access to the subretinal space include the amount of traction being exerted on the edges of the break, the effects of gravity (location of the break above or below the horizontal midline), and the lifestyle and activities of the affected person.
Symptoms of an acute retinal tear include a sudden onset of floaters and debris, photopsia, and blurred vision. These same symptoms can occur in uncomplicated PVD. Photopsia associated with vitreoretinal traction is typically of very short duration, unlike the scintillations encountered with migraine or other vascular disturbances.
When an RD has occurred, patients may additionally become aware of visual field loss, with the perception of a mobile shadow or curtain obscuring their vision. If the macula has become involved, there will usually be significant loss of central visual acuity as well. Patients with chronic, slowly progressive, or demarcated detachments, usually in the inferior retina, may be asymptomatic.
As noted above, PVD is usually a precursor of retinal tear and detachment and is evident clinically in most cases. Slit-lamp exam may reveal the presence of fine pigment granules in the anterior vitreous, which, in the absence of a history of prior intraocular surgery, is highly suggestive of a retinal break. When a vitreous hemorrhage occurs in association with PVD, a retinal tear will be present about 70% of the time.
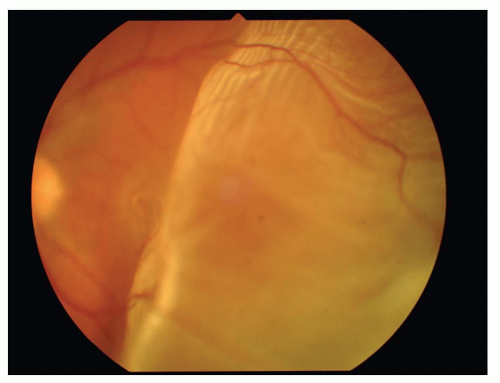
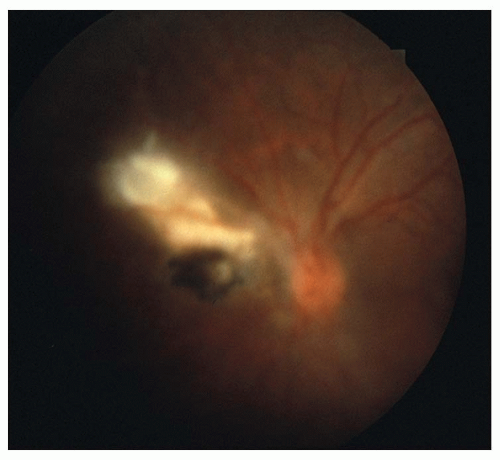
Binocular indirect ophthalmoscopy with scleral depression or peripheral contact lens examination will usually reveal the attached flap of a retinal horseshoe tear under persistent vitreoretinal traction, or a free-floating operculum overlying a retinal hole if the traction has been alleviated. The tear itself usually has a darker orange color than the surrounding retina due to the unimpeded illumination of the underlying choroid.
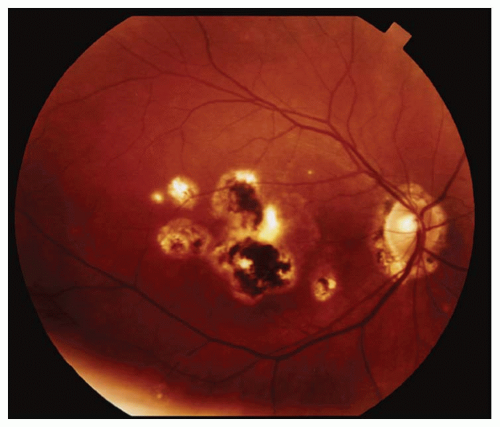
The most obvious visible evidence of the presence of a detached retina is its elevation from the surface. The surface of the detached retina has a corrugated and often dimpled appearance and demonstrates movement with motion of the head and/or eye. This mobility
may be significantly reduced in longstanding RDs or in those complicated by proliferative vitreoretinopathy (PVR).
