. Only hypertension remained significantly associated with vessel caliber on multivariate analyses; for participants grouped by CRAE and AVR, those in the first quartiles (narrowest arterioles; smallest AVR) had the highest prevalence of hypertension. With regard to Karnofsky scores, we found that lower scores (worse health) were strongly related to larger CRVE ( P = .001) and smaller AVR ( P < .001; data not shown).
| Quartile | Comparisons | |||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | Crude | Adjusted a | |||
| OR b | P | OR b | P | |||||
| Comorbidities | ||||||||
| Hypertension (percent, individuals) | ||||||||
| CRAE | 27 | 21 | 17 | 14 | 0.76 | <.001 | 0.75 | <.001 |
| CRVE | 21 | 22 | 19 | 17 | 0.89 | .09 | 0.88 | .08 |
| AVR | 27 | 21 | 16 | 15 | 0.79 | <.001 | 0.83 | .007 |
| Diabetes mellitus (percent, individuals) | ||||||||
| CRAE | 9 | 8 | 10 | 6 | 0.89 | .23 | 0.92 | .41 |
| CRVE | 7 | 11 | 8 | 7 | 0.91 | .34 | 0.96 | .73 |
| AVR | 11 | 8 | 7 | 8 | 0.93 | .47 | 0.93 | .47 |
| Renal disease (percent, individuals) | ||||||||
| CRAE | 5 | 4 | 6 | 5 | 1.03 | .83 | 1.00 | .97 |
| CRVE | 6 | 4 | 5 | 6 | 1.00 | .94 | 1.00 | .97 |
| AVR | 6 | 3 | 5 | 7 | 1.06 | .64 | 1.03 | .77 |
| Coronary heart disease (percent, individuals) | ||||||||
| CRAE | 8 | 7 | 6 | 8 | 0.98 | .88 | 1.05 | .69 |
| CRVE | 8 | 7 | 7 | 8 | 1.01 | .90 | 1.04 | .77 |
| AVR | 7 | 7 | 8 | 6 | 0.98 | .88 | 1.00 | .97 |
| Peripheral vascular disease (percent, individuals) | ||||||||
| CRAE | 5 | 3 | 5 | 3 | 0.89 | .45 | 0.90 | .52 |
| CRVE | 4 | 2 | 4 | 7 | 1.31 | .07 | 1.26 | .15 |
| AVR | 5 | 5 | 5 | 1 | 0.73 | .04 | 0.79 | .12 |
| Stroke (percent, individuals) | ||||||||
| CRAE | 6 | 6 | 3 | 4 | 0.82 | .15 | 0.85 | .26 |
| CRVE | 7 | 4 | 6 | 4 | 0.86 | .27 | 0.88 | .38 |
| AVR | 3 | 9 | 5 | 3 | 0.95 | .73 | 0.96 | .77 |
a Analyses involving CRAE were adjusted for age, race, hematocrit, time since diagnosis of AIDS, HAART (ever used), and CD4+ T lymphocyte count; analyses involving CRVE were adjusted for age, race, mean corpuscular volume, history of smoking, and HAART (ever used and current use); analyses involving AVR were adjusted for age, race, mean corpuscular volume, hematocrit, and current use of HAART.
b Odds ratio defined as change in odds of event per quartile of vascular measurement.
Table 4 shows the relationship of vessel caliber indices at baseline with death of 304 participants during follow-up. Larger CRVE ( P = .006) and smaller AVR ( P < .001) were strongly related to death on crude analyses; only the relationship with AVR remained significant after adjustment for comorbidities ( P = .02). HIV RNA blood level and HAART use at enrollment were the primary confounders that attenuated the effect of CRVE on mortality. The unadjusted relative risk (RR) for mortality per quartile of CRVE was 1.15 ( P = .006, as noted above); with adjustment, RR was 1.07 ( P = .21). During follow-up, 15 of 1250 participants developed cytomegalovirus (CMV) retinitis. Because AIDS-related CMV retinitis is associated with an increased risk of mortality, we repeated our analyses after excluding these 15 individuals, and the relationship between AVR and death remained significant (data not shown).
| Index | Crude | Adjusted b | ||||
|---|---|---|---|---|---|---|
| RR c Death/Quartile | 95% CI | P | RR c Death/Quartile | 95% CI | P | |
| CRAE | 0.98 | 0.89–1.09 | .77 | 0.95 | 0.86–1.06 | .40 |
| CRVE | 1.15 | 1.04–1.27 | .006 | 1.07 | 0.96–1.20 | .21 |
| AVR | 0.84 | 0.76–0.93 | <.001 | 0.88 | 0.79–0.98 | .02 |
a 304 deaths among 1250 study participants; median follow-up, 6.8 years; rate, 3.6/100 person-years.
b Adjusted for the following variables at baseline: current use of HAART, CD4+ T lymphocyte count, HIV RNA blood level; age, black race, mean corpuscular volume, hematocrit, time since diagnosis of AIDS, and hypertension (n = 1152 because of missing values).
c Relative risk estimated from Cox regression per increasing quartile.
Death certificates were available for 92 of 304 participants (30.3%). Table 5 shows the relationships between vessel caliber indices at baseline and causes of mortality during follow-up. There was a weak relationship between smaller CRAE and diseases characterized by microvasculopathy (cardiovascular disease, renal disease, and stroke) as causes of, or contributors to, death ( P = .08).
| Quartile | ||||
|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |
| Number of deaths a | ||||
| CRAE | 78 | 81 | 66 | 79 |
| CRVE | 64 | 72 | 77 | 91 |
| AVR | 96 | 73 | 76 | 59 |
| Number of participants for whom death certificates were available | ||||
| CRAE | 30 | 22 | 23 | 17 |
| CRVE | 20 | 20 | 24 | 28 |
| AVR | 36 | 28 | 18 | 10 |
| Attributed causes of, or contributors to, death | ||||
| Diseases characterized by vasculopathy b | ||||
| Cardiovascular disease | ||||
| CRAE | 4 | 2 | 0 | 0 |
| CRVE | 2 | 2 | 2 | 0 |
| AVR | 3 | 3 | 0 | 0 |
| Renal disease | ||||
| CRAE | 1 | 0 | 1 | 1 |
| CRVE | 1 | 0 | 0 | 2 |
| AVR | 1 | 0 | 2 | 0 |
| Stroke | ||||
| CRAE | 1 | 1 | 0 | 0 |
| CRVE | 0 | 0 | 2 | 0 |
| AVR | 1 | 1 | 0 | 0 |
| Liver disease | ||||
| CRAE | 4 | 0 | 0 | 3 |
| CRVE | 3 | 0 | 2 | 2 |
| AVR | 4 | 1 | 2 | 0 |
| Other specified diseases c | ||||
| CRAE | 4 | 3 | 2 | 4 |
| CRVE | 3 | 4 | 3 | 3 |
| AVR | 5 | 4 | 1 | 3 |
| AIDS-related opportunistic infection or malignancy | ||||
| CRAE | 9 | 6 | 7 | 4 |
| CRVE | 7 | 5 | 5 | 9 |
| AVR | 9 | 8 | 5 | 4 |
| AIDS-related, not otherwise specified | ||||
| CRAE | 7 | 9 | 11 | 5 |
| CRVE | 4 | 9 | 7 | 12 |
| AVR | 13 | 9 | 7 | 3 |
| Trauma | ||||
| CRAE | 0 | 1 | 2 | 0 |
| CRVE | 0 | 0 | 3 | 0 |
| AVR | 0 | 2 | 1 | 0 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


