Purpose
To determine the efficacy of 1 intravitreal bevacizumab injection followed by pro re nata (1 + PRN) injection in cases of subfoveal myopic choroidal neovascularization (CNV) and to identify CNV-recurrence–related prognostic factors.
Design
Retrospective observational case series.
Methods
In total, 103 eyes of 89 consecutive naive patients who had subfoveal myopic CNV and had been followed-up for at least 2 years were included. Of those eyes, 24 had recurrences. The remaining eyes were stable after the initial treatment.
Results
The average patient age was 51.1 ± 15.2 years. The average follow-up duration was 44.1 ± 12.7 months. At baseline and at the 1-year, 2-year, and final visits, the average best corrected visual acuities (BCVAs) were 0.57 ± 0.45, 0.38 ± 0.51, 0.40 ± 0.52, and 0.41 ± 0.41 logMAR, respectively. The recurrence rate during follow-up was 23.3%. The BCVA improved by 0.2 logMAR after 2.7 injections in the eyes without recurrence but by only 0.08 logMAR after 6.9 injections in the eyes with recurrence. In univariate analysis, recurrence was associated with older age, more myopic refraction, thinner choroid, larger CNV lesions, and subfoveal hemorrhage at baseline. In multivariate analysis, only baseline CNV lesion size associated significantly with CNV recurrence ( P = 0.002). Recurrence, baseline BCVA, choroidal thickness, and CNV size associated significantly with final BCVA ( P = 0.026, <0.0001, 0.007, and 0.002, respectively). Baseline choroidal thickness, CNV size, age, and presence of lacquer cracks associated significantly with injection number ( P < 0.0001, <0.0001, 0.026, and 0.035, respectively).
Conclusions
1 + PRN intravitreal bevacizumab monotherapy effectively stabilized subfoveal myopic CNV. The CNV size, the baseline BCVA, and the choroidal thickness were the main prognostic factors of subfoveal myopic CNV after 1 + PRN injection of bevacizumab.
Subfoveal choroidal neovascularization (CNV) secondary to pathologic myopia is one of the main causes of vision-threatening conditions in young and middle-aged patients. Although several procedures have been introduced to halt the activation of myopic CNV, including argon laser photocoagulation, photodynamic therapy (PDT), transpupillary thermotherapy, intravitreal bevacizumab injection, intravitreal steroid injection, and surgical excision, most have not yielded particularly convincing results. The exception is intravitreal bevacizumab injection. Bevacizumab is an anti–vascular endothelial growth factor (VEGF), and 2 recent studies have reported that it yields favorable results in myopic CNV. Two other studies showed that a single intravitreal anti-VEGF injection could stabilize myopic CNV for 1 year. Recently, a new approach that involved a single intravitreal bevacizumab injection followed by additional injections as needed was introduced: the effect of this approach might be comparable to that of the initial approach, in which 3 monthly injections were given. Many studies of the effect of anti-VEGF treatment on CNV in age-related macular degeneration have been performed, but far fewer studies have examined the effect of this therapy on CNV in patients with myopia. Consequently, the anti-VEGF treatment protocol and prognostic factors for myopic CNV have not yet been established. Furthermore, to the best of our knowledge, the pretreatment prognostic factors for naive, subfoveal myopic CNV lesions have never been studied.
The present study was performed to determine the effectiveness of intravitreal bevacizumab injection followed by further injections as needed on naive, subfoveal myopic CNV and to identify the clinical pretreatment prognostic factors that associate with final visual acuity, myopic CNV recurrence, and the total number of intravitreal bevacizumab injections.
Methods
This retrospective, observational, comparative, single-center study was performed in accordance with the 1975 Helsinki Declaration and its 1983 revision and was approved by the institutional review board of Asan Medical Center in Seoul, South Korea. All patients were informed about the intravitreal bevacizumab procedure, and informed consent was obtained before the injection.