The configuration of the detachment is an important clue to help pinpoint the location of the retinal break(s). With a superior detachment, the retinal break is usually at or near the 12 o’clock position. When a detachment of one inferior quadrant has occurred, a break will almost always be found superior to a line radially bisecting the area of detachment. In an inferior detachment, if the subretinal fluid extends higher on one side than the other, the break will be found on the higher side. About half of all RDs are associated with a single break, but the possibility of multiple breaks must always be kept in mind.
Confrontation testing will often demonstrate a visual field defect, and Snellen acuity will be reduced if the macula is detached. Tonometry will usually reveal a relatively low intraocular pressure (IOP) in an eye with a RD, although rarely the pressure may actually be higher than normal. Pupillary testing will often demonstrate a relative afferent pupillary defect (RAPD), although this is typically much less prominent than that encountered with optic nerve disease or with central retinal vascular occlusion. Ultrasonography and occasionally electroretinography (ERG) may be helpful in cases where media opacity prevents adequate visualization of the retina.
Traction detachment is associated with a concave retinal configuration and lack of significant movement. Choroidal tumors and detachment can be distinguished from RD by their smooth contour and the presence of retinal pigment epithelium (RPE) and choroidal markings associated with the elevated retina.
Treatment for an acute, symptomatic flap tear is justified as a means of reducing the risk of progression to RD. Studies have shown such tears to progress to detachment in approximately one third of cases. Treatment for symptomatic operculated holes is also justifiable, although the danger of progression is less than for a flap tear with persistent vitreoretinal traction.
In asymptomatic patients, the decision whether or not to treat must be individualized, taking into account multiple factors. These include the location, type, and size of the break(s), with superior breaks, flap tears, and large breaks being considered for prophylaxis. If there is pigmentation around the break, it is not acute and treatment may not be indicated. If there is significant localized subretinal fluid around the break, treatment may be indicated. The presence of vitreous hemorrhage increases the risk associated with a retinal break.
Other factors that must be weighed in the decision are the age and life expectancy of the patient, the patient’s lifestyle, the status of the fellow eye, the refractive error (in particular, the degree of axial myopia), the presence of aphakia or pseudophakia, anticipated cataract surgery, and the family history. The timing of treatment is dictated by the apparent danger associated with a particular break.
Prophylactic treatment of retinal breaks is usually accomplished using either transconjunctival cryopexy or laser photocoagulation. The choice of technique is guided by the number, size, and position of the break(s), as well as by the preference of the patient and surgeon.
There is some theoretical advantage of laser over cryotherapy due to the possibility of dispersing viable RPE cells with cryotherapy. It is felt by many surgeons that there is a greater risk for the subsequent development of an epiretinal membrane (ERM) and associated macular pucker associated with cryotherapy. There has thus been a trend toward increased utilization of laser for prophylactic treatment. However, there are some cases where cryotherapy still holds a distinct advantage, such as cases of very anterior breaks and when visualization is impaired due to the presence of significant cataract or vitreous hemorrhage.
Other than in cases of giant tear, scleral buckling (SB) is rarely utilized for prophylactic treatment.
Laser photocoagulation of retinal tears can usually be accomplished with only topical anesthesia, unless extensive treatment of multiple breaks or lattice degeneration is contemplated. Transconjunctival cryopexy is generally performed utilizing topical anesthesia augmented with a subconjunctival injection in the area overlying the break(s).
Whichever modality is chosen, the goals of treatment are the same: to insure an adequate width of treatment that completely surrounds the retinal break. The resultant scar will then isolate the break and prevent fluid from the vitreous cavity from gaining access to the subretinal space.
Following surgery, the patient is instructed to restrict activity until
an effective adhesion has developed, usually 7 to 10 days for cryopexy or a slightly shorter period of time for laser.
Prophylactic treatment of high-risk retinal breaks has been shown to be effective in significantly reducing the risk of subsequent RD. Serious complications are rare, with the most frequent visually significant adverse consequence being macular pucker. However, macular pucker may also develop in association with retinal breaks that have not been treated.
TABLE 18-1 Selected Results from the 2006 ASRS PAT Survey | |||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||
The decision to treat an RD must also be individualized, taking into account the likelihood of progression, the residual visual potential, the age, lifestyle, and life expectancy of the patient, the status of the fellow eye, and the patient’s wishes.
