Authors discuss laryngeal lesions, metastases, and relevant anatomy. Outcome of surgical and radiotherapy in terms of voice preservation is discussed. Radiation techniques and outcomes for laryngeal cancer are presented along with discussion of interdisciplinary treatment. Authors review studies and quality of life outcomes of surviving laryngeal cancer patients.
Laryngeal cancer is the most common head and neck cancer, affecting approximately 10,000 Americans per year and accounting for almost one third of all head and neck malignancies . Radiotherapy (RT) has a long and well-established history in the multidisciplinary management of laryngeal cancer, be it as a definitive local treatment for early lesions, or in combination with surgery and chemotherapy for locally advanced disease. In general, surgery and radiation are equally effective for early stage lesions, whereas more advanced stages require a multimodality approach using a combination of surgery and radiation or chemotherapy and radiation. Given the important role that the larynx plays in human speech and communication, most contemporary treatment paradigms are geared toward optimizing cure rates while preserving organ function. In addition to stage and site of disease (supraglottic versus glottic versus subglottic), treatment modality and technique also depend on a host of other factors such as age, performance status, and treatment-related side effects. A multidisciplinary approach is, therefore, essential in incorporating all of these considerations into a cohesive and effective treatment plan. This article reviews the indications for radiotherapy for cancer of the larynx and summarizes some of the important data supporting its use.
Anatomy and patterns of spread
The larynx is divided into three anatomic regions: supraglottic, glottic, and subglottic. The supraglottic larynx is composed of the supra and infrahyoid epiglottis, the aryepiglottic folds, the arytenoids, and the false vocal cords. The glottic larynx encompasses the true vocal cords (including anterior and posterior commissures), and the subglottic larynx extends from the lower edge of the glottis to the inferior aspect of the cricoid cartilage. The thyroid cartilage encloses the larynx anteriorly and laterally. One common pattern of spread in laryngeal carcinoma is local extension into the surrounding structures. Two important membranous barriers to local tumor spread are the quadrangular membrane, which separates the laryngeal vestibule from the hypopharynx and the conus elasticus, which is made up of fibers spanning from the cricoid inferiorly to the vocal ligament and arytenoid cartilage superiorly. In addition, the epiglottic cartilage has numerous dehiscences that facilitate tumor extension into the pre-epiglottic space, which is a fat-filled horseshoe-shaped space extending anteriorly and posterolaterally from the epiglottic cartilage. Yet another area that is vulnerable to local tumor spread is the space between the true and false vocal cords or the so-called laryngeal ventricle (or sinus of Morgagni), as this structure is not protected by the quadrangular membrane.
The most common type of cancer of the larynx is squamous cell cancer, ranging from carcinoma in situ to poorly differentiated carcinoma. Much rarer cell types include verrucous, adenoidcystic, and neuroendocrine carcinomas. Laryngeal cancer is three times more likely to arise in the glottis than the supraglottis, while subglottic cancer is extremely rare, accounting for no more than 2% of all laryngeal cancers . The true vocal cords are on average 2 cm long and are thinnest anteriorly and posteriorly where they insert into the thyroid cartilage and the vocal processes of the arytenoids, respectively . Most lesions arise on the free edge and upper surface of the anterior two thirds of the vocal cords, frequently extending to the anterior commissure. These tumors typically grow very slowly and remain confined to the mucosa of the true vocal cords. Larger or more infiltrative glottic tumors can invade the paraglottic and pre-epiglottic spaces, which offer little resistance to further spread through the ligaments and membranes around the thyroid cartilage or through the thyroid cartilage itself.
Local spread
In contrast to glottic lesions, supraglottic tumors have varying local extension patterns depending on which anatomic subsite they originate from. For example, suprahyoid lesions are often exophytic and can grow to a relatively large size without causing many symptoms. One important consideration is that the marked regression of these lesions after chemoradiation can compromise the anatomic integrity of the epiglottis, thus increasing the risk for aspiration. In contrast, infrahyoid lesions tend to grow anteriorly or circumferentially, whereas lesions from the aryepiglottic folds can extend in virtually all directions. Subglottic lesions spread either inferiorly to the trachea and into the neck or to the glottis superiorly. The location and degree of locoregional spread can often account for the nature and severity of symptoms at presentation, such as hoarseness, sore throat, shortness of breath, dysphagia, and throat “fullness.” For example, patients with supraglottic cancers, which often grow to more advanced stages, can present with sore throat, referred ear pain (via the vagus and the auricular nerve of Arnold), or enlarged neck nodes.