The computerized clinical charts of consecutive treatment-naive patients (>20 years of age) with subfoveal myopic CNV who were treated with at least 1 intravitreal bevacizumab injection and then followed-up for at least 2 years at the Asan Medical Center between January 1, 2007, and December 31, 2010, were reviewed. Eyes were determined to have degenerative high myopia if they had a spherical equivalent refractive error of ≥−8.0 diopters or an axial length of ≥26.5 mm. The presence of subfoveal myopic CNV was determined by using optical coherence tomography (OCT), fluorescein angiography (FA), and indocyanine green angiography. Myopic CNV was considered to be subfoveal if any portion of the CNV was under the center of the fovea. Agreement by two authors (J.K. and S.G.J.) was needed before a patient was considered to have active myopic CNV. An eye was defined to have active myopic CNV if remaining intra- or subretinal fluid on OCT or leakage on FA was observed. The OCT was performed monthly, and FA was performed every 3 months during the treatment. If CNV activity was resolved on the next follow-up OCT after one injection, no further injections were given until a recurrence was observed. However, if CNV activity remained, additional monthly injections were given after monthly OCT examinations until complete resolution or CNV stabilization was observed. Myopic CNV recurrence was defined as the reactivation or aggravation of intra- or subretinal fluid on OCT or leakage on FA at least 3 months after the initial resolution of the fluid. Initial treatment was the total number of intravitreal bevacizumab injections needed to resolve or stabilize the subfoveal CNV activity.
Patients were excluded from this study if they had any of the following: a history of intraocular injection, photodynamic treatment, photocoagulation, intraocular trauma, inflammation, surgery, or ocular infection; thin sclera and corneal abnormalities that could affect intraocular pressure (IOP) measurements; or a history of any kind of glaucoma, severe macular disease, including extensive chorioretinal atrophy, retinal detachment, severe cataract that could affect visual acuity, or severe systemic problems. Of the 107 consecutive treatment-naive eyes (in 92 patients) that satisfied the inclusion and exclusion criteria, 3 were lost to follow-up for unknown reasons in the following 2 years. Thus, 103 eyes (89 patients) with subfoveal myopic CNV were enrolled and analyzed in this study. The patients were divided into 2 groups on the basis of recurrence after myopic CNV activity had resolved or stabilized, first by the initial intravitreal bevacizumab injection; patients in group B but not group A experienced recurrence.
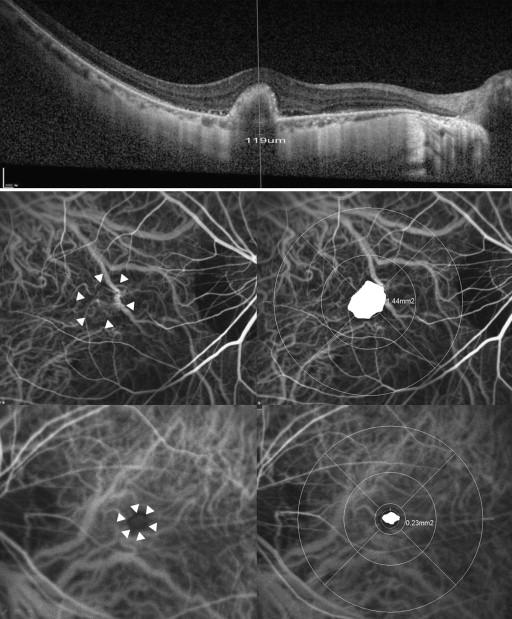
All participants received comprehensive ophthalmologic examinations before the first intravitreal bevacizumab injections, including reviews of their medical and clinical histories, measurements of best corrected visual acuity (BCVA), slit-lamp biomicroscopies, refractions, dilated fundoscopic examinations, measurements of axial length by using IOLMaster (Carl Zeiss Meditec, Jena, Germany), pretreatment IOP measurements by using a Goldman applanation tonometer (Haag-Streit, Koeniz, Switzerland), and OCT (Spectralis, Heidelberg Engineering, Heidelberg, Germany) examinations including extended depth image, FA and indocyanine green angiography (HRA-2; Heidelberg Engineering, Heidelberg, Germany). The size of the CNV lesion and the subfoveal choroidal thickness (between the Bruch membrane and the inner portion of the sclera) were measured manually by 2 authors (H.S.Y. and J.T.K.), who were masked to the clinical information and intravitreal bevacizumab injections of the patients ( Figure 1 ). For these measurements, Heidelberg Eye Explore (v 1.7.1.0, Heidelberg Engineering) was used with the HRA/Spectralis Viewing Module (v 5.6.4.0) and the HSF Region Finder (v 2.4.3.0) using AutoRescan images. The intraclass correlation coefficient (ICC) was used to examine the interobserver agreement in terms of these choroidal thickness and CNV lesion size measurements. The means of the 2 observer values were used in the analysis.