When an acute, macula-threatening, or recent macula-off detachment is encountered, surgery is indicated as soon as possible. Appropriate bedrest and positioning instructions are given to reduce the possibility of extension of the detachment into the macula, if it is still attached. This may also allow for significant absorption of subretinal fluid after RD surgery.
An inferior demarcated detachment may not require surgery. Likewise, the limited visual prognosis associated with a long-standing macula-off detachment may persuade the patient and surgeon not to pursue surgical intervention, particularly if there is excellent vision in the contralateral eye.
Available methods of retinal reattachment surgery include SB, pars plana vitrectomy (PPV), pneumatic retinopexy (PR), and a temporary balloon buckle. The American Society of Retina Specialists (ASRS) polled its membership about preferences for method of surgical repair of RD in the 2006 PAT (Preferences and Trends [PAT]) survey. Several typical scenarios were described, with differing patient characteristics that often influence the choice of surgical technique. The four scenarios and survey results are shown in Table 18-1.
Traditionally, the surgical technique most commonly employed in the
treatment of rhegmatogenous RD has been SB, in combination with retinopexy utilizing cryotherapy or laser photocoagulation.
The purpose of the buckle is to aid in bringing the treated breaks back into contact with the RPE, and to relieve persistent vitreoretinal traction. The scleral buckle is created utilizing either solid silicone rubber elements or soft silicone sponges, which are permanently anchored to the sclera with sutures.
Placement of the buckle can be segmental, with either radial or circumferential elements, or encircling, passing under the four rectus muscles. In some cases, a combination of an encircling band or tire is combined with a radial sponge.
Cryotherapy or laser is necessary in order to produce a controlled inflammatory reaction that will result in a permanent adhesion of the retina once it is brought back into contact with the RPE. Laser cannot be effectively employed until after the retina has been brought back into close proximity with the pigment epithelium, whereas cryotherapy can be applied around the retinal breaks in the presence of subretinal fluid.
Drainage of subretinal fluid is often required in order to bring the retina into contact with the buckle and also to provide space to achieve an adequate buckle height without an excessive elevation of IOP. The need for drainage is influenced by several factors, including the amount of retinal elevation, the number, size, and position of retinal breaks, and the duration of the detachment, as long-standing subretinal fluid becomes highly viscous and resistant to reabsorption.
A successful outcome of surgery is dependent on finding and closing all the retinal breaks, not necessarily only the causative break.
Surgery is usually performed under regional block anesthesia, although general anesthesia may be necessary in young patients or if a lengthy procedure is a possibility. Some surgeons prefer general anesthesia for most cases.
PPV has become the method of choice for treating RDs associated with giant retinal tears and for dealing with other complicated scenarios. Some surgeons now prefer PPV as their first option in the primary repair of most RDs. Vitrectomy is also necessary to repair recurrent RDs associated with significant PVR.
The vitrectomy is often supplemented with adjunctive techniques, including the use of membrane dissection, internal drainage of subretinal fluid, laser, fluid-gas exchange, intraocular silicone oil, and, particularly in the treatment of giant tears, liquid perfluorocarbon. In many cases, it is also necessary to employ a scleral buckle.
PPV has become increasingly popular, particularly in Europe, as a primary procedure for rhegmatogenous RD. In the United States, many surgeons employ primary PPV for pseudophakic detachments, where its tendency for cataractogenesis is a moot point. There has been a significant recent trend in vitreous surgery to employ smaller gauge instrumentation, with 23-gauge and 25-gauge systems increasingly utilized.
Reported advantages of PPV over SB include better intraoperative control, the avoidance of a very soft eye during surgery, the avoidance of drainage complications, and a lower incidence of refractive change and diplopia. Disadvantages include the invasive nature of the surgery and the associated risk of endophthalmitis, cataractogenesis in phakic patients, the higher rate of iatrogenic and missed/new breaks,
more frequent persistent postoperative IOP elevation, the more technologically intense nature of the procedure requiring more skilled assistance and nursing, and probably its increased expense.
PR is an effective method of repair, if cases are selected carefully. It is more successful when limited to detachments with superior breaks and to cases with a single break or breaks which are minimally separated.
