Purpose
To evaluate the retinal functional changes measured by scanning laser ophthalmoscope microperimetry in neovascular age-related macular degeneration treated with ranibizumab injections.
Design
Prospective, interventional case series.
Methods
A total of 42 eyes of 39 patients with neovascular age-related macular degeneration were included. After an initial 3 loading injections of ranibizumab, 0.5 mg per injection per month, injection was performed as needed. Evaluation of best-corrected visual acuity, microperimetry, and optical coherence tomography were performed before treatment and 3 months, 6 months, and 12 months after treatment. According to the appearance of the subfoveal choroidal neovascular membrane on fluorescein angiography, the study group was divided into patients with a predominantly or purely classic choroidal neovascular membrane, those with a minimally classic choroidal neovascular membrane, and patients with occult choroidal neovascular membrane.
Results
In all the subjects, mean retinal sensitivity of the central 12-degree area had increased significantly from 4.89 ± 3.1 dB to 9.82 ± 2.1 dB at month 12 ( P = .01). The number of absolute scotoma points decreased significantly from 11.3 ± 3.2 to 5.9 ± 2.4 at month 12 ( P = .01). However, in the subgroup analysis, the mean retinal sensitivity improvement, decreased absolute scotoma size, best-corrected visual acuity improvement, and central macular thickness improvement did not differ significantly among the groups.
Conclusions
Intravitreal 0.5 mg ranibizumab therapy improves retinal function, quantified not only by visual acuity, but also by mean retinal sensitivity and fixation stability, as assessed by scanning laser ophthalmoscope microperimetry. Measurement of retinal sensitivity may facilitate evaluation of the effectiveness of intravitreal ranibizumab treatment in patients with neovascular age-related macular degeneration.
Neovascular age-related macular degeneration (AMD) is the leading cause of irreversible loss of central vision in people of older than 50 years in industrial countries. Loss of visual acuity typically results from choroidal neovascularization (CNV) beneath the macula. Vascular endothelial growth factor (VEGF)-A is a key factor in the pathogenesis of CNV. Ranibizumab (Lucentis; Novartis Pharma AG, Basel, Switzerland, and Genentech, Inc, South San Francisco, California, USA) is a recombinant, fully humanized, affinity-matured monoclonal antigen-binding antibody fragment that inhibits the binding of multiple biologically active forms of VEGF-A to their receptors.
Two pivotal phase III trials, MARINA ( M inimally classic/occult trial of the A nti-VEGF antibody R anibizumab I n the treatment of N eovascular A ge-related macular degeneration) and ANCHOR ( AN ti-VEGF antibody for the treatment of predominantly classic CHOR oidal neovascularization in age-related macular degeneration), have demonstrated the efficacy of monthly dosing regimens of ranibizumab in improving visual acuity in patients with subfoveal CNV secondary to AMD. These studies also described the safety and tolerability profile of intravitreal treatment using ranibizumab.
In most previous studies, visual acuity was the standard way of measuring visual performance, but it inadequately described the functional impact in patients with compromised central visual fields resulting from AMD. Perimetry examines the light differential threshold, which differs from the minimal angle of spatial resolution (also termed visual acuity ). The latter markedly depends on the translucency of the optic media, whereas the light differential threshold mainly depends on the intactness of the photoreceptors. Therefore, the evaluation of retinal sensitivity and central retinal field function by using microperimetry, which is one of the functional evaluation techniques, is more informative compared with only visual acuity testing.
Several studies have shown that microperimetry allows an accurate measurement of retinal sensitivity in neovascular AMD treated with an anti-VEGF agent. However, to the best of our knowledge, most of the previous studies were retrospective, and there have been no reports on differences in the change of retinal sensitivity among neovascular membrane types. In this study, we evaluated the changes in macular sensitivity for patients with neovascular AMD treated with intravitreal ranibizumab therapy in association with subfoveal neovascular membrane type.
Methods
Patient Selection
A prospective, nonrandomized interventional case series was conducted in patients with neovascular AMD referred to the Retina Center of Kim’s Eye Hospital, Konyang University College of Medicine, from November 2008 through January 2011.
Patients were included if they met all of the following criteria: (1) age 50 years or older; (2) naïve to treatment; (3) subfoveal CNV attributable to AMD diagnosed by fluorescein angiography (FA); (4) presence of subretinal fluid, cystic maculopathy, or central retinal thickness of more than 250 μm on optical coherence tomography (OCT); (5) best-corrected vision of more than 20/400 (Snellen equivalent); (6) CNV of less than 5400 μm maximum linear dimension; and (7) ability to understand and sign the consent form.
