Purpose
To evaluate the efficacy and safety of reduced-fluence photodynamic therapy (PDT) combined with bevacizumab for polypoidal choroidal vasculopathy (PCV).
Design
Prospective, noncomparative, interventional case series.
Methods
Sixteen treatment-naïve patients with polypoidal choroidal vasculopathy were treated with reduced-fluence PDT combined with bevacizumab. All patients were followed up monthly for 12 months with measurements of best-corrected visual acuity (BCVA) and central foveal thickness by optical coherence tomography. Indocyanine green angiography and fluorescein angiography were performed every 3 months. Patients were re-treated with reduced-fluence PDT combined with bevacizumab or with sole injection of bevacizumab when indicated.
Results
The mean logMAR BCVA showed significant improvement from 0.76 at baseline to 0.46 at 12 months ( P = .002). At 12 months, the BCVA improved in 9 eyes (56.3%) by 3 lines or more, was stable in 6 eyes (37.5%), and decreased in 1 eye (6.3%) because of recurrence of polyps. During the study period, 3 patients (18.8%) had recurrence of polyps and 2 patients (12.5%) had persistent polyps. Mean episodes of reduced-fluence PDT and mean injections of intravitreal bevacizumab over 12 months were 1.44 and 2.44, respectively. Although 3 patients had mild choroidal nonperfusion—1 eye after 1 session of PDT and 2 eyes after 2 sessions—no severe complications, including endophthalmitis, uveitis, or subretinal hemorrhage, developed.
Conclusion
Reduced-fluence PDT combined with bevacizumab for PCV seemed to be effective for improving vision and reducing complications. Further study to optimize the light dose of PDT in combination therapy is needed in order to achieve better treatment outcomes for PCV.
Polypoidal choroidal vasculopathy (PCV) is a peculiar choroidal vascular lesion appearing as interconnecting branches of vessels with terminal polyps, which is characterized by multiple, recurrent, serosanguineous pigment epithelium detachment and neurosensory retinal detachment. The prevalence of PCV is higher in Asian populations than in white patients, and has been reported to be 23% to 54.7% in Japanese patients, 22.3% in Chinese patients, and 24.6% in Korean patients diagnosed with neovascular age-related macular degeneration (AMD). The pathogenesis of PCV still is not fully understood; it is considered to be a variant of AMD or a choroidal vascular abnormality characterized by protruding aneurysmal vascular lesions derived from the inner choroidal circulation.
Treatment of PCV is not well established, and a conservative approach is advocated if visual acuity is not affected by progressive exudative changes. Previous studies have indicated that photodynamic therapy (PDT) with verteporfin may be an effective treatment for symptomatic PCV. However, subretinal and subpigment epithelial hemorrhage are potential complications of PDT, with the incidence ranging from 4.7% to 30.8%, and recurrence of polyps has been reported from 4.5% to 50% over 1 year. Several studies have shown that conventional PDT following the Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) guidelines causes damage to the physiological choroid in most eyes. This has been demonstrated by indocyanine green angiography (ICGA) showing choriocapillary nonperfusion, by histology showing dose-dependent thrombosis of the choriocapillaris, and by immunochemical staining showing reactive upregulation of vascular endothelial growth factor (VEGF). Furthermore, increased expression of VEGF after PDT might potentially increase the risk of angiogenesis recurrence. Eandi and associates reported that selective PDT treating only the area of the active PCV or “hot spot” evident on ICGA without extra margin might reduce collateral damage to the choriocapillaris and upregulation of VEGF. The Visudyne in Minimally Classic Choroidal Neovascularization (VIM) trial demonstrated a trend toward better visual outcomes when reduced-fluence PDT was used, compared with standard-fluence PDT. The Verteporfin and Bevacizumab (VIA) study showed that 2 groups treated by combination of intravitreal bevacizumab and 25 J/cm 2 or 12 J/cm 2 PDT had favorable results, reducing the number of intravitreal bevacizumab treatments compared to bevacizumab alone. Yamashita and associates showed favorable 1-year results using reduced-fluence PDT for PCV.