The technique consists of a pars plana injection of perfluorocarbon gas, again combined with cryopexy and/or laser photocoagulation. The patient must be willing and able to sustain a strict postoperative positioning regimen, or the chances of success are dramatically reduced. The head is positioned so as to bring the position of the break to the 12:00, so that the gas bubble completely covers the break. This must be continued until the break has been brought back into contact with the RPE, and a good cryopexy or laser scar is developing.
In general, better results have been obtained in phakic patients. PR is contraindicated in the presence of extensive lattice degeneration or significant PVR.
Gas injection may also be utilized in conjunction with SB, in order to facilitate the reabsorption of subretinal fluid.
The placement of a temporary balloon buckle is also an effective method of repairing selected RDs, although its use never became widespread in the United States. Similar to PR case selection, the temporary balloon buckle is almost always limited to detachments with a single break. Unlike PR, the balloon technique can be employed to treat an inferior break. Currently, the material is not commercially available.
Approximately 80% to 85% of primary RDs can be successfully repaired utilizing a single SB operation, and approximately 92% of detachments overall can be successfully repaired, including those that require two or more procedures.
The success rates for PPV are similar to those of SB in uncomplicated cases, and possibly superior in some hands. The success rates for PPV in complicated cases of RD are significantly higher than with SB.
The one-operation success rates with PR are somewhat less than with either SB or PPV, but failed attempts at this procedure do not seem to significantly jeopardize the ultimate final outcome. An important advantage of PR is its significantly lower overall costs relative to SB and PPV, which must be performed in the operating room. There may also be faster rehabilitation with PR, at least in some cases. When careful case selection is adhered to, the use of this modality can be advocated.
Visual results following RD surgery are in large part determined by the preoperative visual acuity and the status of the macula. Overall, 55% of anatomic surgical successes will achieve at least 20/50 vision within 6 months of surgery, but about 15% will be left with 20/400 or worse. The average long-term visual result for a repaired macula-off detachment is about 20/70 acuity. A patient with a macula-on detachment should be advised preoperatively that the cost of repairing the detachment may be a mild decrease in visual acuity, even with successful surgery.
Patients with a retinal tear and/or a RD require careful follow-up to watch for the development of complications and to monitor the status of the fellow eye.
The most common cause of failed surgery is the development of PVR. PVR is characterized by the development of fibrous cellular proliferation that leads to the formation of epiretinal and/or subretinal membranes that distort the retinal surface into fixed folds and often reopen existing breaks or cause new ones to form. Its development portends a relatively poor prognosis, particularly in regards to the visual outcome.
Other significant complications include intraoperative subretinal hemorrhage, retinal incarceration, and new retinal break formation, and postoperative choroidal detachment, diplopia, strabismus, cystoid macular edema (CME), and macular pucker.
is much less common. It is not certain why in some cases cystoid degeneration progresses to degenerative retinoschisis. There does not appear to be a genetic predisposition to this process. Bullous retinoschisis seems to be more common when zones of typical and reticular cystoid degeneration overlap.
The vast majority of patients (˜99%) with retinoschisis are asymptomatic. Although retinoschisis causes an absolute visual field defect, most patients are unaware of these, except in those rare cases where the schisis extends posteriorly into the macular region. The reasons for the usual lack of recognition of the field cut include the typically very slow or lack of progression of the retinoschisis and the fact that nasal field loss associated with the usual temporally located schisis is often overlooked.
Complications of retinoschisis include RD and vitreous hemorrhage. The localized, nonprogressive type of RD does not usually cause symptoms, but progressive, clinically significant detachments obviously do. Vitreous hemorrhage, causing an acute onset of floaters, or hemorrhage into the schisis cavity may rarely be associated with retinoschisis, but such hemorrhages are usually minor. Incidental PVD is a more common source of both vitreous hemorrhage and floaters in general.
