The nose is the most prominent feature of the human face. The importance of the nose to facial harmony has been well recognized throughout history. Nasal mutilation as a form of humiliation or punishment dates back to 1500 BC when Prince Lakshmana in India deliberately amputated the nose of Lady Surpunakha. King Ravana arranged for the reconstruction of Lady Surpunakha’s nose by his physicians, documenting one of the earliest accounts of nasal reconstruction in India . The historical account of nasal reconstruction in premodern times is well established, and many advancements have been made during the last few decades. Nonetheless, despite the fact that the repair of nasal defects is recognized as the oldest form of facial reconstructive surgery , its complexity continues to challenge reconstructive surgeons. This article focuses on the repair of small nasal defects following Mohs’ surgery; however, the principles discussed can be applied to most forms of nasal surgery.
Small nasal defects are repaired using several options, including healing by secondary intention, primary closure, local and regional flaps, skin grafts, and composite grafts. Occasionally, cartilage grafts or subcutaneous augmentation flaps are also necessary to establish appropriate support as well as contour match with the tissue surrounding the defect. Selection of the optimal reconstructive method is influenced by the size, depth, and location of the nasal defect.
Analysis of a nasal defect includes determining the depth of the wound, the color and texture of the missing skin, and the extent of involved esthetic nasal units and adjacent facial regions. In addition, the defect is inspected for any missing muscle, cartilage, or internal lining. The thickness, texture, and mobility of remaining nasal skin are important in determining reconstructive options; furthermore, any medical, social, and psychologic issues pertaining to the patient should be considered. Paramount to a successful reconstruction is appropriate assessment and treatment of any existing or potential nasal airway compromise.
Nasal anatomy
A thorough understanding of nasal anatomy is essential to successful nasal reconstruction. Given that the focus of this article is on small external nasal defects, an anatomic review pertinent to these types of repairs is provided.
The external nose consists of overlying skin, soft tissue, blood vessels, and nerves. Skin is thin and mobile over the dorsum, whereas it is thick and adherent to the underlying nasal framework at the nasal tip and alae. At the cephalic portion of the nasal sidewalls, the skin is thin; however, caudally, it becomes thicker near the nasal groove. Despite thicker skin at the nasal tip, the skin rapidly transitions to very thin skin covering the nostril margins and columella. The close approximation of the dermis of the skin lining and covering the nasal facets and nostril margins makes these areas especially vulnerable to notching and contour irregularities following reconstruction.
Four layers compose the soft tissue between skin and the bony cartilaginous skeleton of the nose. These layers are (1) the superficial fatty panniculus, (2) the fibromuscular layer, (3) the deep fatty layer, and (4) the periosteum or perichondrium . The superficial fatty panniculus is located immediately below the skin and consists of adipose tissue with interlacing vertical fibrous septi running from the deep dermis to the underlying fibromuscular layer. This layer is thicker in the glabellar and supratip areas. The fibromuscular layer contains the nasal musculature and the nasal subcutaneous muscular aponeurotic system, which is a continuation of the facial aponeurotic system. The deep fatty layer located between the subcutaneous muscular aponeurotic system and the thin covering of the nasal skeleton contains the major superficial blood vessels and nerves. This layer of loose areolar fat has no fibrous septae; as a result, immediately below it is the preferred plane for undermining nasal skin. The nasal bones and cartilages are covered with periosteum and perichondrium, which provide nutrient blood flow to these respective tissues. The periosteum of the nasal bones extends over the upper lateral cartilages and fuses with the periosteum of the piriform process laterally . Perichondrium covers the nasal cartilages, and dense interwoven fibrous interconnections can be found between the tip cartilages.
The nasal musculature has been described and classified by Greisman and Letourneau . The greatest concentration of muscle is located at the junction of the upper lateral and alar cartilages, allowing for muscular dilation and stenting of the nasal valve area. All nasal musculature is innervated by the zygomaticotemporal division of the facial nerve .
The internal and external carotid arteries contribute to the superficial arterial supply of the nose and adjacent area. The angular artery arises from the facial artery and provides a rich blood supply for laterally based nasal flaps. A branch of the angular artery, the lateral nasal artery, supplies the lateral surface of the caudal nose. The lateral nasal artery passes deep to the nose in the sulcus between ala and cheek and is covered by the levator labii superioris alaeque nasi. The artery branches multiple times to enter the subdermal plexus of the skin covering the nostril and cheek. The dorsal nasal artery, a branch of the ophthalmic artery, pierces the orbital septum above the medial palpebral ligament and travels along the side of the nose to anastomose with the lateral nasal artery. The dorsal nasal artery provides a rich axial blood supply to the dorsal nasal skin and serves as the main arterial contributor to dorsal nasal flaps ( Fig. 1 ).