Anti-VEGF drugs, including pegaptanib, bevacizumab, and ranibizumab, have recently become available for use in treatment of choroidal neovascularization (CNV) in AMD. However, the efficacy for PCV seems to be limited. Previous studies have shown that intravitreal bevacizumab resulted in reduction of exudative fluid, but that it seems to be ineffective in causing regression of polypoidal lesions in ICGA.
Thereafter, combination therapy of PDT and intravitreal bevacizumab was introduced as an additional strategy for treatment of PCV, considering synergetic effects. It resulted in favorable outcomes, including improved visual acuity, reduced recurrence rate, and low complications. However, it is uncertain whether combined therapy with reduced-fluence PDT and intravitreal bevacizumab would be effective. Thus, we aimed to investigate the efficacy and safety of combined therapy with reduced-fluence PDT and intravitreal bevacizumab in treatment of PCV.
Methods
This was a prospective, consecutive, open-label, noncomparative, interventional study conducted at the Eye Clinic of Yeungnam University Hospital.
Patient Selection
Sixteen eyes of 16 patients with PCV were enrolled in this study between December 1, 2008 and August 31, 2009. Inclusion criteria of this study included 1) age older than 50 years, 2) symptomatic PCV in the macular area, 3) best-corrected visual acuity (BCVA) of 20/40 or worse, 4) presence of polyps with or without branching vascular network vessels on ICGA, and 5) a greatest linear dimension (GLD) of 5400 μm or less. Exclusion criteria included 1) previous treatment for PCV, 2) existence of drusen suggesting CNV secondary to AMD, 3) refractive error of higher than -6 diopters (D), 4) large subretinal hemorrhage, and 5) an area of scar tissue accounting for 50% or more of the lesion of PCV.
All participants received a comprehensive ocular examination at baseline and at monthly-interval visits for 12 months. The examination included BCVA, dilated fundus, and macular examination with a 90-D lens; color fundus photography; and central foveal thickness (CFT) by optical coherence tomography (OCT) (OCT Model 3000; Zeiss, Dublin, California, USA). The BCVA was converted to logarithm of the minimal angle of resolution (logMAR) equivalents. Each 0.10-unit difference in logMAR visual acuity (VA) was considered as 1 line. Fluorescein angiography (FA) and ICGA were performed using a scanning laser ophthalmoscope (Heidelberg Retinal Angiography II; Heidelberg Engineering, Heidelberg, Germany) at baseline and at every 3-month visit.
Initial Treatment
We planned combined therapy with an intravitreal bevacizumab (Avastin; Genentech, San Francisco, California, USA) and a session of reduced-fluence PDT (Visudyne; Novartis AG, Basel, Switzerland) as the initial treatment. PDT was performed approximately 7 days after intravitreal bevacizumab. It was assumed that intravitreal bevacizumab might permit more efficient delivery of PDT laser to sub–retinal pigment epithelium (RPE) lesions and reduced-fluence PDT following intravitreal bevacizumab might be enough to resolve polypoidal lesions. The interval of combined therapy was established based on previous studies demonstrating significant regression of macular edema 1 week after intravitreal bevacizumab in the patients with diabetic macular edema and macular edema secondary to retinal vein occlusion.
Bevacizumab (1.25 mg/0.05 mL) was injected 3.5 mm posterior to the limbus using a 30-gauge needle under strict aseptic techniques with topical anesthesia. A topical antibiotic was applied 4 times daily for 7 days.
PDT was administered with a reduced fluence of 25 J/cm 2 for 83 seconds (300 mW/cm 2 ) after verteporfin injection in standard fashion. The laser spot size was chosen by addition of 1000 μm to the entire PCV lesions, including polypoidal lesions and branching vascular network vessels determined by ICGA.
Re-Treatment
FA and ICGA were performed at baseline and every 3 months to assess the resolution and recurrence of PCV lesions. If active polypoidal lesions associated with exudative fluid were seen on ICGA, re-treatment with reduced-fluence PDT combined with intravitreal bevacizumab was performed using the same method as that of the initial treatment described previously. However, if ICGA could not identify active polyps associated with exudative fluid demonstrated by FA or OCT (if the exudative fluid was associated only with the vascular network component of PCV on ICGA), intravitreal bevacizumab without reduced-fluence PDT was solely administered. During intermediate visits, if exudative fluid was present on OCT, intravitreal bevacizumab was solely chosen as the option for re-treatment. Figure 1 shows a summary of re-treatment protocol. Treatment endpoint was the disappearance of active polypoidal lesions on ICGA with complete resolution of exudative fluid on OCT.

Main Outcomes and Statistical Analysis
Visual and tomographic outcomes were analyzed every month during a 12-month follow-up period. Visual outcomes included the mean BCVA change in logMAR and the proportion of eyes that had improved (≥3 lines), stable (within 2 lines), or decreased (≥3 lines) vision after the initial treatment when compared to the baseline. Tomographic outcomes included the mean change of CFT from baseline after the initial treatment. Wilcoxon signed rank test was used to determine the significance of changes of the mean BCVA and CFT from the baseline.
Angiographic outcomes were evaluated every 3 months during the study period, which included the proportion of eyes with complete resolution of leakage on FA and total regression of polyps on ICGA from baseline after the initial treatment. Other important parameters included the number of treatments required during the study period.
Results
Clinical data from 16 eyes of 16 patients enrolled consecutively during the study period are shown in Table 1 .
| Best-Corrected Visual Acuity (BCVA) | OCT | Polyp change in ICGA | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | 12 mo | ||||||||||||||||||
| No. | Age | GLD (μm) | Baseline | 3 mo | 6 mo | 9 mo | 12 mo | CFT (μm) | Findings | CFT (μm) | Findings | Initial FA | 3 mo | 6 mo | 9 mo | 12 mo | No. of PDT | No. of IVB | Cx |
| 1 | 65/M | 4253 | 1.20 | 1.00 | 0.78 | 1.00 | 1.02 | 396 | SRF | 213 | N-S | O-CNV | Di | Di | Di | Di | 1 | 5 | Mild a |
| 2 | 61/F | 2467 | 0.40 | 0.20 | 0.12 | 0.20 | 0.18 | 304 | SRF | 184 | N-S | O-CNV | Di | Di | Di | Di | 1 | 2 | No |
| 3 | 69/M | 1472 | 1.20 | 1.00 | 1.20 | 1.20 | 1.00 | 332 | SRF+PED | 225 | N-S | O-CNV | Di | Di | Di | Di | 1 | 4 | No |
| 4 | 75/F | 2637 | 0.42 | 0.20 | 0.18 | 0.30 | 0.70 | 442 | SRF | 310 | SRF | O-CNV | Di | Di | Di | Re | 2 | 4 | No |
| 5 | 69/F | 4526 | 0.48 | 0.40 | 0.40 | 0.30 | 0.30 | 408 | SRF+PED | 245 | N-S | MC-CNV | Pe | Di | Di | Di | 2 | 3 | No |
| 6 | 71/M | 4320 | 1.00 | 0.52 | 0.40 | 0.40 | 0.40 | 263 | SRF | 183 | N-S | O-CNV | Di | Di | Di | Di | 1 | 1 | No |
| 7 | 52/M | 3876 | 0.50 | 0.20 | 0.10 | 0.10 | 0.10 | 300 | SRF | 192 | N-S | O-CNV | Di | Di | Di | Di | 1 | 1 | No |
| 8 | 58/M | 4354 | 0.70 | 0.38 | 0.20 | 0.30 | 0.30 | 314 | SRF+PED | 214 | N-S | O-CNV | Di | Di | Di | Di | 1 | 1 | No |
| 9 | 59/M | 5247 | 1.20 | 0.80 | 0.70 | 0.70 | 0.70 | 463 | SRF+PED | 193 | N-S | O-CNV | Di | Di | Di | Di | 1 | 1 | No |
| 10 | 69/M | 4876 | 0.70 | 0.30 | 0.20 | 0.20 | 0.30 | 399 | SRF | 224 | N-S | O-CNV | Di | Di | Di | Di | 1 | 1 | No |
| 11 | 69/M | 2245 | 0.70 | 0.40 | 0.30 | 0.36 | 0.30 | 272 | SRF | 220 | SRF | O-CNV | Di | Re | Re | Re | 4 | 5 | No |
| 12 | 72/M | 2673 | 0.42 | 0.20 | 0.20 | 0.14 | 0.10 | 377 | SRF | 228 | N-S | O-CNV | Di | Di | Di | Di | 1 | 1 | No |
| 13 | 72/M | 2379 | 0.38 | 0.54 | 0.40 | 0.40 | 0.30 | 391 | SRF+PED | 193 | PED | O-CNV | Di | Di | Di | Di | 1 | 2 | No |
| 14 | 70/M | 3663 | 0.50 | 0.36 | 0.50 | 0.50 | 0.40 | 331 | SRF | 176 | N-S | O-CNV | Pe | Di | Di | Di | 2 | 2 | Mild |
| 15 | 65/F | 2478 | 2.00 | 1.20 | 1.20 | 1.20 | 1.10 | 252 | SRF | 158 | N-S | O-CNV | Di | Di | Di | Di | 1 | 1 | No |
| 16 | 69/M | 3125 | 0.40 | 0.30 | 0.30 | 0.20 | 0.10 | 412 | SRF+PED | 216 | PED | O-CNV | Di | Re | Di | Di | 2 | 5 | Mild |
| Average | 66.6 | 3411 | 0.76 | 0.50 | 0.45 | 0.47 | 0.46 | 352.9 | 210.4 | 1.44 | 2.44 | ||||||||
a Mild = mild choriocapillary nonperfusion (grade 1 based on grading by Michels and associates ).
Visual Outcomes
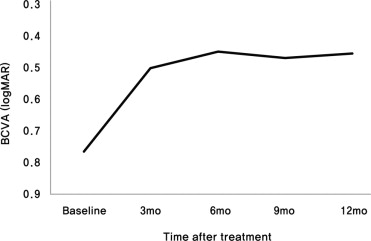
The logMAR BCVA (mean ± standard deviation) was 0.76 ± 0.45 at baseline, 0.50 ± 0.32 at 3 months, 0.45 ± 0.35 at 6 months, 0.47 ± 0.36 at 9 months, and 0.46 ± 0.34 at 12 months ( Figure 2 ) . Changes in BCVA showed significant difference when compared to baseline at 3, 6, 9, and 12 months (2.62 lines and P = .001, 3.12 lines and P = .001, 2.95 lines and P = .001, and 3.06 lines and P = .002, respectively). Data showed that the significant gain from the baseline of the mean BCVA was already achieved at 3 months and was maintained until 12 months.
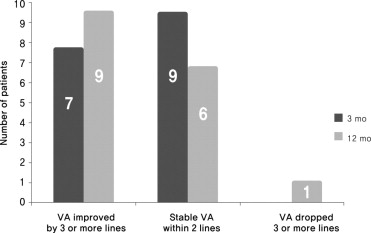
The proportions of eyes with improved vision of 3 lines or more was 7 of 16 eyes (43.7%) and 9 of 16 eyes (56.3%), and the proportions of eyes with stable vision was 9 of 16 eyes (56.3%) and 6 of 16 eyes (37.5%), at 3 and 12 months, respectively ( Figure 3 ) . One eye (6.3%, Patient 4) had decreased vision attributable to newly developed polyps at 12 months. Patient 4 had been treated with 1 session of PDT and 3 injections of intravitreal bevacizumab before the recurrence of lesions. We did not define any eyes with a decrease in vision, except for this one, during the study period.
Angiographic and Tomographic Outcomes
Baseline classification of the neovascular lesions of the 16 study eyes, using FA, demonstrated occult CNV in 15 eyes (93.8%) and minimally classic CNV in 1 eye (6.3%). ICGA showed that 15 eyes (93.8%) had a dilated network of inner choroidal vessels with polyps and 1 eye (6.3%) had only polyps without the vascular network.
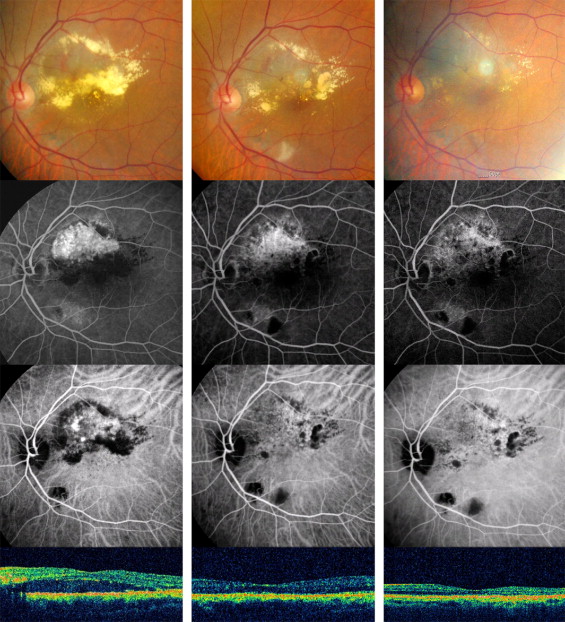
After the first session of combined therapy, ICGA showed resolution of polypoidal lesions in 14 of 16 eyes (87.5%) at 3 months and persistent polyps in 2 eyes (12.5%). By 6 months, the 2 eyes with persistent polyps showed complete regression with additional combined therapy. During the study period, recurrent polypoidal lesions on ICGA appeared in 3 eyes (18.8%). Two eyes (12.5%; Patients 11 and 16) had reactive polyps on the same location and 1 eye (6.3%; Patient 4) had newly developed polyps on a location that differed from that of the original lesions. However, in all 15 eyes that had both polyps and branch vascular network, the abnormal choroidal vascular network showed no definite changes in size and shape.
Of particular interest, many eyes had remaining or recurrent leakage on FA or fluid accumulation on OCT, although they had complete polyp regression on ICGA. At 3 months, although 14 of 16 eyes (87.5%) had polyp regression on ICGA, 3 of the 14 eyes had remaining leakage on FA. During the same follow-up period, 4 of 14 eyes experienced recurrent fluid accumulation on monthly OCT. By 6 months, although 14 of 16 eyes (87.5%) achieved polyp regression on ICGA, 3 of 14 eyes still had leakage on FA. From 4 to 6 months, 4 of 14 eyes showed recurrent fluid accumulation on monthly OCT. By 9 months, although 15 of 16 eyes (93.8%) achieved polyp regression on ICGA, 1 of the 15 eyes still had leakage on FA. During the follow-up period from 7 to 9 months, 2 of 15 eyes showed recurrent fluid accumulation on monthly OCT. By 12 months, 14 eyes had polyp regression on ICGA and all of the 14 eyes showed no leakage on FA and no fluid accumulation on OCT.
At baseline, OCT showed that all eyes had subretinal fluid involving the fovea, and 4 of 16 eyes (25.0%) had associated PED. At the 12-month follow-up, OCT demonstrated an absence of subretinal fluid in 14 of 16 eyes (87.5%), with persistent subretinal fluid and residual PED in 2 eyes and 3 eyes, respectively. The mean CFT showed a significant decrease, from 352.9 ± 66.8 μm at baseline to 236.4 ± 59.0 μm at 3 months ( P = .001), which was maintained throughout follow-up with a mean CFT of 210.9 ± 34.9 μm at 12 months ( P = .000).
Fundus photographs, FA, ICGA, and OCT of Patient 11 before and after treatment are displayed in Figure 4 .