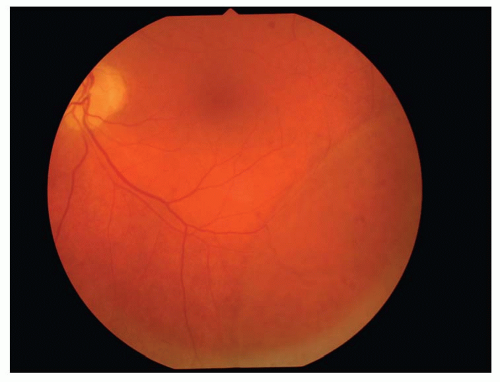
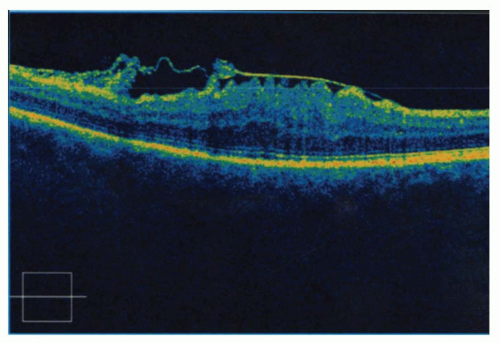
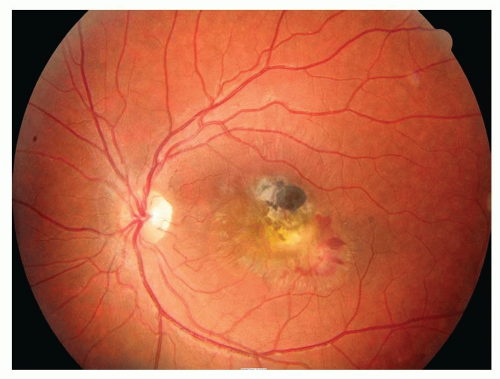
The inner layer of the retinoschisis is visible as a smooth-domed elevation containing the retinal vessels. The retinal vessels may appear white due to sheathing. Retinoschisis is found predominantly in the temporal retina and is usually bilateral, although it may be asymmetric in extent and elevation. The schisis cavity may increase or decrease in height over time, and complete disappearance has been noted in approximately 2% of cases. Tiny glistening flecks are often seen on the inner layer, although these have little clinical significance. New areas of schisis may develop elsewhere in about 10% of cases. Posterior extension of the retinoschisis may occur, although involvement of the macula is rare. Most cases stop within 3 disc diameters of the macula.
Holes may develop in either or both of the walls of a retinoschisis cavity in about 15% of cases, with outer layer holes much more common than inner layer holes. Clinically, what appears to be an inner layer hole may actually represent only an area of extreme thinning. When an inner layer hole does develop, it is usually quite small and difficult to appreciate clinically. Outer layer holes are usually much more obvious, as they tend to be large. They have a rounded contour, may be single or multiple, and are seen typically in association with reticular retinoschisis. Outer layer holes often have a posteriorly rolled edge. Fundus contact lens examination is useful in the recognition of outer and inner layer holes.
Although progressive rhegmatogenous RD is the most important clinical complication of degenerative retinoschisis, this is actually rare, accounting for probably no more than 2.5% of all detachments. In about 6% of cases of retinoschisis, localized, asymptomatic, nonprogressive, or very slowly progressive RDs develop, and these do not extend much beyond the border of the area of schisis. There may be a fine demarcation line present if one of these localized subclinical RDs is present. All schisis-related RDs are associated with outer layer holes. The viscosity of the fluid within the retinoschisis cavity is probably an important factor in inhibiting the progression of schisis-related detachments with outer layer holes. Inner layer holes or traction-related tears are also sometimes present, particularly with progressive, symptomatic RDs.
The differential diagnosis of degenerative retinoschisis includes RD, choroidal detachment/tumors, and juvenile (X-linked) retinoschisis. The latter two groups are usually easily excluded with a thorough history and careful examination. In some cases, the differentiation of retinoschisis from a chronic RD can be challenging. Table 18-2 illustrates some distinguishing characteristics, which are helpful in this distinction.