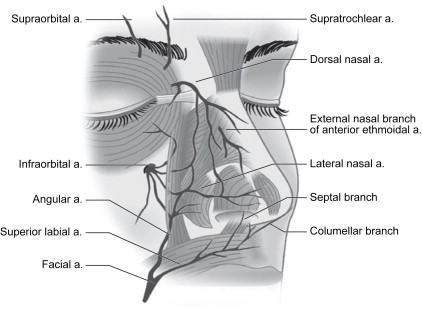
The nostril sill and columellar base are supplied by branches of the superior labial artery. A branch of the superior labial artery, the columellar artery, ascends superficial to the medial crura and is transected with a transcolumellar incision during an external rhinoplasty approach. In addition to the columellar artery, the nasal tip is supplied by the external nasal branch of the anterior ethmoidal artery.
The anterior ethmoidal artery, a branch of the ophthalmic artery, pierces bone on the medial wall of the orbit at the point where the lamina papyracea of the ethmoid bone articulates with the orbital portion of the frontal bone (frontoethmoid suture). The vessel enters the ethmoid sinuses to supply the mucosa and sends branches to the superior aspect of the nasal cavity. The external nasal branch of the anterior ethmoidal artery emerges between the nasal bone and the upper lateral cartilage to supply the skin covering the nasal tip. The venous drainage of the external nose consists of veins with names that correspond to the associated arteries. These veins drain via the facial vein, the pterygoid plexus, and the ophthalmic veins.
The sensory nerve supply of the nasal skin is supplied by the ophthalmic and maxillary divisions of the fifth cranial nerve. Branches of the supratrochlear and infratrochlear nerves supply the skin covering the radix, rhinion, and cephalic portion of the nasal sidewalls. The external nasal branch of the anterior ethmoidal nerve supplies the skin over the caudal half of the nose. The infraorbital nerve provides sensory branches to the skin of the lateral aspect of the nose.
The caudal third of the nose consists of the lobule (tip), columella, and alae. It is structurally supported by paired alar (lower lateral) cartilages, the caudal septum, accessory cartilages, and fibrous fatty connective tissue. The variable configuration of the nasal tip is dependent on the size, shape, orientation, and strength of the alar and septal cartilages, and by the quality and thickness of overlying soft tissue and skin. The alar cartilages are attached to the upper lateral cartilages and the septum, and they provide most of the support for the tip ( Fig. 2 ).
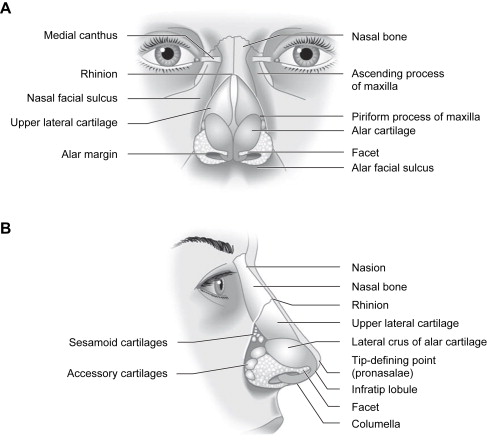
The alar cartilage is subdivided into medial, intermediate, and lateral crura. On lateral view, the internal structure responsible for the prominence of the tip-defining point is the cephalic edge of the domal segment of the intermediate crus. The shape, length, and angulation of the intermediate crura determine the configuration of the infratip lobule and the position of the tip-defining point. The lateral crus is the largest component of the alar cartilage and provides support to the anterior one half of the alar rim. Resection or weakening of the lateral crus predisposes to alar retraction and notching, an important consideration in nasal reconstruction. Laterally, small sesamoid cartilages are interconnected by a dense fibrous connective tissue that is contiguous with the superficial and deep perichondrium of the upper lateral cartilage and lateral crus. Inferolaterally, the ala contains fat and fibrous connective tissue but no cartilage. The shape and resiliency of the nostril depend on the dense fibrous fatty connective tissue located within the confines of the ala, and the integrity of this area should be restored with cartilage grafting when necessary.
The cartilaginous dorsum consists of paired upper lateral cartilages and the cartilaginous septum. The cephalic two thirds of the cartilaginous dorsum is a single cartilaginous unit, whereas, caudally, there is gradual separation of the upper lateral cartilages from the septum. The lateral borders of the upper lateral cartilages are rectangular in shape and are connected to the piriform aperture by an aponeurosis . The lateral border of the upper lateral cartilage creates a space known as the external lateral triangle. This space is bordered by the lateral edge of the upper lateral cartilage, the extreme lateral portion of the lateral crus, and the edge of the piriform fossa. The space is lined by mucosa and covered by the transverse portion of the nasalis muscle. It may contain accessory cartilages and fibrous fatty tissue that contribute to the lateral aspect of the internal nasal valve. Nasal obstruction may occur from medialization of this space secondary to scar contracture associated with trauma or surgery. The bony dorsum consists of paired nasal bones and paired ascending processes of the maxillae. The bony vault is pyramidal in shape, and the narrowest part is at the level of the intercanthal line.