Nodal metastases
In contrast to the supraglottis, the glottis has virtually no lymphatic drainage and, therefore, there is a very small risk of lymph node metastasis with T1 or small T2 lesions ( Table 1 ). The incidence of lymphadenopathy at diagnosis is about 5% for early glottic lesions and 20% for the more extensive T3/4 tumors . Additionally, according to Byers and colleagues , the rate of occult nodal involvement is approximately 16% in patients with T3/4, N0 disease at the time of elective nodal dissection, with the most frequently involved nodes being the upper and mid-jugular nodes. As smaller tumors grow, however, they begin to involve the supraglottic or subglottic structures, which have more extensive lymphatic networks, thus significantly increasing the risk of nodal involvement. For example, older studies have shown that 55% of patients with supraglottic cancers have clinically involved lymph nodes and that up to one half of the remaining clinically node-negative patients have pathologic nodal involvement on further evaluation . Subglottic tumors have a similarly high rate of lymph node involvement, but in contrast to supraglottic cancers, which tend to preferentially involve the upper and mid-jugular nodes, they tend to spread to the lower jugular, pretracheal, and prelaryngeal (Delphian) nodes first. Based on these and other studies, it is generally accepted that patients with T3/4 glottic as well as patients with supraglottic and subglottic tumors merit comprehensive treatment of the nodal chains in addition to the primary tumor.
| Tis: Carcinoma in situ | |
| T1: Supraglottis: tumor limited to one subsite of supraglottis with normal cord mobility | |
| Glottis: T1a, tumor limited to one vocal cord or T1b, tumor limited to both vocal cords with normal cord mobility | |
| Subglottis: tumor limited to subglottis | |
| T2: Supraglottis: tumor invades more than one supraglottic subsite or glottis or region outside supraglottis without larynx fixation | |
| Glottis: tumor extends to supra- or subglottis and/or with impaired cord mobility | |
| Subglottis: tumor extends to glottis with normal or impaired cord mobility | |
| T3: Supraglottis: tumor limited to larynx with cord fixation or invades laryngeal structures and/or erodes inner cortex of thyroid cartilage | |
| Glottis: tumor limited to larynx with cord fixation and/or invades paraglottic space and/or erodes inner cortex of thyroid cartilage | |
| Subglottis: tumor limited to larynx with cord fixation | |
| T4: T4a, tumor invades through thyroid cartilage or invades cricoid (for subglottis) and/or tissues beyond larynx (trachea, thyroid, deep tongue muscles, esophagus) | |
| T4b, tumor invades prevertebral space, encases carotid artery, or invades mediastinal structures | |
| N1: Single ipsilateral node < 3 cm | MX: distant metastasis cannot be assessed |
| N2: a, single ipsilateral node 3–6 cm | M0: No distant metastasis |
| b, multiple ipsilateral nodes < 6 cm | M1: Distant metastasis |
| c, bilateral or contralateral nodes < 6 cm | |
| N3: node(s) > 6 cm | |
Distant metastases
In addition to direct extension and nodal involvement, laryngeal cancer can occasionally spread distantly, usually to the lungs and/or bones. Early studies have shown a rather low (less than 10%) overall incidence of distant metastases from laryngeal carcinoma, with supraglottic and subglottic cancers accounting for most cases. For example, Marks and colleagues found that 23% of patients with pyriform sinus cancers develop metastatic disease, whereas only 1% of patients with glottic lesions fail distantly once their neck disease is controlled . In addition, other investigators have reported distant metastasis rates approaching 15% in patients with primary subglottic squamous cell carcinomas . A recent study from the Netherlands also examined risk factors associated with metastatic disease and concluded that lymph node involvement increases the risk for distant metastases three- to fourfold, whereas lymph node involvement with extracapsular extension increases this risk by a factor of 10 . All of these distinctions are becoming increasingly important as refinements in surgical and chemoradiation approaches continue to affect better local control rates, thus necessitating new and more effective approaches of preventing and managing distant failures.