Injection Procedure
After obtaining the informed consent of the patient, the intravitreal bevacizumab injection was performed in a sterile manner. First, topical anesthesia was applied and 10% povidone-iodine was used to scrub the eyelids and lashes. Povidone-iodine eyedrops (5%) were then applied to the conjunctiva bulbi and the fornices. The intravitreal dose of bevacizumab was 1.25 mg per 0.05 mL. Thus, a 0.05 mL volume of bevacizumab was injected through a 30-gauge needle at 3.5 mm of the inferotemporal or inferonasal limbus by two skilled retinal surgeons (J.K. and S.G.J.). After the injection, prophylactic topical antibiotics were applied for 1 week.
Statistical Analysis
All statistical analyses were performed by using Statistical Package for Social Sciences (v 12.0; SPSS, Chicago, IL, USA). Groups A and B were compared in terms of their clinical pretreatment characteristics and post-treatment BCVA by using the Student paired t test for continuous variables and the χ 2 test for categorical variables. To identify clinical factors that associated significantly and independently with myopic CNV recurrence, multivariate logistic regression analysis was performed with backward elimination. The correlation between myopic CNV size and choroidal thickness was analyzed by using the Spearman correlation coefficient. Multivariate stepwise linear regression analysis was used to identify the factors that associate significantly with the final BCVA and total number of intravitreal bevacizumab injections. P < 0.05 was considered to be significant. In multivariate logistic regression analysis, a P value >0.05 was considered to be the appropriate model in the Hosmer–Lemeshow test.
Results
Patient Demographic Data
In total, 103 subfoveal active myopic CNV eyes in 89 Korean patients were included. The patients were followed up for 44.1 ± 12.7 (24-64) months, and the recurrence rate in the 103 eyes during the follow-up period was 23.3%. The participants consisted of 24 men and 79 women, and their average age was 51.1 ± 15.2 years.
Groups A and B consisted of 79 and 24 eyes, respectively. The 2 groups did not differ in terms of gender distribution ( P = 0.424), but the patients in group A were 10 years younger than those in group B, on average; this difference was statistically significant ( P = 0.004). Groups A and B differed significantly in terms of preinjection refraction, axial length, choroidal thickness, CNV lesion size, presence of subfoveal hemorrhage, and diffuse macular atrophy at final visit but not in terms of follow-up duration, preinjection IOP, presence of lacquer cracks, diabetes, hypertension, or baseline focal atrophy ( Table 1 ). With regard to the manual measurements of choroidal thickness and CNV size, the interobserver agreements were good (ICC = 0.925 and 0.947, respectively).
| Characteristic | Total Group | CNV Without Recurrence | CNV with Recurrence | P Value |
|---|---|---|---|---|
| Number of eyes | 103 | 79 | 24 | |
| Number of bevacizumab injections (before/after recurrence) | 3.7 ± 2.3 (2.8/0.9) | 2.7 ± 1.3 (2.7/0) | 6.9 ± 1.7 (3.3/3.7) | <0.0001 a |
| Age at baseline (year) | 51.1 ± 15.2 | 48.7 ± 13.8 | 58.9 ± 17.1 | 0.004 a |
| Sex (men/women) | 24/79 | 17/62 | 7/17 | 0.424 b |
| Total follow-up period (months) | 44.1 ± 12.7 | 43.9 ± 13.7 | 44.8 ± 9.1 | 0.563 a |
| Initial BCVA (logMAR) | 0.57 ± 0.45 | 0.54 ± 0.45 | 0.70 ± 0.44 | 0.115 a |
| 1-year BCVA (logMAR) | 0.38 ± 0.51 | 0.32 ± 0.49 | 0.59 ± 0.58 | 0.021 a |
| 2-year BCVA (logMAR) | 0.40 ± 0.52 | 0.30 ± 0.48 | 0.69 ± 0.63 | 0.001 a |
| BCVA at final follow-up visit (logMAR) | 0.41 ± 0.41 | 0.34 ± 0.38 | 0.62 ± 0.47 | 0.017 a |
| Baseline refraction | −11.55 ± 4.69 | −11.24 ± 4.84 | −12.56 ± 4.07 | 0.028 a |
| Baseline axial length | 28.2 ± 1.4 | 27.8 ± 1.4 | 28.4 ± 1.4 | 0.018 a |
| Baseline intraocular pressure (mm Hg) | 15.8 ± 3.4 | 15.5 ± 3.6 | 16.7 ± 2.7 | 0.143 a |
| Baseline choroidal thickness (μm) | 70.6 ± 26.4 | 76.3 ± 25.5 | 51.8 ± 20.2 | <0.0001 a |
| Baseline CNV lesion size (mm 2 ) | 0.87 ± 0.31 | 0.79 ± 0.29 | 1.14 ± 0.25 | <0.0001 a |
| Presence of subfoveal hemorrhage at baseline (no. eyes) | 21 | 12 | 9 | 0.039 b |
| Presence of lacquer cracks at baseline (no. eyes) | 23 | 14 | 9 | 0.053 b |
| Diabetic mellitus at baseline (no. patients) | 14 | 11 | 3 | 1.0 b |
| Presence of hypertension at baseline (no. patients) | 23 | 21 | 2 | 0.091 b |
| Focal subfoveal and/or juxtafoveal atrophy at baseline | 29 | 19 | 10 | 0.093 b |
| Subfoveal and/or juxtafoveal atrophy at final visit | 19 | 11 | 8 | 0.032 b |
Visual Outcome
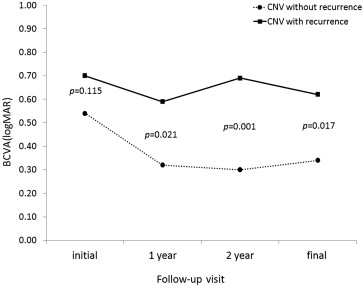
The initial BCVA of the whole cohort was 0.57 ± 0.45 (0.54 ± 0.45 in group A and 0.70 ± 0.44 in group B). Group A tended to have better baseline BCVAs than group B, but this difference did not achieve statistical significance ( P = 0.115). For the whole cohort, the mean BCVA values 1 and 2 years after the initial intravitreal bevacizumab treatment and at the final visit were 0.38 ± 0.51, 0.40 ± 0.52, and 0.41 ± 0.41, respectively. In group A, the mean BCVA improved at the 1-year and final visits compared with baseline ( P < 0.0001 and 0.021, respectively) ( Figure 2 ). However, the difference between the 1-year and final BCVA values did not achieve statistical significance ( P = 0.221). In group B, the mean BCVA improved significantly at the 1-year and final visits compared with baseline ( P = 0.017 and 0.036), but the BCVA improvement between the 1-year and final visits was not significant ( P = 0.091). The mean BCVAs in group A and B differed from each other at the 1-year, 2-year and final follow-up visits (0.021, 0.001 and 0.017 at the 1-year, 2-year and final visits, respectively; Figure 2 ).