Exclusion criteria were as follows: (1) retinal angiomatous proliferation or polypoidal choroidal vasculopathy; (2) submacular hemorrhage or scarring involving the fovea; (3) corneal, lenticular, or vitreous opacification that prevented good-quality angiograms or OCT; (4) history of uveitis or vitrectomy; (5) diabetic retinopathy; (6) other ocular conditions that affect vision; or (7) cardiovascular, cerebrovascular, or peripheral vascular events within 6 months of enrollment. Only patients with typical neovascular AMD were included, and those with retinal angiomatous proliferation or polypoidal choroidal vasculopathy were excluded, because we considered that retinal angiomatous proliferation or polypoidal choroidal vasculopathy may respond to treatment differently from typical neovascular AMD lesions.
Approval for this study was obtained from the Institutional Review Board of the Konyang University College of Medicine. The procedure and the treatment options were explained extensively to the patients; those who agreed to participate signed a consent form.
Documentation and Follow-Up
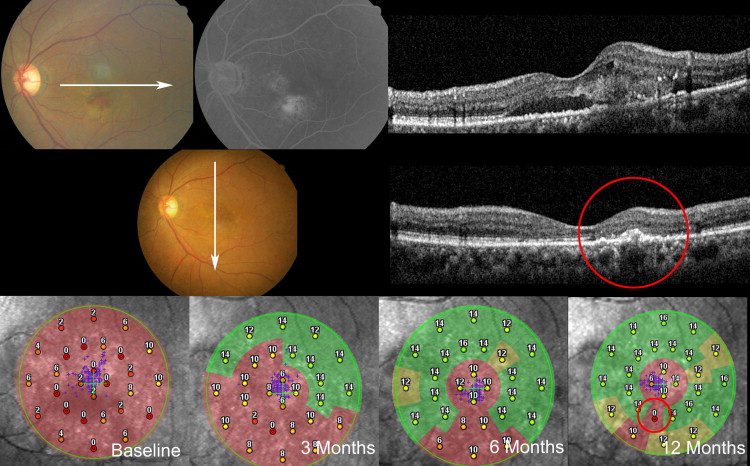
All patients underwent a complete ophthalmic evaluation 1 week before the treatment and at follow-up examinations at 3 months, 6 months, and 12 months after the treatment. Visual acuity measurement using a Snellen chart, slit-lamp examination, tonometry, binocular ophthalmoscopy, color fundus photography, OCT (Spectral OCT/SLO; OTI Ophthalmic Technologies, Inc, Miami, Florida, USA), FA, indocyanine green angiography performed using a confocal laser scanning system (HRA-2; Heidelberg Engineering, Dossenheim, Germany), and microperimetry (spectral-domain OCT/scanning laser ophthalmoscopy with an add-on microperimetry module; OPKO, Miami, Florida, USA) all were performed before intravitreal injection. Further, microperimetry and OCT were performed at each follow-up examination.
Treatment Protocol
Intravitreal ranibizumab (0.5 mg) injection was performed for all patients with the same treatment and retreatment protocols. Evidence of neovascular AMD with recent visual deterioration was an indication for treatment. We performed 3 consecutive monthly loading dose injections. After performing these 3 loading injections, retreatment for each patient was performed on a “retreated as needed” basis if any of the following were observed: (1) visual deterioration of more than 2 lines; (2) evidence of persistent fluid or hemorrhage that involved the macula on OCT at least 1 month after the previous injection; (3) newly developed macular hemorrhage; or (4) evidence of an active neovascular AMD lesion on FA, indocyanine green angiography, or OCT.
Optical Coherence Tomography Examination
OCT imaging was performed with the spectral-domain OCT module integrated in the OPKO/OTI device. It captured 28 000 A-scans per second, which enable the acquisition of up to 128 longitudinal OCT scans in 2 seconds over a macular area of 5 mm. Central macular thickness (CMT) was assessed by retinal thickness of the 1-mm central retina obtained by the macular scan. Only well-centered scans, without overt algorithm failure messages, were selected for analysis.
