Purpose
To evaluate efficacy and safety of quarterly (and then monthly) ranibizumab during the 2-year P hase IIIb, multicenter, randomized, double-masked, sham i njection–controlled study of the e fficacy and safety of r anibizumab in subjects with subfoveal CNV with or without classic CNV secondary to AMD (PIER) study.
Design
Phase IIIb, multicenter, randomized, double-masked, sham injection–controlled trial in patients with choroidal neovascularization (CNV) secondary to age-related macular degeneration (AMD).
Methods
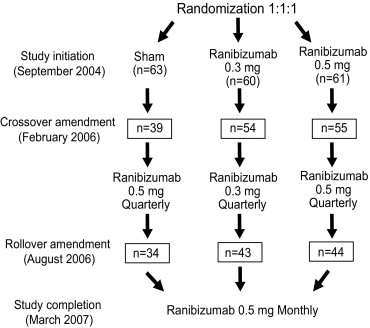
Patients were randomized 1:1:1 to sham injection (n = 63) or 0.3 mg (n = 60) or 0.5 mg (n = 61) intravitreal ranibizumab monthly for 3 months and then quarterly. During study year 2, eligible sham-group patients crossed over to 0.5 mg ranibizumab quarterly. Later in year 2, all eligible randomized patients rolled over to 0.5 mg ranibizumab monthly. Key efficacy and safety outcomes of the 2-year trial are reported.
Results
At month 24, visual acuity (VA) had decreased an average of 21.4, 2.2, and 2.3 letters from baseline in the sham, 0.3 mg, and 0.5 mg groups ( P < .0001 for each ranibizumab group vs sham). VA of sham patients who crossed over (and subsequently rolled over) to ranibizumab decreased across time, with an average loss of 3.5 letters 10 months after crossover. VA of 0.3 mg and 0.5 mg group patients who rolled over to monthly ranibizumab increased for an average gain of 2.2 and 4.1 letters, respectively, 4 months after rollover. The ocular safety profile of ranibizumab was favorable and consistent with previous reports.
Conclusions
Ranibizumab provided significant VA benefit in patients with AMD-related CNV compared with sham injection. Ranibizumab appeared to provide additional VA benefit to treated patients who rolled over to monthly dosing, but not to patients who began receiving ranibizumab after >14 months of sham injections.
Neovascular age-related macular degeneration (AMD) is characterized by new vessel growth and leakage in the choroidal vascular network beneath the macula, with extension and leakage into the subretinal space. Although the pathologic events that precede choroidal neovascularization (CNV) are not clearly understood, disrupting the activity of vascular endothelial growth factor A (VEGF-A), a diffusible cytokine that promotes angiogenesis and vascular permeability, effectively treats CNV secondary to AMD.
Ranibizumab (Lucentis; Genentech, South San Francisco, California, USA) is an intravitreally administered recombinant, humanized, monoclonal antibody antigen-binding fragment that neutralizes all known active forms of VEGF-A. In 2 Phase III pivotal studies––the MARINA study in patients with minimally classic or occult with no classic CNV and the ANCHOR study in patients with predominantly classic CNV––monthly intravitreal injections of 0.3 mg or 0.5 mg ranibizumab not only prevented vision loss but also improved visual acuity (VA) compared with sham injections or photodynamic therapy (PDT) with verteporfin.
Subsequently, a P hase IIIb, multicenter, randomized, double-masked, sham i njection–controlled study of the e fficacy and safety of r anibizumab in subjects with subfoveal CNV with or without classic CNV secondary to AMD (PIER) evaluated adverse events and VA benefit of quarterly dosing in patients with neovascular AMD. The PIER dosing schedule—monthly for 3 months and then quarterly—was selected based on Phase I and II studies, which indicated that the VA benefits of 0.3 mg and 0.5 mg ranibizumab administered intravitreally monthly for 3 months may last up to 90 days.
While ranibizumab administered on the PIER dosing schedule provided significant VA benefit compared to sham injections in patients with neovascular AMD, quarterly dosing with ranibizumab did not provide the VA benefit demonstrated by monthly dosing in the MARINA and ANCHOR studies. In fact, during study year 2, after careful review of available clinical data, including the 12-month data from MARINA and ANCHOR, the PIER protocol was amended to provide all PIER patients the opportunity to receive ranibizumab.
Here, we present VA and safety outcomes over 2 years in the PIER study, showing that the VA benefit of quarterly ranibizumab treatment was maintained well into the second year of the study. Furthermore, switching to monthly ranibizumab treatment late in year 2 appeared to provide increased VA benefit to patients who had previously been treated quarterly, while ranibizumab treatment appeared not to provide a VA benefit to control patients who began receiving ranibizumab after a year without treatment.