Retreatment and Complications
On average, the eyes in groups A and B had 2.7 ± 1.3 and 6.9 ± 1.7 intravitreal bevacizumab injections in total, respectively. Of the 103 eyes of the whole cohort, myopic CNV activity subsided in 86 eyes (83.5%: 67 eyes in group A and 19 eyes in group B, P = 0.844) and stabilized in 17 eyes (16.5%: 12 eyes in group A and 5 eyes in group B, P = 0.586) without complete remission of intraretinal and subretinal fluid after the initial intravitreal bevacizumab injections. Complete resolution of CNV activity after only 1 injection occurred in 21 eyes (20.4%; 24.1% in group A and 8.3% in group B, P = 0.002).
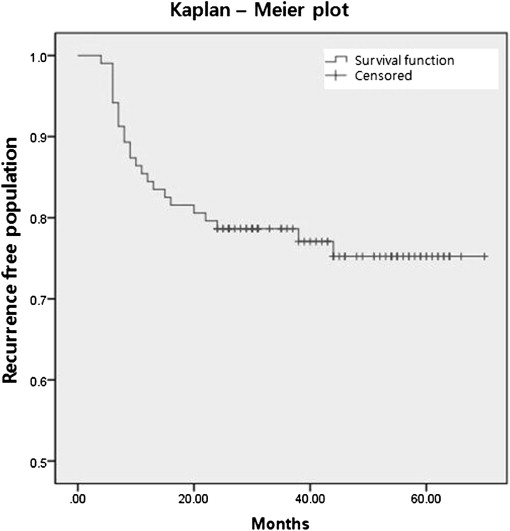
Group B had 1.3 ± 0.4 recurrences on average, and 3.7 additional injections were given on average after the first recurrence. Most of the first recurrences occurred during the first year ( Figure 3 ; 16 eyes, 72.7%). At the 2-year visit, 22 of the 24 (91.7%) group B eyes had experienced a recurrence. Thus, only 2 eyes (8.3%) recurred after 2 years. The 2 groups did not differ in terms of the frequency of intravitreal bevacizumab treatment before the first stabilization of subfoveal myopic CNV was achieved ( P = 0.476). However, group B had a significantly higher total number of injections than group A ( P < 0.0001).
Systemic intravitreal bevacizumab-related complications did not develop in any of the patients. Ocular complications, including endophthalmitis, cataract, glaucoma, and retinal detachment, were not observed during the follow-up period. However, 11 eyes (13.9%) in group A and 8 eyes (33.3%) in group B showed subfoveal or juxtafoveal atrophy ( P = 0.032). Of these, 2 eyes in group B showed diffuse subfoveal chorioretinal atrophy that extended to the extrafoveal area.
Prognostic Factors That Predict Final BCVA, Recurrence, and Frequency of Intravitreal Bevacizumab Injection
Univariate analysis revealed that final BCVA associated significantly with baseline age, baseline BCVA, recurrence, total number of injections, baseline choroidal thickness, baseline CNV size, baseline presence of subretinal hemorrhage, and baseline presence of lacquer crack. However, in multivariate stepwise linear regression analysis, final BCVA associated significantly with baseline BCVA, recurrence, baseline choroidal thickness, and baseline CNV size only ( Table 2 ).
| Factor | Univariate Analysis | Multivariate Analysis (corrected R 2 = 0.593) | ||
|---|---|---|---|---|
| Correlation Coefficient | P value | Regression Coefficient B | P value | |
| Age at baseline (year) | 0.299 | 0.001 | ||
| Initial BCVA (logMAR) | 0.785 | <0.0001 | 0.616 | <0.0001 |
| Recurrence | 0 | 0.005 | 0.100 | 0.026 |
| Injection number | 0.268 | 0.003 | ||
| Follow-up duration | 0.135 | 0.086 | ||
| Baseline choroidal thickness | −0.225 | 0.011 | −0.008 | 0.007 |
| Baseline refraction | –0.030 | 0.032 | ||
| Baseline IOP | 0.302 | 0.381 | ||
| Baseline CNV lesion size | 0.246 | 0.001 | 0.065 | 0.002 |
| Presence of subretinal hemorrhage at baseline | 0.115 | 0.048 | ||
| Presence of lacquer cracks at baseline | 0.226 | 0.011 | ||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