Defect preparation
Nasal defects occasionally involve the nose and an adjacent area of the face. When planning reconstructive options, it is helpful to demarcate the division between the nose and surrounding facial regions (forehead, periorbita, cheeks, and lips) such that defects involving multiple facial regions are repaired with separate methods addressing each region. Adhering to this principle places eventual scars along lines that separate each esthetic region, helping to preserve the natural contours of the face.
Within each region, individual esthetic units should be identified. The esthetic units of the nose are based on variations in skin thickness and texture, as well as variations in nasal contour created by the underlying nasal framework. Optimal repair of a nasal defect may require repositioning of skin and soft tissue within an involved esthetic unit, allowing eventual scars to lie within zones of transitions between adjacent units ( Fig. 3 ). In addition, small defects are often enlarged to facilitate repair of an entire esthetic unit by a single regional flap (often a forehead or melolabial flap). This article focuses on repair of small defects that do not require reconstruction with regional flaps.
It is equally important to establish a uniform depth while maintaining symmetry with contralateral nasal units. Beveled tissue at the periphery of the Mohs’ defect is removed if flaps are planned to optimize the eversion of skin edges at closure. In addition, cutaneous edges of the flap and the recipient tissue are trimmed appropriately to establish uniform thickness at the line of closure. Whenever possible, the primary defect should be deepened to establish uniformity rather than thinning the flap. One exception to removing beveled edges is when skin grafts are performed. In this circumstance, beveled edges are freshened but maintained to provide a gradual contour transition between recipient tissue and the skin graft. Additional techniques helpful in optimizing repair include angulation of curvilinear defects, because round defects are more likely to undergo concentric scar contraction and result in a trapdoor deformity. Modifying the periphery of the defect by creating acute angles may reduce the risk of this deformity.
Defect preparation
Nasal defects occasionally involve the nose and an adjacent area of the face. When planning reconstructive options, it is helpful to demarcate the division between the nose and surrounding facial regions (forehead, periorbita, cheeks, and lips) such that defects involving multiple facial regions are repaired with separate methods addressing each region. Adhering to this principle places eventual scars along lines that separate each esthetic region, helping to preserve the natural contours of the face.
Within each region, individual esthetic units should be identified. The esthetic units of the nose are based on variations in skin thickness and texture, as well as variations in nasal contour created by the underlying nasal framework. Optimal repair of a nasal defect may require repositioning of skin and soft tissue within an involved esthetic unit, allowing eventual scars to lie within zones of transitions between adjacent units ( Fig. 3 ). In addition, small defects are often enlarged to facilitate repair of an entire esthetic unit by a single regional flap (often a forehead or melolabial flap). This article focuses on repair of small defects that do not require reconstruction with regional flaps.
It is equally important to establish a uniform depth while maintaining symmetry with contralateral nasal units. Beveled tissue at the periphery of the Mohs’ defect is removed if flaps are planned to optimize the eversion of skin edges at closure. In addition, cutaneous edges of the flap and the recipient tissue are trimmed appropriately to establish uniform thickness at the line of closure. Whenever possible, the primary defect should be deepened to establish uniformity rather than thinning the flap. One exception to removing beveled edges is when skin grafts are performed. In this circumstance, beveled edges are freshened but maintained to provide a gradual contour transition between recipient tissue and the skin graft. Additional techniques helpful in optimizing repair include angulation of curvilinear defects, because round defects are more likely to undergo concentric scar contraction and result in a trapdoor deformity. Modifying the periphery of the defect by creating acute angles may reduce the risk of this deformity.
Healing by secondary intention
Small cutaneous and superficial soft tissue defects involving concave surfaces of the nose may granulate and epithelialize with an acceptable esthetic result. Ideal locations for healing by secondary intention include the bony nasal sidewall and lateral alar groove, where the force of wound contracture is stabilized by the underlying soft tissue and nasal framework. This anatomy minimizes distortion of natural nasal contours . It is imperative to keep wounds moist during the healing phase. Wounds should be cleaned twice daily, removing fibrinous debris during each cleaning, and then covered with a topical petroleum-based ointment. For patients with sensitivity to petroleum-based ointments, a topical water-based ointment containing mupirocin may be substituted and applied more frequently. Patients are counseled that wounds often take 4 to 6 weeks to heal, and that after the wound has completely healed, massage may be indicated to address any contour irregularities. Patients are sometimes offered dermabrasion to the involved nasal esthetic units if surface irregularities are noted.
