Fig. 3.1
Anatomy of ear
Bone exposed defect (BD) of the EAM can result from various causes including injury, disease, congenital anomalies, and postoperative damage. They are a frequent cause of recurrent infection often accompanied by pain, malodor, otorrhea, and bleeding. This condition imposes many restrictions on the patient’s daily life such as the inability to take a bath, shampoo one’s hair, or go swimming. BD of the EAM is a particularly serious problem for patients who need to wear hearing aids.
To date, almost all treatments for these types of defects have involved surgical repair requiring incisions of the postauricular skin, the harvesting of autologous tissue, and other invasive procedures. These surgical treatments demand that patients endure multiple discomforts including anesthesia, bed rest, and/or hospitalization, the high-priced cost of the operation, restrictions on daily life, and cosmetic sequelae to name a few. More importantly, these types of invasive treatments are not always successful and often pose other risks including the development of iatrogenic cholesteatomas and deformity of the EAM. Additionally, conventional surgical techniques are unable to restore the normal shape of the EAM. The undamaged EAM has a self-cleaning function, which keeps the sound pathway clear of debris. A mechanism of epithelial migration moves surface keratin laterally from the tympanic membrane toward the ear canal opening. This desquamation and skin migration aids in moving objects out of the ear canal. Therefore, in order to maintain normal EAM morphology, it is very important that the EAM retain these functions.
The need for an effective treatment strategy is not limited to large BD of the EAM, as even acute BD often fails to heal spontaneously. This is due to the fact that the soft tissue of the EAM is very thin and somewhat limited in blood supply. Thus, it is very important that regeneration strategies for BD of the EAM consist of a better arrangement of regenerative conditions such as ensuring an abundant blood supply as well as an efficient seal for the prevention of infection and the maintenance of moisture within the tissue.
As present treatments for BD of the EAM stand on the concept of reconstructive surgery, they are woefully insufficient from the points of both morphology and function.
We newly developed the regenerative treatment for BD of the EAM as follows (Fig. 3.2).
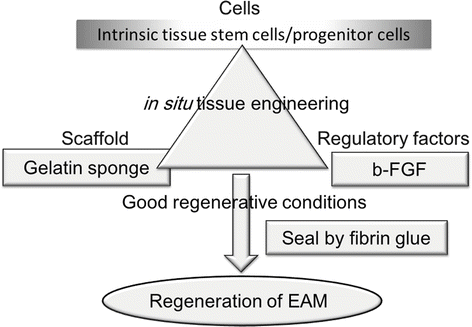

Fig. 3.2
Schematic of the tissue engineering triad and our strategy for regenerating BDs of the EAM. According to the principles of in situ tissue engineering, regeneration of tissues/organs requires the introduction of three basic elements – cells, scaffolds, and regulatory factors – under suitable conditions. In this clinical study, we introduced just two of these elements – scaffolding and a regulatory factor – as cells were expected to be endogenously supplied from the mechanically disrupted edge of the defect of EAC. That is, mechanical disruption of the defect edge may stimulate the activity of tissue stem cells/progenitor cells of the EAC in this region
3.2 Patients and Therapeutic Materials
Thirty patients consisting of 12 males and 18 females participated in this clinical study. These patients were selected from outpatients with EAM cholesteatomas (n = 13), EAM cholesteatomas following middle ear surgery (n = 4), traumatic BDs of the EAM (n = 6), BDs of the EAM following middle ear surgery (n = 3), and benign tumor of the EAM (n = 4). Patient ages ranged from 15 to 82, with an average age of 55.
They were classified into three grades based on the size of their BDs: below 100 mm2 as Grade I (n = 11), from 100 to 200 mm2 as Grade II (n = 12), and over 200 mm2 as Grade III (n = 7). Patients were divided into two groups: the b-FGF group (n = 20: Grade I = 7, II = 8 and III = 5) and the control group (n = 10: Grade I = 4, II = 4, and III = 2).
Materials used for repairing the BD included a gelatin sponge (Spongel: Astellas Pharma Inc. Tokyo, Japan) with basic fibroblast growth factor (b-FGF, Fibrast: Kaken Pharma Co., Ltd Tokyo, Japan) and a fibrin glue (Bolheal: Kaketsuken, Kumamoto, Japan or Beriplast: CSL Behring K.K., Tokyo, Japan) (Fig. 3.3a, b).


Fig. 3.3
(a-1) b-FGF. (a-2) Gelatin sponge. (b-1) Trimmed gelatin sponge. (b-2) Gelatin sponge with b-FGF
The study subjects were limited to patients who fully understood the procedure and agreed to pursue this new treatment by signing “Informed Consent” documents. This new therapy was approved by the ethical committee of the Medical Research Institute Kitano Hospital in Osaka and Kanai Hospital in Kyoto Japan; all procedures followed were in accordance with the Helsinki Declaration.
3.3 Therapeutic Procedures
Figure 3.4 shows all procedures. The soft tissue of the EAM was fully anesthetized by applying a cotton ball soaked in 4 % lidocaine for 15 min in most cases of Grade I or II. The patients of Grade III and a part of Grade I and II were anesthetized by injecting 1.0 % lidocaine with epinephrine. In cases with cholesteatomas or benign tumors, the entire growth was removed along with the surrounding tissue. In cases of simple soft tissue defects, a mechanical disruption of the defect edge was created. Both of the procedures described above were performed under the microscope by oto-microsurgery. In the b-FGF group, a gelatin sponge slightly larger than the defect was immersed in b-FGF (10–300 μg: 100 μg/mL of Trafermin, a recombinant human b-FGF); the sponge was placed such that it was in contact with the edge of the defect. The bone surface had been made smooth using a micro-osteotrite in the case of severe cholesteatoma. In the control group, the same treatment was performed using a gelatin sponge that had been immersed in saline. Fibrin glue was then dripped over the sponge to form a seal. In cases with very large BDs comprising the entire area of the EAM, the EAM was packed with gelatin sponge soaked in b-FGF and sealed with fibrin glue. In these extreme cases, the auricle was then dressed with a waterproof transparent dressing (Tegaderm: Sumitomo 3M Limited, Tokyo, Japan) (Fig. 3.4).
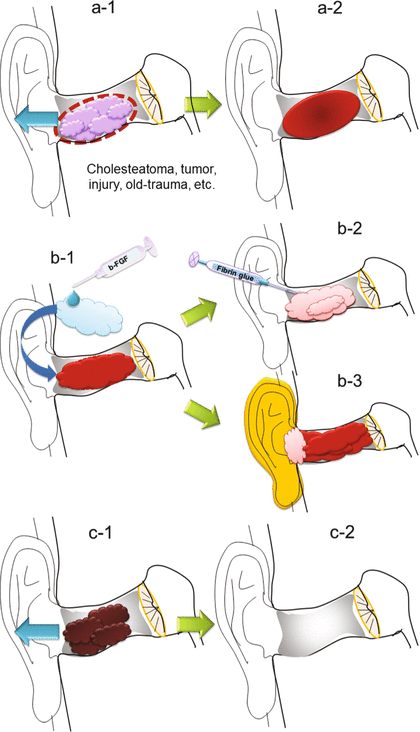

Fig. 3.4
Procedures used in this treatment: (a-1) After fully anesthetizing the EAM by application of a cotton ball soaked in 4 % lidocaine for 15 min, any lesions such as cholesteatomas or benign tumors are resected along the dotted line. (a-2) A mechanical disruption of the EAM defect edge is created. Depending on the cases, a concave of the bone is made smooth by a drill/a micro-sharp spoon after removing the matrix of cholesteatoma or a tumor. (b-1) Trimmed gelatin sponge soaked in b-FGF is placed over the defect. (b-2) The gelatin sponge is sealed with fibrin glue. (b-3) In cases with very large BDs comprising the entire area of the EAM, after the EAM was packed with gelatin sponge soaked in b-FGF and sealed with fibrin glue, the auricle was then dressed with a waterproof transparent dressing. (c-1) Three weeks after treatment, crust over the EAM defect is removed. (c-2) In case complete regeneration has not been achieved, the above same treatment is performed repeatedly until three times
Following the procedure, patients were advised to avoid touching their ear and to keep their ear dry and were ordered to revisit the hospital 2 or 3 weeks after treatment at which point any crust over the defect was removed.
3.4 Evaluation
Three weeks after the procedure, the effectiveness of this treatment was evaluated by the images of fiberscope and microscope. In cases in which complete closure of the defect was not achieved, the above treatment was repeated up to three times. The final evaluation was performed 3 months after the initial treatment. The success rate was determined based on the rate of complete closure of the EAM BDs within three courses of treatment.
The occurrence of negative side effects and sequelae were also evaluated. Average follow-up period was 24 months (from 3 to 30 months).
In five cases, in order to evaluate a self-cleaning function, which keeps regenerated EAM clear of debris, we observed migration of pigment (Pyoctanin/Crystal violet: Methylrosanilinium Chloride) marked on the regenerated site for 4 weeks (Fig. 3.8).
3.5 Results
3.5.1 Complete BD Closure Rates
Figures 3.5, 3.6, 3.7, and 3.8 illustrate typical cases of regenerated BDs.
Get Clinical Tree app for offline access