TABLE 18-2 Clinical Signs: Retinoschisis Versus Retinal Detachment | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Floaters
Transient photopsia
Blurred vision
Metamorphopsia
Prepapillary floater in the shape of an annular ring, line, or other geometric form
Occasionally, preretinal and vitreous hemorrhage, retinal hemorrhage, retinal tear, and RD subtle signs
Macular edema
Macular hole
Pseudo-operculum anterior to macula
ERM
Retinal striations
White without pressure
Schafer’s sign (tobacco dust)-pigmented particles (red blood cells or retinal pigment) in anterior vitreous
Peripheral retinal operculum (if associated with a retinal break)
PVD rarely occurs in patients younger than 45 years of age unless associated with chorioretinitis or trauma.
Vitreous detachment of the optic disc is present in approximately one-half of all patients older than 50 years of age.
By age 65, approximately 31% of the population has experienced a complete PVD.
Complete PVD is much more common than incomplete PVD.
Up to 50% of PVDs are associated with photopsia.
Approximately 90% of patients who report concurrent flashes and floaters have PVD, and 11% have retinal breaks.
Retinal breaks occur in up to 15% of patients with symptomatic PVD.
7.5% of PVDs have an associated vitreous hemorrhage.
Traction at the posterior pole resulting in macular edema occurs in about 2.5% of symptomatic PVDs.
Trauma
Chorioretinitis
May potentially be precipitated by aphakia, pseudophakia, myopia, heavy lifting, and vigorous exercise or isolated head movement
adherent (blood vessels and optic disc), stimulating a reparative process in which Muller cell processes and astroglial cells migrate through ILM breaks to proliferate on the retinal surface (within the subhyaloid space). Other IERMs that are composed of collagen (“cortical vitreous preretinal membranes”) or a combination of collagen and retinal glial cells may also occur.
Decreased visual acuity (dependent upon the extent and location of membrane formation and presence of secondary sequelae)
Amsler grid metamorphopsia
Loss of foveal reflex
Semitranslucent to opaque gatheredpleat or volcano pattern appearance of IERM with deviation of retinal vasculature and retinal folds
Cellophane reflex of retinal surface (best observed with the binocular indirect ophthalmoscope, accessory biomicroscopic lenses, and/or red-free filter of the direct ophthalmoscope)
Perimacular vasculature may be slightly tortuous, and vessels may appear to straighten toward the area of the IERM
IERM is much more prevalent in patients over the age of 50 (6.4% of individuals within this age group are thought to have the condition).
Unilateral IERM occurs in 70% to 90% of cases.
Approximately 1% to 2% of unilateral cases eventually become bilateral.
IERM affects both sexes equally.
PVD occurs in 80% to 90% of cases.
CME has been reported to occur in 16% of patients with IERM.
In 77% of cases, vision is 20/60 or better, while 61% have acuity of 20/40 or better.
Usually none (possibly PVD)
droppings from chickens, other birds, and bats) are inhaled into the pulmonary system and then travel via the bloodstream to the eye (choroid) and/or other organ systems.
Classic triad of peripapillary atrophy, peripheral chorioretinal atrophic spots, and macular subretinal neovascularization
Absence of anterior uveitis
Disciform macular scar
Metamorphopsia or relative scotoma on Amsler grid
Hemorrhagic/serous retinal and RPE detachment
Optic disc edema, choroidal hemorrhage
Vitreous hemorrhage (infrequent)
Absence of vitritis (vitritis concurrent with OHS-like signs may indicate an inflammatory disorder, such as pseudo-POHS, a subtype of acute zonal occult outer retinopathy)
Gray-green area in macular or peripapillary area indicating the presence of an SRNVM
Linear streak lesions at the equator, which run parallel to the oral serrata (infrequent)
OHS patients typically range in age from 20 to 50 years.
OHS occurs six times more frequently in whites than in blacks.
OHS occurs with equal frequency between the sexes.
Bilateral macular involvement is more common in men than in women.
Maculopathy presents in individuals between the ages of 20 to 40 years.
Each year in the United States, at least 2,000 individuals experience significant visual loss because of OHS.
most common manifestation resulting in irreversible damage to the retina in utero. Toxoplasma organisms invade the retina of the fetus, where they may change into the cystic form. Most serious, active cases are thought to represent a late manifestation of an intrauterine infection, where the cysts rupture and liberate parasites in surrounding cells.
of acquired infections is probably much greater than originally suspected. There is clear evidence that acquired toxoplasmosis can induce ocular lesions; however, many of these acquired cases produce self-limited and benign lesions.