Radiotherapy for early glottic carcinoma
Both radiotherapy and voice-preserving surgery (excision, cordectomy, or hemilaryngectomy) are accepted treatment modalities for early (Tis, T1/T2 N0) glottic lesions. Even though large individual series have demonstrated comparable local control rates, there has been no direct comparison of these two treatment modalities in a prospective trial. A recent Cochrane review tried to compare the effectiveness of radiotherapy and surgery in early laryngeal cancer but concluded ultimately that there is not enough evidence to establish a clear superiority . Additionally, there are no randomized data on functional outcomes after either treatment to help guide clinicians in their recommendations. In general, the choice of therapy is dictated by both the need for tumor control and the desire to preserve phonation after therapy, which in turn depend on a variety of factors, such as tumor location, volume of disease, extent of invasion, anterior commissure involvement, and nodal status. In addition, socioeconomic considerations, such as patient age, occupation, and likelihood of compliance can also inform the physician as to what treatment method may be most appropriate for a particular patient.
Carcinoma in situ (Tis)
The management of carcinoma in situ (Tis) of the glottis is controversial, with treatment practices ranging from primary radiation or surgery (vocal cord stripping, microexcision, cordectomy, or partial laryngectomy) to observation after biopsy. A few studies on the latter approach, however, have documented high rates of progression to invasive squamous cell carcinoma that is often no longer amenable to voice-preserving therapy . In addition, some series have shown that, due to limited biopsy sampling, many patients with Tis actually harbor invasive disease . Therefore, most physicians advocate treatment, either with minimally invasive surgery or radiation. Complete stripping of the mucosa of the cord is often curative for early dysplastic lesions (leukoplakia, erythroplakia, hyperkeratosis); however, the disadvantage with this method is that patients need careful observation and frequently require repeat procedures that may eventually scar the cords, thus obscuring new lesions and leading to hoarseness. Furthermore, many radiation oncologists believe that surgical techniques tend to be more operator-dependent than the well-established approaches and treatment paradigms with radiation, and that, unless patients are undergoing these procedures at high volume head and neck cancer centers, they should preferentially be steered toward radiation. These considerations favor RT, which has traditionally conferred excellent local control rates in the range of 85% to 100% ( Table 2 ). In addition, surgical salvage rates after recurrence are excellent, ranging from 90% to 100%, whereas the salvage potential of radiation after surgery in early glottic lesions has not been as extensively investigated. The main disadvantages of radiotherapy are the acute side effects from treatment and the length of treatment. Also, some critics point out that most patients salvaged with surgery after recurrence require total laryngectomy and that initial treatment with surgery tends to be more cost effective .
| Study | N | 5-year local control, % |
|---|---|---|
| Tis | ||
| Wang | 60 | 92 |
| Spayne, et al | 67 | 98 |
| Garcia-Serra, et al | 30 | 88 |
| Cellai, et al | 89 | 84 |
| T1 | ||
| Wang | 665 | 93 |
| Warde, et al | 449 | 91 (T1a) |
| 82 (T1b) | ||
| Mendenhall, et al | 291 | 94 (T1a) |
| 93 (T1b) | ||
| Cellai, et al | 831 | 84 |
| T2 | ||
| Wang | 237 | 71–77 |
| Warde, et al | 230 | 69 |
| Frata, et al | 256 | 73 |
T1 glottic lesions
Similar concerns regarding the advantages and disadvantages of surgery versus radiation stimulate the debate on the most appropriate treatment for T1 glottic lesions. In general, these lesions can also be treated very effectively with either surgery or radiation. For example, in one of the largest surgical series reported to date, the 3-year local control rates for patients with T1a and T2b lesions undergoing laser cordectomy were 94% and 91%, respectively . In another large study using radiation as the treatment of choice, the 5-year local control rates were comparable at 94% for T1a and 93% for T1b tumors . Because T1 lesions can be so diverse in terms of presentation, many clinicians base their treatment recommendations on ancillary criteria, such as lesion size, shape, location, and anterior commissure involvement. Some of these criteria, such as involvement of the anterior commissure, are thought to be poor prognostic factors that negatively impact the likelihood for local control and voice preservation after therapy . The largest retrospective study on radiotherapy outcomes in T1 glottic cancer, recently published by Cellai and colleagues from Italy, lends further support to these findings. A total of 831 patients with T1N0 glottic tumors were analyzed and a 5-year local control rate of 84% was reported (see Table 2 ). This compares favorably with local control rates from other published series, but more importantly, in this study lesion extent and anterior commissure involvement were shown to predict in a statistically significant way for local or locoregional failure on both uni- and multivariate analysis. Additionally, some