Fluorescein Angiography and Subgrouping of the Study Group
At baseline, according to the appearance of the subfoveal choroidal neovascular membrane on FA, the study group was divided into the following: patients with a predominantly or purely classic type of subfoveal neovascular membrane (with a more than 50% classic component); those with a minimally classic type of subfoveal neovascular membrane (with a less than 50% classic component); and patients with occult-type subfoveal neovascular membrane. The fluorescein angiograms were examined in a blinded manner by the main investigators (H.J.C. and S.J.Y.) and, in case of doubt, additionally by the senior authors (C.G.K. and J.W.K.).
Microperimetry
Microperimetry was performed by an automatic fundus-related perimeter, which included the spectral-domain OCT (OTI Spectral OCT/SLO; OTI Ophthalmic Technologies, Inc). The standard Goldmann III size was used as a stimulus, and the duration of the stimulus was 200 milliseconds. The fixation target was a red cross of 2 degrees and varied in size according to the patient’s visual acuity (range, 2 to 10 degrees). A white monochromatic background at 1.27 cd/m 2 was used, and a 4-2-1 double-staircase strategy was applied. Before testing the eye, each subject was given pretest training and a demonstration of the test environment, which included actual presentations of the stimulus in the area to be tested. This was carried out to eliminate or minimize any learning effect that might have affected the results. During the examination, fixation behavior and response reliability of the participants were monitored, and only tests with fewer than 10% false-positive or false-negative answers were included in the study.
An automatic protocol was used with a customized radial grid to test the 28 locations that cover the central 12 degrees (28 dots; 4 central, 12 middle, and 12 outer rings). An autonomic follow-up protocol was performed at each control examination that selects the same retinal landmark as in the baseline evaluation. Macular sensitivity was defined as the mean retinal sensitivity of the 28 locations that cover the central 12-degree field.
The size of the dense relative scotoma was evaluated by the number of measurement points with a sensitivity of less than 5 dB, whereas the size of the absolute scotoma was evaluated by the number of points with sensitivity of 0 dB. The number of points of dense relative and absolute scotoma was measured before and after the treatment.
The stability of fixation, which was graded on the basis of the variation of the preferred retinal locus, was assessed according to the classification of Fujii and associates as follows. If less than 75% of fixation points were located within a predetermined limited area of the 4-degree diameter circle centered in the gravitational center of all fixation points, regardless of the position of the foveal center, fixation was classified as unstable. If less than 75% of fixation points were located within a 2-degree circle, but more than 75% of fixation points were located within a 4-degree circle, fixation was classified as relatively unstable. Furthermore, if more than 75% of fixation points was located within a 2-degree circle, the fixation was classified as stable. For statistical analysis, fixation stability was graded as follows: stable = 3, relatively stable = 2, and unstable = 1.
Statistical Analysis
Data are expressed as the mean ± standard deviation and 95% confidence intervals (CIs) for continuous variables and as frequencies for categorical variables. Statistical analysis was performed using a commercially available statistical software package (SPSS for Windows, version 13.0; SPSS, Inc, Chicago, Illinois, USA). Frequencies were compared between the treatment groups using chi-square tests. When appropriate, a 1-way analysis of variance was used. The changes in retinal sensitivity, BCVA, and CMT between the baseline and the follow-up were analyzed with a 1-tailed paired t test. A P value of less than .05 was considered significant.
Results
Baseline Characteristics
Forty-two eyes of 39 patients (19 men and 20 women) were enrolled in this study. The average age for the entire study group was 67.62 ± 10.42 years. At the baseline, the mean logarithm of the minimal angle of resolution BCVA of the entire study group was 0.86 ± 0.58, and the mean CMT was 297 ± 120.4 μm. Subjects received a mean of 3.9 ± 1.2 injections within the 12-month period ( Table 1 ).