Is the patient aware of having contracted any congenital infections?
Does the patient eat raw or undercooked lamb, pork, or chicken?
Is there an exposure to cats or cat litter?
Infected water, organ transplantation, and blood transfusions are potential sources of transmission, but are rare in the Unites States
Inquire about immunosuppression or risk factors for AIDS in patients with active bilateral, atypical, or multifocal lesions.
Serology is supportive but not diagnostic in adults, as 20% to 70% of the general population will show positive titers.
The most commonly used serologic tests are the IFA and ELISA for toxoplasmosis which can be done on blood, cerebrospinal fluid (CSF), and aqueous. These tests permit separation of IgM and IgG antibodies. The presence of IgM antitoxin titers indicates a recent acquisition of infection. Recurrent retinochoroiditis is usually associated with stable, low IgG titers and no IgM antibody. A positive test, even at a 1:1 dilution, corroborates the diagnosis.
In difficult or atypical cases, anterior chamber paracentesis to detect local antibody production may be helpful. If IgG antibodies in the aqueous are higher than in the serum, the diagnosis is supported. In addition, polymerase chain reaction tests to detect T. gondii DNA in the aqueous can help confirm the diagnosis and help differentiate ocular toxoplasmosis from similar retinal lesions caused by herpes virus or other infectious agents.
The Sabin-Feldman dye test, which was the original standard test for toxoplasmosis, requires working with live Toxoplasma organisms and is seldom used today.
Fluorescein angiography and optical coherence tomography may be useful in helping to identify subretinal neovascular membranes and/or macular edema.
lesion may persist for up to 4 months, but it will spontaneously resolve.
Prednisone 40 to 80 mg p.o. q.d. began after the second or third day of antitoxoplasmic therapy until day 10 and then tapered gradually over a period of 4 to 5 weeks. Stop steroids before discontinuing antitoxoplasma medications.
Pyrimethamine—loading dose of 100 to 200 mg p.o. the first day and then 25 mg p.o. b.i.d. for 3 to 6 weeks. Follow complete blood counts and give folinic acid (leucovorin) 5 mg p.o. three to four times weekly to minimize bone marrow depression (thrombocytopenia, leukopenia, megablastic anemia). Continue leucovorin for 1 week after stopping pyrimethamine.
Sulfadiazine—loading dose of 2 to 4 g p.o. the first day, then 1 g p.o. q.i.d. for 3 to 6 weeks depending on clinical response.
Clindamycin—150 to 300 mg p.o. q.i.d. Useful adjunct to above therapy or may be used with only sulfadiazine. If the patient has a sulfa allergy, clindamycin may be used with pyrimethamine. Clindamycin concentrates in the choriod but is expensive. Monitor for pseudomembranous colitis. Limit treatment to 4 weeks.
Azithromycin—500 mg on day 1 followed by 250 mg/d, alone or in combination with pyrimethamine, appears to be effective in treating sight-threatening lesions with fewer side effects than other antibiotics.
Tetracycline—loading dose of 1 g p.o. on the first day, then 500 mg p.o. q.i.d. for 3 to 6 weeks.
Trimethoprim (160 mg)/sulfamethoxazole (800 mg)—1 tablet p.o. b.i.d., with or without clindamycin. Many uveitis specialists concur that TMP/SMX is an effective therapy and is generally well tolerated. There is also some evidence that long-term treatment (every 3 days for up to 20 months) with TMP/SMX significantly reduces the rate of recurrence of retinochoroiditis in patients with a history of multiple previous recurrences.
treatment with antitoxoplasmic agents prior to and after surgery might be prudent for patients at risk for visual loss. Consider TMP/SMX p.o. b.i.d.
Peripheral chorioretinal granuloma (˜50% of patients)—usually associated with dense fibrous bands in the vitreous with dragging of the macula; may mimic pars planitis.