investigators have reported significantly lower actuarial local control rates after radiotherapy for bulky versus small T1 lesions, lending some credence to the approach of laser excision followed by RT for more extensive T1 tumors . In general, however, either RT or surgery alone is used and both are given equal consideration if the lesion is small, limited to one vocal cord (T1a), and if there is no extension to the arytenoids or the anterior commissure. However, occasionally endoscopic laser resection is recommended over RT for the well-defined superficial lesion located in the middle third of the vocal cord, particularly when it is along the cord’s free edge, as RT takes much longer to administer. This recommendation is also supported by studies that have shown very similar voice quality measures in patients with mid-cord tumors undergoing limited excision as well as those receiving definitive RT for such early glottic lesions . In contrast, because the surgical management of larger (especially T1b) lesions requires the removal of greater amounts of tissue with an increasing potential for poor voice quality as a result, radiation is usually the preferred initial treatment for those tumors.
Even though many clinicians take into consideration the potential for dysphonia after either mode of therapy, there are really very few good studies comparing voice quality after radiation versus laser excision. In fact, most of them are retrospective single institution series that when taken together yield conflicting impressions. On the one hand, Hirano and colleagues concluded in the 1980s that hoarseness and poor approximation of the vocal cords were more frequently observed in patients treated with laser excision. Similarly, data from our institution suggested a few years later that a higher proportion of patients treated with radiation for T1 glottic carcinoma retain normal to near-normal voice quality when compared with patients treated with laser excision . On the other hand, however, more recent evidence seems to contradict these findings, perhaps reflecting recent improvements in endoscopic microsurgical techniques. A recent study from Canada, for example, suggests that even though vocal cord function (as assessed by videostroboscopy) was superior in the patients who received RT, the patients who underwent endoscopic resection of their tumors actually scored higher on subjective and objective voice assessments (Voice Handicap Index questionnaire and Visipitch parameters) . In addition, a recent meta-analysis of almost 300 patients with T1 glottic tumors treated either with laser excision or radiotherapy concluded that both treatment methods result in comparable levels of voice handicap .
Factors affecting voice quality
Other investigators have attempted to single out specific patient- or treatment-related characteristics that predict for poor voice quality after treatment. For instance, McGuirt and colleagues observed that the greater the amount of vocalis muscle resected during laser excision, the greater the decline of voice quality posttreatment . As far as radiation is concerned, continued heavy tobacco exposure during RT and extensive vocal cord stripping or multiple biopsies before RT have been postulated by some to adversely affect postirradiation voice quality . Others still have suggested that voice overuse during radiation may also degrade one’s voice after treatment . In summary, it is very challenging to discern any significant benefit of one treatment approach versus another in patients with Tis or T1 lesions of the glottis. RT continues to be one standard approach in treating these patients. Endoscopic laser excision can provide comparable local control rates and posttreatment voice quality in selected patients and has the main advantage of brevity at a potentially lower cost, being in most cases a single outpatient procedure.
T2 glottic lesions
Given that T2 lesions are by definition larger, often extending beyond the glottis or causing some impairment of vocal cord mobility, it is not surprising that these tumors are somewhat more difficult to control with either surgery or radiation. Local control rates with radiation alone range from 65% to 80%, ultimately reaching 70% to 90% after surgical salvage (see Table 2 ). As far as surgery is concerned, both a vertical partial laryngectomy or a partial supracricoid laryngectomy are acceptable options for bulkier and more invasive T2 lesions, especially since local control rates are excellent and preservation of vocal cord function is somewhat less of an issue once phonation has been compromised by tumor invasion. Endoscopic resection is also an option but only in selected patients with T2 glottic tumors that are small, superficial, and with normal vocal cord mobility. Nonetheless, radiation alone is still the preferred treatment approach at many institutions due to somewhat better functional outcomes. Some investigators have even shown improved rates of disease control (92% versus 72%, 5-year disease-free survival) and better voice preservation rates (89% versus 61%) for T2N0 tumors treated with concurrent chemoradiation; however, given the low incidence of distant dissemination in these patients and the potential for greater toxicity, this approach should not be routinely recommended without further validation .
Loco-regional control after treatment for T2 glottic cancer
The wide range in local control rates for T2 glottic cancer is, in part, due to the heterogeneity of presentations within this stage of disease. The second largest experience to date on T2 lesions treated with RT alone was recently reported by Frata and colleagues who attempted to single out features associated with a poor outcome. Over 250 cases were analyzed retrospectively and a cumulative local control probability of 73% was reported at 5 years. The main determinants of inferior local control on both univariate and multivariate analysis in this study were tumor extent and impaired cord mobility. Other investigators have looked for prognostic factors in this group of patients as well and have found very similar results. For example, patients with T2 lesions treated with radiation at the Massachusetts General Hospital were found to have a slightly better local control at 5 years if they did not have impaired vocal cord mobility (77% versus 71%) . In contrast, studies from the M.D. Anderson Cancer Center did not show a clear relationship between cord mobility and outcomes after RT . Furthermore, the influence of impaired cord mobility on surgical salvage rates after radiation is far from clear and anterior commissure involvement has failed to emerge as an independent prognostic factor in this group of patients. If anything, many investigators believe nowadays that it is simply a marker of more extensive disease, which is the actual and more significant determinant of outcome . What has more clearly emerged as a predictor of local control in T2 glottic cancer is the way that radiation is delivered, namely over what period of time and to what total dose. For example, investigators from Japan recently reiterated that treatment duration of 54 days or less and RT doses 67 Gy or more correlated with significantly better 5-year local control rates (87% versus 71%, P = .023 and 91% versus 60%, P = .0013) .
Technical considerations
Tis, T1 lesions
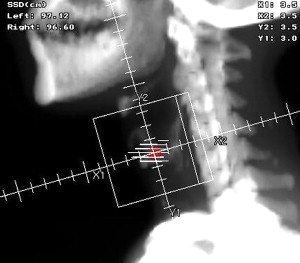
As mentioned earlier, the radiation techniques for Tis and T1 lesions are pretty standard and setup is quite simple. Patients are usually treated with two 5 × 5-cm opposed lateral fields that cover the glottic larynx with a 1- to 2-cm margin. The superior border of the field is placed at the top of the thyroid cartilage and the inferior border is extended to the bottom of the cricoid. Anteriorly the field extends 1 to 2 cm past the skin surface and posteriorly it stops at the anterior edge of the cervical vertebral bodies ( Fig. 1 ). Occasionally, the fields are turned (collimated) slightly to match the pre-vertebral line posteriorly and tilted inferiorly by 5 to 10 degrees to avoid the shoulders, especially in patients with short necks. Both Cobalt-60 and 4- to 6-megavoltage (MV) photons can provide adequate coverage of the glottis, whereas higher energy beams are generally avoided due to a greater risk of underdosing the anterior commissure. This consideration arises from a phenomenon called dose-buildup, which essentially means that higher energy beams deposit most of their energy deeper into tissues while sparing/underdosing superficial structures. Even with 6 MV there is some risk of underdosing anterior tumors, especially in very thin patients, thus necessitating the use of tissue-equivalent bolus material to allow for adequate build-up. Bolus is a general term used in radiation oncology that defines various ways of simulating additional tissue in the patient so that the dose can be effectively modified to cover more superficial structures (ie, skin, subcutaneous structures). Computed tomography (CT)-based planning is generally recommended to better assess dose distribution and to determine if wedges should be used. Wedges are beam modifiers that when placed in front of the beam allow for a varying dose distribution in tissue depending on the angle of the wedges employed. In laryngeal cancer, two 15-degree wedges are usually used to improve homogeneity and dose distribution to the mid and posterior portions of the vocal cords. Treating without wedges, on the other hand, is recommended for anterior lesions, as small hot spots in areas of disease are generally acceptable.