Skin grafts
Occasionally, superficial nasal defects are best addressed by repair with a skin or composite graft. Young patients with tight nasal skin and a propensity for multiple nasal skin cancers and elderly patients with thin atrophic skin are good candidates for this repair method. Placement of a graft obviates the need for additional scars on the nose as is seen with other reconstructive methods; however, most grafts have a “patch” appearance, with discrepancies in color and texture between native and grafted nasal skin. Post-operative dermabrasion minimizes these color and texture discrepancies.
Skin graft viability depends on several factors, including the blood supply to the recipient site, the microcirculation on the surface of the recipient site, the vascularity of donor graft tissue, contact between the graft and recipient site, and certain systemic illnesses. Contact between the skin graft and recipient site is essential. A bolster dressing prevents fluid collections and shearing forces from disrupting fibrous connections between the graft and wound bed. Systemic illnesses that may compromise graft survival include rheumatoid arthritis, lupus, hematologic disorders, diabetes, nutritional deficiencies, and hypoxemia . Use of tobacco products is also detrimental to the survival of skin grafts.
Recipient site conditions that are not favorable to graft survival include irradiated tissue, excessive fibrosis, and exposed bone, cartilage, or tendon. Grafts placed over avascular defects smaller than 1 cm 2 may survive through nutritional support via wound edges; however, grafting over avascular wounds larger than this is unlikely to succeed .
For deeper wounds, skin grafting may be delayed until granulation tissue has filled the wound bed (2 to 3 weeks), reducing the likelihood of a step-down deformity at the edges of the graft. In addition, the delay provides an improved vascular recipient site for the graft , which is especially important in areas of exposed periosteum or perichondrium. The wound is cared for in a similar fashion as healing by secondary intention. Any epithelium on the surface of the granulation tissue is removed before grafting, and the tissue is crosshatched so that myofibrils are released. Granulating wounds normally contain bacteria; bacterial counts greater than 10 5 organisms per gram of tissue often lead to graft loss . When delayed grafting is planned, the patient should be started on a course of an antistaphylococcal antibiotic several days before grafting.
Split-thickness skin grafts
Split-thickness grafts consist of epidermis and a variable portion of underlying dermis. Because of their poor color and texture match with normal skin and their tendency to contract, split-thickness skin grafts are rarely used to replace skin in nasal reconstruction.
Full-thickness skin grafts
Full-thickness skin grafts consist of epidermis and full-thickness dermis. They resist contraction, have a texture and pigmentation similar to normal skin, and require a well-vascularized, uncontaminated wound site for survival. Full-thickness grafts survive initially by diffusion of nutrition from fluid in the recipient site, a process known as plasma imbibition. This process is followed by vascular inosculation, which usually occurs during the first 24 to 48 hours. After 48 to 72 hours, capillaries in the recipient site begin to grow into the graft to provide new circulation. By 3 to 5 days, a new blood supply has been established. Initially, full-thickness skin grafts appear blanched; however, over 3 to 7 days, a pink color develops, signaling neovascularization. After 4 to 6 weeks, the pink color begins to fade, but the graft will often remain lighter than the surrounding skin, especially in darker skinned individuals.
When compared with split-thickness grafts, full-thickness grafts have the advantage of better color and texture match, less contour irregularities, no need for special equipment, and easier donor site wound care. The disadvantages include a reduced survival rate for larger grafts and a longer healing time .
The ideal nasal defect to repair with a full-thickness skin graft is superficial, with loss of skin but not underlying muscle. The vascularity of shallow wounds is greater than for defects extending through the muscle to the underlying cartilage or bone. The ideal defect is separated from the free margin of the nostril by 5 mm and is located in thin-skinned areas of the nose. These areas include the cephalic side-walls and dorsum and the infratip lobule. Shallow wounds in these areas are typically completely filled by a full-thickness skin graft, establishing a confluent contour with the surrounding skin.
Areas of the nose that are covered with thicker skin include the tip, ala, nasion, and caudal aspect of the sidewalls and dorsum. Full-thickness skin grafts used to repair defects of the nose in regions of thicker nasal skin tend to heal with a contour depression and noticeable textural discrepancies between the graft and adjacent nasal skin, because the nasal skin in these areas tends to have a more sebaceous nature than the graft. If a full-thickness graft has been performed and a contour depression exists, the appearance can be improved by subsequent placement of a dermal fat graft after the skin graft has healed ( Fig. 4 ). Meyers and coworkers describe the use of dermal grafts at the time of skin graft placement to help prevent contour depression following the repair of deeper defects. Their technique involves the placement of dermal tissue in linear strips within the wound bed, leaving adequate exposure of the underlying wound bed to provide nourishment to an overlying skin graft .