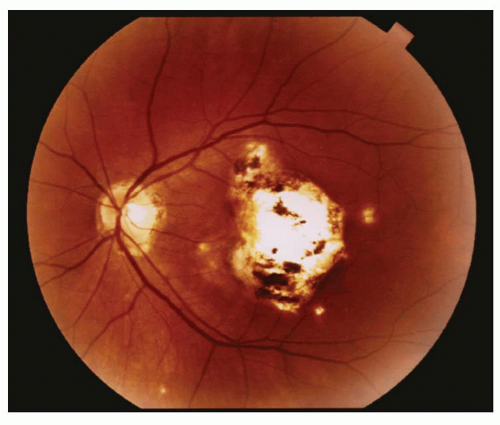
Posterior pole chorioretinal granuloma (˜25% to 50% of patients)—a white mass, 1 to 6 mm in diameter, is seen with an accompanying vitritis; usually located in the macula or in the papillomacular bundle (Fig. 18-8).
Endophthalmitis (<25% of patients)— chronic, diffuse, painless uveitis with a cloudy vitreous. There may be a secondary cataract, cyclitic membrane, and/or RD.
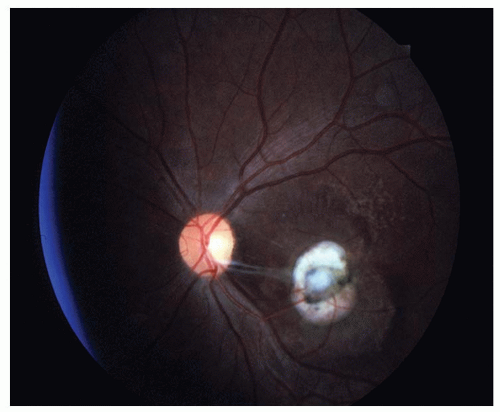 Figure 18-8. Toxocara posterior pole granuloma with fibrous vitreous bands extending from the lesion. |
The overall prevalence of Toxocara infection of the eye is about 1% of the general uveitis population.
Visceral larva migrans usually presents around the age of 2 years.
Most cases of ocular toxocariasis occur in children 5 to 10 years of age, with a range of 2 to 30 years. By the time the child seeks eye care, systemic manifestations have usually resolved.
The largest number of cases of ocular toxocariasis has been reported in the United States; however, the disease has been seen in many countries worldwide.
Is there known exposure to puppies, especially 2 weeks to 6 months of age? (>50% of puppies and 20% of adult dogs harbor the parasite)
Is there a history of geophagia?
Did the child play in a sand box?
Is there a history of ingesting uncooked foods?
is self-limiting, the amount of ocular absorption is unknown, and the reaction to a dead nematode can increase ocular inflammation. If oral antihelmenthics are employed, it is recommended that they be used in conjunction with steroids to decrease the inflammatory process.
albendazole 400 mg p.o. b.i.d. for 5 days. Use is controversial
prednisone 30 to 60 mg p.o. q.d. for 5 to 10 days followed by taper to control the vitreitis (in some cases, periocular injections of steroids may be preferable to oral usage).
the etiology of AMD. An inflammatory model was supported by genetic association studies which revealed allelic variants of genes coding the alternate complement pathway, particularly compliment factor H. Two genetic mutations, namely Tyr402HIS and Ala69Ser (both on the long arm of chromosome 10), may explain 75% of the genetic risk of AMD. These and other mutations have been clearly validated in white populations but do not seem to confer additional risks in other races. Other factors that predispose to AMD include systemic hypertension, cardiovascular disease, and smoking. Exposure to sunlight (ultraviolet light [UV]) was thought to play a role in the disease process, but no causal relationship was established.
Initially may be asymptomatic
Blurred vision
Metamorphopsia
Central blind spot
changes of AMD (large drusen and pigment mottling in the macula), and it is currently estimated that 1% will be legally blind (20/200 or less vision bilaterally) from the disorder. AMD affects primarily blue-eyed, white individuals, women more often than men. It rarely causes visual loss in African Americans. Many individuals with AMD have other family members affected by the disease.
advances in pharmacotherapy have considerably limited its use.
cells may represent the future of surgical approaches to AMD.
the later stages of the disease, there may also be noticeable color vision defects. Night blindness is usually not a complaint. Photophobia is rare as an early complaint, but some patients report that they are bothered by glare.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree