CHAPTER 48 Ptosis surgery
Introduction
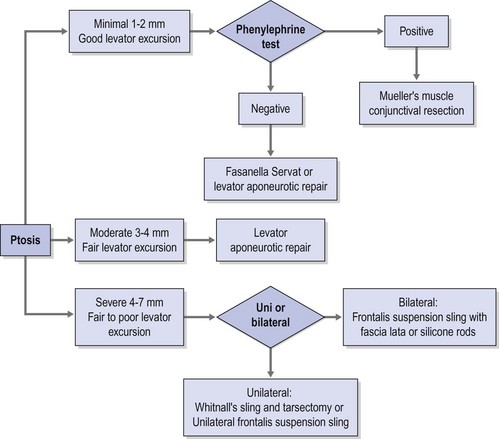
A practical clinical algorithm that allows the surgeon to select a reliable procedure for the management and treatment of the ptosis patient is provided. The algorithm’s goal is to help the surgeon with the selection of the best surgical procedure based on the amount of ptosis and levator function present the time of diagnosis, to achieve successful correction of the ptosis present with good postoperative results and patient satisfaction (Fig. 48.1).
Epidemiologic consideration and terminology
Congenital ptosis can affect one or both eyes; however, in approximately 70% of known cases, congenital ptosis has unilateral affectation. Congenital ptosis may be present at birth, or it may develop later in life. A droopy eyelid(s) that is present at birth, or that develops within the first year of life, is considered congenital in nature. In most cases of congenital ptosis, the problem is isolated and does not affect the vision if the eyelid doesn’t obstruct the visual axis. This holds particularly true in the critical period of visual development, corresponding to the first 3 months of life. The pupil obstruction caused by the droopy eyelid obscures the pediatric patient’s visual field, and permanent loss of vision may occur as a result of amblyopia. Congenital ptosis has no mortality and its morbidity arises from occlusion (deprivation) amblyopia, astigmatism induced from the compression of the droopy eyelid on the corneal tissue, and ocular torticollis. The astigmatism induced by congenital ptosis is generally anisometropic and can be uni or bilateral depending on whether one or both eyelids are affected by the condition. In instances where the degree of anisometropia is significant, anisometropic amblyopia can also develop. In the presence of amblyopia and/or functional limitation caused by congenital ptosis, surgery must be performed to correct the problem early in life, since recent evidence supports that the incidence of deprivation or anisometropic amblyopia in congenital ptosis can be reduced significantly1.
Congenital ptosis can also be seen as part of many congenital syndromes including blepharophimosis syndrome, and certain forms of congenital fibrosis of the extraocular muscles syndromes (CFEOM)2, Marcus Gunn jaw winking syndrome, and congenital Horner syndrome
Clinical features, diagnosis, and differential diagnosis
Although there are numerous classifications for ptosis; such as congenital versus acquired, neurogenic, myogenic, traumatic, and mechanical3, none of those classifications provides a complete approach to the ptosis patient, nor does any of them guide the clinician to the development of a system for adequate repair. Classifying ptosis as minimal = 0.5–1.5 mm, moderate = 2.0–3 mm, or severe >3 mm based on the amount of ptosis provides a practical framework for a logical system of repair when this system is used in conjunction with the amount of levator excursion. This last measurement is an indirect measurement of the viability of the levator palpebrae superioris and an excellent predictor of the surgical outcome. Using this matrix the appropriate choice of surgical strategy can then be applied to each of these three scenarios.
Preoperative assessment
Assessment of ptosis and documentation
Ptosis is documented by the margin to reflex distance 1 (MRD1)4, which is the distance from the central pupillary light reflex to the upper eyelid margin, measured in millimeters. It is important to document the amount of ptosis to the nearest 0.5 mm, if possible. The margin to reflex distance 2 (MRD2) is the distance from the central pupillary light reflex to the lower eyelid margin. The MRD1 plus the MRD2 should equal the palpebral fissure.
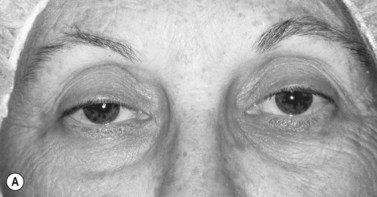
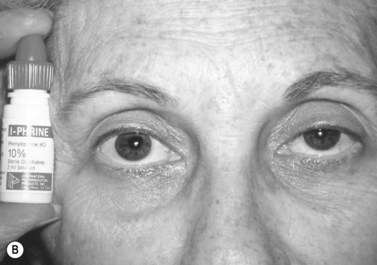
Patients with minimal ptosis (2 mm or less) should have a phenylephrine test performed in the involved eye or eyes after appropriate ptosis measurements have been documented. Either 2.5% or 10% phenylephrine is instilled in the affected eye or eyes. We prefer to use 2.5% phenylephrine. Usually two drops are instilled and the patient is re-examined 5 minutes later. The MRD1 is rechecked in the affected and unaffected eyes (Fig. 48.2). A rise in the MRD1 of 1.5 mm or greater is considered a positive phenylephrine test. This indicates that Müller’s muscle is viable, and the Müller’s muscle conjunctival resection procedure can be performed, also giving the patient a reasonable prediction of the desired result.
The contralateral eye must also be rechecked in patients with unilateral ptosis. When the ptotic eye is occluded, if the MRD1 decreases appreciably in the opposite eye, this usually indicates that bilateral ptosis is present; this finding is consistent with Hering’s law5. The patient may require bilateral surgery. A negative phenylephrine test precludes the use of the Müller’s muscle conjunctival resection procedure, because the outcome of the procedure is unpredictable in this setting. Callahan and Beard3
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


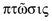 , which means ‘to fall’. In ophthalmology and oculoplastics this term invariably and unequivocally refers to the pathologic condition in which the upper eyelid margin is inferiorly displaced. In the setting of a patient with ptosis, the abnormal eyelid position may cover a significant portion of the cornea and hence the pupillary aperture, sometimes to a degree that is enough to cause visual impairment from the obstruction created. A more suitable term to describe the condition would be that of blepharoptosis, but for the purposes of this chapter the terms ptosis and blepharoptosis will be used interchangeably.
, which means ‘to fall’. In ophthalmology and oculoplastics this term invariably and unequivocally refers to the pathologic condition in which the upper eyelid margin is inferiorly displaced. In the setting of a patient with ptosis, the abnormal eyelid position may cover a significant portion of the cornea and hence the pupillary aperture, sometimes to a degree that is enough to cause visual impairment from the obstruction created. A more suitable term to describe the condition would be that of blepharoptosis, but for the purposes of this chapter the terms ptosis and blepharoptosis will be used interchangeably.