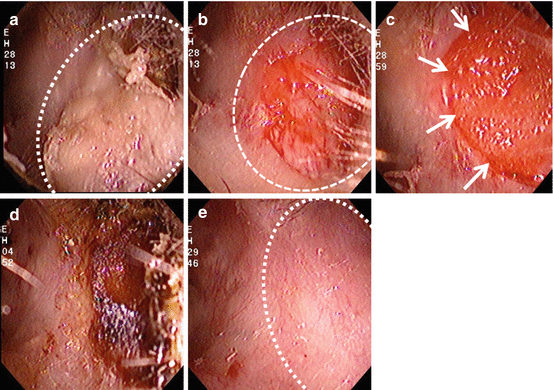
Fig. 3.5
Typical case of BD regeneration in a 67-year-old female with EAM cholesteatoma after middle ear surgery (Grade II). (a) White dotted circle indicates the cholesteatoma. (b) After the cholesteatoma was removed, a portion of the bone was exposed (white dotted circle). (c) Gelatin sponge containing b-FGF was placed into/over the defect and sealed with fibrin glue (white arrows). (d) Three weeks after treatment, dried gelatin sponge and crust covered the defect. (e) One month after treatment, the BD of the EAM was completely regenerated (white dotted circle)

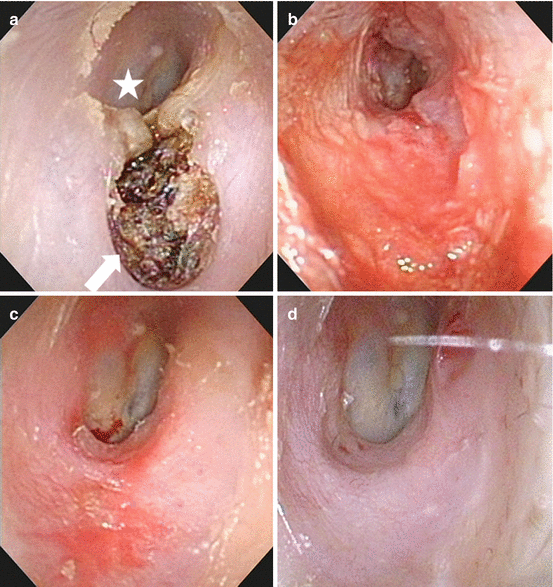
Fig. 3.6
Example of cholesteatoma with extreme bone defect of the EAM in a 71-year-old male. (a) Before treatment. White asterisk tympanic membrane, white arrow cholesteatoma with extreme bone defect of the EAM. (b) One month after treatment. (c) Two months after treatment. (d) One year after treatment, the BD of the EAM was completely regenerated

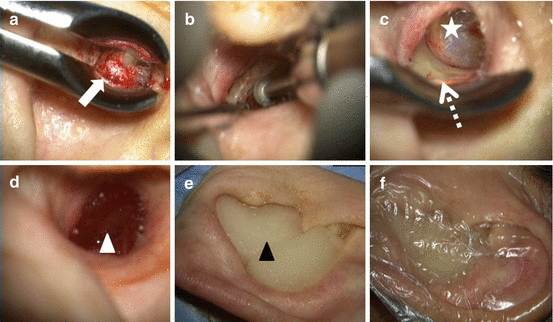
Fig. 3.7
Example of cholesteatoma with a very large BD encompassing the entire area of the EAM in a 72-year-old female. (a) White arrow indicates the cholesteatoma in the left EAM. (b) After resection of cholesteatoma, the bone surface of EAM is making smooth by diamond bar. (c




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