Methods
PIER methodology, including study design, eligibility, masking, treatment, assessments, and analyses, has been published in detail. All patients provided informed written consent prior to participation. Briefly, eligible patients were at least 50 years of age with a diagnosis of primary or recurrent subfoveal CNV (predominantly classic, minimally classic, or occult with no classic) secondary to AMD and baseline best-corrected VA of 20/40 to 20/320 Snellen equivalent, measured using the Early Treatment Diabetic Retinopathy Study (ETDRS) chart at a distance of 4 meters.
Classic and/or occult CNV comprised ≥50% of the total AMD lesion area, and the total lesion was ≤12 disc areas (DA). If a CNV lesion was minimally classic or occult with no classic component, the treated eye was required to meet protocol-defined criteria for disease progression (ie, a 10% increase in lesion size based on fluorescein angiography [FA] obtained 1 month prior to study initiation [ie, day 0] compared to FA obtained 6 months prior to day 0; ≥5 ETDRS letter [1 Snellen line] VA loss within 6 months prior to day 0; or CNV-associated subretinal hemorrhage 1 month prior to day 0). Patients who had fibrosis or atrophy involving the center of the fovea or subretinal hemorrhage ≥1 DA or ≥50% of total lesion area with foveal involvement were excluded. One eye per patient was studied.
Eligible patients were randomized 1:1:1 to sham injections, 0.3 mg intravitreal ranibizumab, or 0.5 mg intravitreal ranibizumab ( Figure 1 ). Patients were masked to treatment. Randomization was stratified by best-corrected VA (≤54 ETDRS letters, ∼20/80 or worse Snellen equivalent vs ≥55 ETDRS letters, ∼20/80 or better Snellen equivalent) at day 0, CNV type (minimally classic vs occult with no classic vs predominantly classic), and study center. The protocol mandated that patients receive sham injections or intravitreal injections of their assigned ranibizumab dose once a month for 3 months (day 0, month 1, month 2) and every 3 months thereafter (months 5, 8, 11, 14, 17, 20, and 23), for the duration of the 2-year study. Fluorescein angiography and fundus photography were performed at months 3, 5, 8, 12, and 24 and were evaluated by a central reading center (Fundus Photograph Reading Center, University of Wisconsin, Madison, Wisconsin, USA). Patients underwent complete ocular examination, including VA assessment at each study visit (ie, the first 3 months and then quarterly). The Vision Functioning Questionnaire-25 (VFQ-25) was administered at baseline and at months 3, 8, 12, and 24, prior to patients completing any other study-related procedures. In addition to injection visits, clinic visits were scheduled at months 3, 12, and 24. Subsequent protocol amendments (crossover and rollover amendments described below) increased subject assessments from quarterly to monthly. The monthly assessments were identical to the previous quarterly assessments.
The incidence and severity of ocular and nonocular adverse events (AEs) and changes in vital signs were assessed at all study visits. In accordance with the criteria established by the worldwide Antiplatelet Trialists’ Collaboration, arterial thromboembolic events (ATEs), such as vascular death, nonfatal myocardial infarction, nonfatal ischemic stroke, and nonfatal hemorrhagic stroke, were documented.
After careful review of 12-month data from the pivotal MARINA and ANCHOR trials, the study sponsor believed it to be in the best interest of sham group patients to be treated with ranibizumab. Thus, the protocol was amended on February 27, 2006 to provide sham-injection patients the opportunity to cross over to receive 0.5 mg ranibizumab quarterly after completing the month-12 visit (ie, the assessment time point for the primary analysis). Subsequently, after careful review of the 12-month PIER data, the protocol was amended again, on August 21, 2006, to provide all patients remaining in the study the opportunity to roll over to receive 0.5 mg ranibizumab monthly for the remainder of the 2-year study. No patients were unmasked to their original treatment assignment as a result of the crossover and rollover amendments.
The primary endpoint of PIER was mean change in best-corrected VA at month 12. Key visual outcomes at month 24 were mean change from baseline VA, proportion of patients who lost <15 VA letters from baseline, proportion of patients who gained ≥15 VA letters from baseline, proportion of patients with Snellen equivalent VA of 20/200 or worse, mean change from baseline VFQ-25 near and distance activities and vision-specific dependency subscale scores, mean change from baseline total area of CNV, and total area of CNV leakage plus retinal pigment epithelium (RPE) staining. Safety endpoints were incidence and severity of ocular and nonocular AEs, incidence of positive serum antibodies to ranibizumab, and changes in vital signs.