T2 lesions
The fields for T2 lesions are generally slightly larger to account for the greater extent of disease and the potential for supra- or subglottic involvement. They usually measure 6 × 6 cm and encompass at least one tracheal ring below the cricoid, especially if there is subglottic extension. Posteriorly, the field covers the anterior one third to one half of the cervical vertebral bodies. At our institution we also extend the superior border to 2 cm above the angle of the mandible to cover the upper jugular nodes, particularly when there is clear evidence of supraglottic extension ( Fig. 2 ). In addition, some radiation oncologists bring in the posterior edges of their T1 and T2 fields by 1 and 1.5 cm, respectively, for the last few fractions as a way of reducing some of the dose to the pharynx. However, caution must be exercised with this technique so as not to underdose lesions that extend to the posterior glottis.
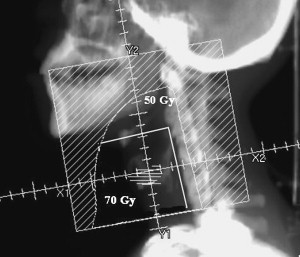
Dose of radiation
Radiation doses have traditionally ranged from 60 to 70 Gy administered in 2-Gy fractions. This approach is based on results from various studies demonstrating superior local control with fraction doses larger than 1.8 Gy ( Table 3 ). Usually, a dose of 60 Gy is administered after surgery when there is no evidence of gross disease. For intact Tis and T1 lesions, a dose of 66 Gy has been employed, whereas T2 lesions have traditionally received 70 Gy. More recently, however, several studies, including a prospective randomized study from Japan, have convincingly shown that fraction doses greater than 2 Gy are even more effective in controlling early glottic cancer than conventional fraction sizes without much added toxicity (see Table 3 ). For example, the Japanese study by Yamazaki and colleagues demonstrated a 5-year local control rate of 92% for patients with T1 lesions receiving 2.25 Gy per fraction (total 56.25 to 63 Gy) and 77% for those receiving 2 Gy per fraction (total 60 to 66 Gy). This difference in local control was statistically significant with a P value of .004. As a result of this and other studies, some centers now irradiate with fractions of 2.25 Gy, administering total doses of 56.25 to 58.5 Gy for Tis, 63 Gy for T1N0, and 65.25 Gy for T2N0 lesions.
| Study | n | Stage | Radiation, Gy (dose/fraction) | 5-year local control, % | P value | |
|---|---|---|---|---|---|---|
| Schwaibold, et al | 56 | T1 | 1.8 | 75 | <.01 | |
| 2 | 100 | |||||
| Le, et al | 398 | T1, T2 | <1.8 | 79 (T1) | 44 (T2) | NS for T1 |
| ≥1.8–2.24 | 81–92 (T1) | 73–79 (T2) | .003 for T2 | |||
| >2.25 | 94 (T1) | 100 (T2) | ||||
| Burke, et al | 100 | T1, T2 | <2 | 44 | <.01 | |
| ≥2 | 92 | |||||
| Garden, et al | 146 | T2 | 2 | 59 | <.001 | |
| 2.06–2.26 | 80 | |||||
| Yamazaki, et al | 180 | T1 | 2 | 77 | .004 | |
| T1 | 2.25 | 92 | ||||
Nontraditional fractionation
Hyperfractionation and accelerated fractionation schemes have also been extensively investigated in both early and more advanced glottic cancer. Garden and co-investigators reported on 230 patients with T2 glottic cancer, more than a third of whom were treated with twice-daily fractionation. The 5-year local control rate was 79% for patients treated with the twice-daily regimen and 67% for those receiving once-daily treatments . The largest retrospective series from the Massachusetts General Hospital also supports the use of hyperfractionation, especially in patients with T3 lesions (5-year local control 67% versus 42%, P = .03) . A more recent study from Japan suggests that hyperfractionation, when compared with conventional RT dosing in T2 glottic cancer, may also improve the 5-year laryngeal preservation rate (95.3% versus 52%–60%, P < .05) . In addition, hyperfractionation and accelerated fractionation schemes have also been shown to improve outcomes after radiotherapy for head and neck cancer in a few randomized trials . For example, two of the more recent trials, Radiation Therapy Oncology (RTOG) 90-03 and the Continuous Hyperfractionated Accelerated Radiotherapy (CHART) trials, demonstrated a benefit to altered fractionation for locally advanced squamous cell cancers of the head and neck but did so at the expense of added toxicity. In the RTOG trial, concomitant boost and hyperfractionated RT improved 2-year local control, disease-free survival, and overall survival when compared with standard or split-course accelerated regiments . In the CHART trial, investigators compared traditional radiotherapy to a continuous regimen over 12 successive days for a total dose of 54 Gy. Even though overall disease-free survival was similar in both groups, for T3 and T4 laryngeal tumors the 3 year local control rates were significantly better with the continuous course of therapy than with conventional fractionation (70% versus 47% and 78% versus 38% respectively) . RTOG 95-12, which randomized patients with T2 glottic tumors to a standard course of 70 Gy in 2 Gy per fraction or to 79.2 Gy in twice-daily 1.2-Gy fractions recently completed accrual and will hopefully add more to our knowledge of hyperfractionation in head and neck cancer.
Radiotherapy for early glottic carcinoma
Both radiotherapy and voice-preserving surgery (excision, cordectomy, or hemilaryngectomy) are accepted treatment modalities for early (Tis, T1/T2 N0) glottic lesions. Even though large individual series have demonstrated comparable local control rates, there has been no direct comparison of these two treatment modalities in a prospective trial. A recent Cochrane review tried to compare the effectiveness of radiotherapy and surgery in early laryngeal cancer but concluded ultimately that there is not enough evidence to establish a clear superiority . Additionally, there are no randomized data on functional outcomes after either treatment to help guide clinicians in their recommendations. In general, the choice of therapy is dictated by both the need for tumor control and the desire to preserve phonation after therapy, which in turn depend on a variety of factors, such as tumor location, volume of disease, extent of invasion, anterior commissure involvement, and nodal status. In addition, socioeconomic considerations, such as patient age, occupation, and likelihood of compliance can also inform the physician as to what treatment method may be most appropriate for a particular patient.
Carcinoma in situ (Tis)
The management of carcinoma in situ (Tis) of the glottis is controversial, with treatment practices ranging from primary radiation or surgery (vocal cord stripping, microexcision, cordectomy, or partial laryngectomy) to observation after biopsy. A few studies on the latter approach, however, have documented high rates of progression to invasive squamous cell carcinoma that is often no longer amenable to voice-preserving therapy . In addition, some series have shown that, due to limited biopsy sampling, many patients with Tis actually harbor invasive disease . Therefore, most physicians advocate treatment, either with minimally invasive surgery or radiation. Complete stripping of the mucosa of the cord is often curative for early dysplastic lesions (leukoplakia, erythroplakia, hyperkeratosis); however, the disadvantage with this method is that patients need careful observation and frequently require repeat procedures that may eventually scar the cords, thus obscuring new lesions and leading to hoarseness. Furthermore, many radiation oncologists believe that surgical techniques tend to be more operator-dependent than the well-established approaches and treatment paradigms with radiation, and that, unless patients are undergoing these procedures at high volume head and neck cancer centers, they should preferentially be steered toward radiation. These considerations favor RT, which has traditionally conferred excellent local control rates in the range of 85% to 100% ( Table 2 ). In addition, surgical salvage rates after recurrence are excellent, ranging from 90% to 100%, whereas the salvage potential of radiation after surgery in early glottic lesions has not been as extensively investigated. The main disadvantages of radiotherapy are the acute side effects from treatment and the length of treatment. Also, some critics point out that most patients salvaged with surgery after recurrence require total laryngectomy and that initial treatment with surgery tends to be more cost effective .
| Study | N | 5-year local control, % |
|---|---|---|
| Tis | ||
| Wang | 60 | 92 |
| Spayne, et al | 67 | 98 |
| Garcia-Serra, et al | 30 | 88 |
| Cellai, et al | 89 | 84 |
| T1 | ||
| Wang | 665 | 93 |
| Warde, et al | 449 | 91 (T1a) |
| 82 (T1b) | ||
| Mendenhall, et al | 291 | 94 (T1a) |
| 93 (T1b) | ||
| Cellai, et al | 831 | 84 |
| T2 | ||
| Wang | 237 | 71–77 |
| Warde, et al | 230 | 69 |
| Frata, et al | 256 | 73 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