| Total (n = 42) | Purely or Predominantly Classic Type (n = 10) | Minimally Classic Type (n = 13) | Occult Type (n = 19) | P Value | |
|---|---|---|---|---|---|
| Age (mean ± SD), year | 67.62 ± 10.42 | 67.22 ± 11.48 | 66.25 ± 9.54 | 68.23 ± 7.65 | .31 a |
| Gender | |||||
| Male | 20 (47.6%) | 4 (69.1%) | 8 (61.5%) | 8 (42.1%) | |
| Female | 22 (52.4%) | 6 (30.9%) | 5 (38.5%) | 11 (57.9%) | .24 b |
| BCVA (Snellen equivalent) | |||||
| ≤ 20/200 | 8 (19.0%) | 1 (10.0%) | 2 (15.4%) | 5 (16.7%) | |
| > 20/200 to ≤ 20/40 | 27 (64.3%) | 7 (70.0%) | 8 (61.5%) | 12 (63.2%) | |
| > 20/40 | 7 (26.7%) | 2 (20.0%) | 3 (23.1%) | 2 (20.1%) | .58 b |
| Total CNV size (DA) | |||||
| Mean ± SD | 4.1 ± 1.8 | 3.8 ± 2.1 | 4.2 ± 2.2 | 4.1 ± 2.4 | .64 a |
| Median (range) | 3.6 (0.1-11.8) | 3.3 (0.0-9.4) | 3.7 (0.2-10.2) | 3.5 (0.1-8.5) | .74 a |
| Mean baseline CMT ± SD (μm) | 297 ± 120.4 | 317 ± 106.9 | 302 ± 150.2 | 284 ± 134.6 | .29 a |
| Mean no. of injections ± SD | 3.9 ± 1.2 | 3.8 ± 1.7 | 4.0 ± 1.8 | 3.9 ± 1.5 | .84 a |
According to the appearance of the subfoveal choroidal neovascular membrane on FA, the group of predominantly or purely classic type consisted of 10 eyes, the group of minimally classic type comprised 13 eyes, and the group of occult type included 19 eyes. All 3 subgroups had similar baseline characteristics for age, sex, distribution of the baseline BCVA, total CNV size, CMT, and mean number of injections with no significant differences ( Table 1 ).
All patients were Korean, and no systemic adverse events were recorded for any of the patients treated with intravitreal injection. No complications, including endophthalmitis, traumatic lens injury, or retinal detachment, were associated with the intravitreal injections.
Functional Changes of the Macula With Microperimetry
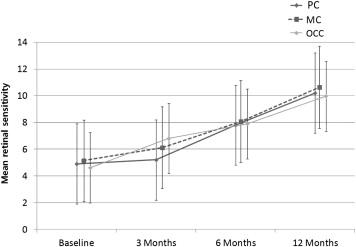
There was significant improvement of mean retinal sensitivity of all the subjects by microperimetry at each examination (4.89 ± 3.1 dB at baseline, 6.52 ± 2.1 dB at 3 months, 8.78 ± 3.1 dB at 6 months, and 9.82 ± 2.1 dB at 12 months; Table 2 ). In each group, mean retinal sensitivity increased significantly during the follow-up ( Table 2 ; Figures 1 and 2 ). The mean changes in retinal sensitivity compared with the baseline were the largest in the occult CNV group: 2.20 at 3 months, 4.68 at 6 months, and 5.46 at 12 months. However, there was no statistical difference of mean retinal sensitivity achieved among the groups ( Table 3 ).
| Mean Retinal Sensitivity ± SD | ||||
|---|---|---|---|---|
| Baseline | 3 Months | 6 Months | 12 Months | |
| Total (n = 42) | 4.89 ± 3.1 | 6.52 ± 2.1 | 8.78 ± 3.1 | 9.82 ± 2.1 |
| P value a | .03 | .01 | .01 | |
| Purely or predominantly classic type (n = 10) | 4.24 ± 2.5 | 5.01 ± 2.5 | 7.42 ± 2.8 | 8.23 ± 1.9 |
| P value a | .16 | .04 | .01 | |
| Minimally classic type (n = 13) | 4.90 ± 2.8 | 6.21 ± 3.2 | 8.12 ± 2.4 | 10.2 ± 2.2 |
| P value a | .03 | .01 | .01 | |
| Occult type (n = 19) | 4.42 ± 2.5 | 6.62 ± 3.1 | 9.12 ± 2.7 | 9.88 ± 2.9 |
| P value a | .02 | .01 | .01 | |
| Mean Change (95% Confidential Interval) | |||
|---|---|---|---|
| Baseline to 3 Months | Baseline to 6 Months | Baseline to 12 Months | |
| Total (n = 42) | 1.63 (1.13 to 2.13) | 3.89 (2.88 to 4.90) | 4.93 (2.91 to 6.94) |
| Purely or predominantly classic type (n = 10) | 0.77 (0.27 to 1.27) | 3.18 (1.68 to 4.68) | 3.99 (2.09 to 5.89) |
| Minimally classic type (n = 13) | 1.31 (0.71 to 1.91) | 3.22 (2.11 to 4.33) | 5.30 (3.19 to 7.41) |
| Occult type (n = 19) | 2.20 (1.42 to 2.98) | 4.68 (3.45 to 5.91) | 5.46 (3.34 to 7.58) |
| P value a | .18 | .24 | .47 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