The intent-to-treat approach was used for visual and anatomic analyses and included all patients as randomized. Missing values were imputed using the last-observation-carried-forward method. All pairwise comparisons between the ranibizumab groups and the sham group were based on statistical models with 2 groups (ranibizumab vs sham) at a time. A type I error management plan was used to adjust for multiplicity of treatment comparisons and visual and anatomic endpoints. Unless otherwise noted, analyses were stratified by CNV type at baseline (minimally classic vs occult with no classic vs predominantly classic), as determined by the central reading center, and by baseline VA (≤54 vs ≥55 letters). For binary endpoints, stratified Cochran χ 2 tests were used for between-group comparisons of the proportion of patients meeting the endpoint. Analysis-of-variance and analysis-of-covariance models were used to analyze continuous endpoints.
The study sample size was based on the primary endpoint (ie, change from baseline best-corrected VA at month 12). The target sample size of 180 subjects (determined by clinical trial simulation) provided 90% power in the intent-to-treat analysis to detect a 9-letter difference between 1 or both ranibizumab dose groups and the sham-injection group in mean change from baseline VA at month 12, according to the Hochberg-Bonferroni criterion (assumptions based on results of the TAP and VIP trials and anticipated proportions of each CNV type). Safety analyses were performed using descriptive statistics and included all treated patients. All analyses were performed with SAS software (SAS Institute Inc, Cary, North Carolina, USA).
Results
Between September 7, 2004 and March 16, 2005, 184 patients were randomized 1:1:1 to receive sham injection (n = 63), 0.3 mg ranibizumab (n = 60), or 0.5 mg ranibizumab (n = 61) at 43 US investigative sites. Baseline demographic and ocular characteristics were similar across treatment groups ( Table 1 ). Groups were predominantly white and nearly two-thirds female, with a mean age of 78 years. Mean baseline VA was 53 to 56 letters (Snellen equivalent ∼20/63 to 20/80) across groups.
| Sham (n = 63) | 0.3 mg (n = 60) | 0.5 mg (n = 61) | |
|---|---|---|---|
| Gender | |||
| Male | 20 (31.7) | 26 (43.3) | 28 (45.9) |
| Female | 43 (68.3) | 34 (56.7) | 33 (54.1) |
| Race/ethnicity | |||
| White | 59 (93.7) | 57 (95.0) | 56 (91.8) |
| Other | 4 (6.3) | 3 (5.0) | 5 (8.2) |
| Age, years | |||
| Mean (SD) | 77.8 (7.1) | 78.7 (6.3) | 78.8 (7.9) |
| Range | 59–92 | 60–93 | 54–94 |
| Age group, years | |||
| 50–<65 | 4 (6.3) | 1 (1.7) | 4 (6.6) |
| 65–<75 | 12 (19.0) | 12 (20.0) | 12 (19.7) |
| 75–<85 | 36 (57.1) | 37 (61.7) | 31 (50.8) |
| ≥85 | 11 (17.5) | 10 (16.7) | 14 (23.0) |
| Prior therapy for AMD | |||
| Any | 35 (55.6) | 35 (58.3) | 33 (54.1) |
| Laser photocoagulation | 3 (4.8) | 5 (8.3) | 7 (11.5) |
| Medication | 1 (1.6) | 1 (1.7) | 2 (3.3) |
| Supplements | 34 (54.0) | 33 (55.0) | 28 (45.9) |
| Years since first diagnosis of neovascular AMD | |||
| n | 62 | 59 | 61 |
| Mean (SD) | 0.3 (0.5) | 0.7 (1.6) | 0.7 (1.2) |
| Range | 0.0–3.0 | 0.0–9.1 | 0.0–5.0 |
| Visual acuity (ETDRS letters) | |||
| n | 63 | 60 | 61 |
| Mean (SD) | 55.1 (13.9) | 55.8 (12.2) | 53.7 (15.5) |
| Range | 25–76 | 18–79 | 13–79 |
| ≤ 54, 20/80 | 25 (39.7) | 29 (48.3) | 27 (44.3) |
| ≥ 55, 20/80 | 38 (60.3) | 31 (51.7) | 34 (55.7) |
| Visual acuity (approximate Snellen equivalent) | |||
| Median | 20/63 | 20/63 | 20/80 |
| 20/200 or worse | 10 (15.9) | 3 (5.0) | 10 (16.4) |
| Better than 20/200 but worse than 20/40 | 42 (66.6) | 49 (81.6) | 36 (58.9) |
| 20/40 or better | 11 (17.5) | 8 (13.3) | 15 (24.6) |
| CNV lesion subtype | |||
| n | 63 | 60 | 61 |
| Predominantly classic | 13 (20.6) | 8 (13.3) | 12 (19.7) |
| Minimally classic | 30 (47.6) | 22 (36.7) | 19 (31.1) |
| Occult without classic | 20 (31.7) | 29 (48.3) | 30 (49.2) |
| Not classified | 0 | 1 (1.7) | 0 |
| Total area of lesion (DA) | |||
| n | 63 | 59 | 61 |
| Mean (SD) | 4.34 (3.23) | 4.36 (3.27) | 4.04 (2.61) |
| Range | 0.1–17.0 | 0.1–20.3 | 0.05–10.0 |
| ≤4 DA | 32 (50.8) | 32 (54.2) | 31 (50.8) |
| >4 DA | 31(49.2) | 27 (45.8) | 30 (49.2) |
| Total area of CNV (DA) | |||
| n | 63 | 59 | 61 |
| Mean (SD) | 3.61 (3.23) | 3.77 (3.40) | 3.29 (2.27) |
| Range | 0.02–17.0 | 0.0–20.3 | 0.03–9.6 |
| Leakage from CNV plus RPE staining (DA) | |||
| Mean (SD) | 4.25 (3.55) | 4.47 (3.56) | 3.99 (2.61) |
| Range | 0.20–19.0 | 0.0–22.5 | 0.50–9.70 |
The first diagnosis of neovascular AMD was within the previous year for 87% of patients. Overall, 80% of patients had either occult with no classic or minimally classic CNV lesions, but occult with no classic CNV was more common in the ranibizumab groups than in the sham injection group (nearly 50% vs <33% of study eye lesions, respectively). Nearly 50% of the study eyes in each group had lesions ≥4 DA. The mean total area of CNV lesion and CNV leakage plus RPE staining at baseline was similar across groups.
Forty-six of 63 (73%), 53 of 60 (88.3%), and 54 of 61 (88.5%) patients randomized to the sham-injection, 0.3 mg, and 0.5 mg groups, respectively, completed the study through month 24 ( Table 2 ). By month 24, 48 of 184 (26.1%) patients had discontinued treatment (25 of 184 [13.6%] at month 12), usually because the patient’s condition mandated other therapeutic intervention.
| Ranibizumab | |||
|---|---|---|---|
| Sham (n = 63) | 0.3 mg (n = 60) | 0.5 mg (n = 61) | |
| Received assigned treatment | 62 (98.4) | 59 (98.3) | 61 (100.0) |
| Completed study | 46 (73.0) | 53 (88.3) | 54 (88.5) |
| Discontinued from study | 17 (27.0) | 7 (11.7) | 7 (11.5) |
| Patient’s decision | 8 (12.7) | 1 (1.7) | 4 (6.6) |
| Patient noncompliance | 1 (1.6) | 2 (3.3) | 1 (1.6) |
| Patient’s condition mandated other therapeutic intervention | 3 (4.8) | 0 | 0 |
| Discontinued treatment | 27 (42.9) | 11 (18.3) | 10 (16.4) |
| Adverse event | 6 (9.5) | 4 (6.7) | 4 (6.6) |
| Patient’s decision | 7 (11.1) | 4 (6.7) | 4 (6.6) |
| Physician’s decision | 2 (3.2) | 1 (1.7) | 1 (1.6) |
| Patient’s condition mandated other therapeutic intervention | 12 (19.0) | 2 (3.3) | 1 (1.6) |
| Eligible to participate in crossover | 40 (63.5) | — | — |
| Crossed over and received 0.5 mg ranibizumab | 39 (61.9) | — | — |
| Visit at which patient crossed over to quarterly 0.5 mg ranibizumab | — | ||
| Month 14 | 15 (38.5) | — | — |
| Month 17 | 17 (43.6) | — | — |
| Month 20 | 7 (17.9) | — | — |
| Mean (SD) duration of crossover treatment, days | 188.3 (75.5) | ||
| Eligible to participate in rollover | 35 (55.6) | 43 (71.7) | 44 (72.1) |
| Participated in rollover amendment | 34 (54.0) | 43 (71.7) | 44 (72.1) |
| Visit at which patient rolled over to monthly 0.5 mg ranibizumab | |||
| Month 19 | 3 (8.8) | 3 (7.0) | 3 (6.8) |
| Month 20 | 14 (41.2) | 14 (32.6) | 16 (36.4) |
| Month 21 | 2 (5.9) | 6 (14.0) | 4 (9.1) |
| Month 22 | 3 (8.8) | 7 (16.3) | 5 (11.4) |
| Month 23 | 12 (35.3) | 13 (30.2) | 16 (36.4) |
| Mean (SD) number of rollover injections | 2.6 (1.5) | 2.6 (1.3) | 2.5 (1.3) |
| Randomized patients (intent-to-treat efficacy analysis) | 63 (100.0) | 60 (100.0) | 61 (100.0) |
At the time of the February 2006 crossover amendment, 40 of 63 (63.5%) patients in the sham-injection group who had not discontinued study treatment were eligible to cross over to receive 0.5 mg ranibizumab quarterly, and 39 (61.9%) of those received at least 1 intravitreal injection, beginning at month 14. At the time of the August 2006 rollover amendment 34 of 63 (54.0%), 43 of 60 (71.7%), and 44 of 61 (72.1%) patients in the sham-injection, 0.3 mg, and 0.5 mg groups, respectively, who had not discontinued study treatment or completed the month-24 visit, rolled over to receive 0.5 mg ranibizumab monthly, beginning month 19. Results are presented according to group assignment at randomization and include post-crossover (sham) and post-rollover (sham, 0.3 mg, 0.5 mg) data.
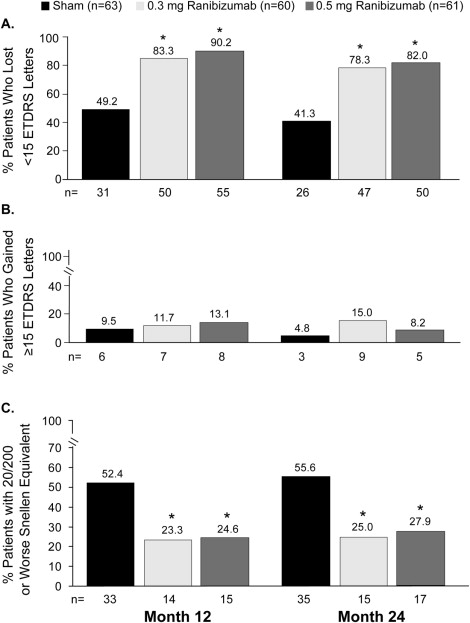
At month 24, VA had decreased from baseline an average of 21.4 letters in the sham group, 2.2 letters in the 0.3 mg group, and 2.3 letters in the 0.5 mg group ( P < .0001 each ranibizumab dose vs sham), with about a 19-letter difference between sham-group and treated patients. The group differences at month 24 were similar to those at month 12 ( Table 3 ). At month 24, 47 of 60 (78.2%) patients in the 0.3 mg group and 50 of 61 (82.0%) of patients in the 0.5 mg group had lost <15 letters from baseline VA compared with 26 of 63 (41.3%) sham-injection patients ( P < .0001 each ranibizumab dose vs sham) ( Figure 2 ); and 21 of 63 (33.3%) patients in the sham group had lost ≥30 VA letters from baseline. Such severe vision loss was uncommon (∼3.0%) in patients who were originally randomized to ranibizumab treatment groups. Ranibizumab groups did not differ significantly from the sham group in the proportion of patients who gained ≥15 VA letters: 3 of 63 (4.8%) in the sham-injection group, 9 of 60 (15.0%) in the 0.3 mg group, and 5 of 61 (8.2%) in the 0.5 mg group.
| Ranibizumab | |||
|---|---|---|---|
| ETDRS Letters | Sham (n = 63) | 0.3 mg (n = 60) | 0.5 mg (n = 61) |
| Month 12 | |||
| Mean (SD) | −16.3 (22.3) | −1.6 (15.1) | −0.2 (13.1) |
| 95% CI a | −21.9 to −10.7 | −5.4 to 2.3 | −3.5 to 3.2 |
| P value (vs sham) b | .0001 | <.0001 | |
| Month 24 | |||
| Mean (SD) | −21.4 (21.8) | −2.2 (15.6) | −2.3 (14.4) |
| 95% CI a | −26.8 to −15.9 | −6.3 to 1.8 | −6.0 to 1.4 |
| P value (vs sham) b | <.0001 | <.0001 | |
a Derived from t distribution.
b Based on pairwise analyses of variance adjusted for stratification of baseline choroidal neovascularization classification (minimally classic vs occult without classic vs predominantly classic) and baseline visual acuity (≤54 vs ≥55 letters).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


