Current diagnostic approaches
Symptoms based
Mechanism based
Angle-closure configuration based
International Society of Geography and Epidemiology of Ophthalmology (ISGEO)
Acute angle-closure
Pupillary block
Pure pupillary block
Primary angle-closure suspect (PACS)
Angle closes suddenly with high intraocular pressure (IOP), symptoms, and rapid optic nerve damage
Restriction of aqueous flow from posterior to anterior chamber
Shallow anterior chamber (AC), more anterior lens position, strong pupillary block tendency
Angle occludable, no peripheral anterior synechiae (PAS) normal (Nl) IOP, optic nerve, Visual Field (VF)
Subacute-angle-closure
Plateau iris
Non-pupillary block
Primary angle-closure (PAC)
Milder, self-limiting acute angle-closure may be intermittent
Anteriorly positioned ciliary processes push iris forward
Deeper AC, more posterior lens position, anterior positioned ciliary body (plateau), iris crowding
Angle occludable, increased IOP and/or PASNl optic nerve, VF
Chronic angle-closure
Lens-induced angle-closure
Multiple mechanisms
Primary angle-closure glaucoma (PACG)
Partially closed angle, increased IOP, symptoms mild or absent until very late
Block results from swollen or mobile lens
Features of both pupillary block and non-pupillary block, difficult to distinguish prior to laser iridotomy
Primary angle-closure abnormal optic nerve, Visual Field
Latent angle-closure
Retrolenticular mechanisms
Shallow AC, narrow angle, at risk for closure, nl IOP, nl optic nerve
Increased retrolenticular pressure, from ciliary block, posterior aqueous diversion
Note: Symptom-based classification does not describe degree of angle obstruction or presence of nerve damage
Note: Mechanism-based classification is easy to understand and aids in determining the choice of treatment
Note: The angle-closure approach is limited to primary angle-closure glaucoma; it does not consider degree of angle obstruction or abnormal optic nerve
Note: The ISGEO classification defines three stages in the natural history; glaucoma is defined by abnormal optic nerve, visual field
International Society of Geography and Epidemiology of Ophthalmology Classification
Foster and associates have published a classification of angle-closure that has been widely used in prevalence studies and epidemiological research [7]. It specifies three stages: Primary angle-closure suspect (PACS), Primary angle-closure (PAC) and (PACG) in the progression of angle-closure from ITC and from signs of disease, such as elevated intraocular pressure and peripheral anterior synechia, to frank optic nerve damage as the result of chronically elevated pressure.
Primary Angle-Closure Suspect
The individual with an anatomically narrow angle can be classified as a primary angle-closure suspect (PACS) when gonioscopy reveals a shallow peripheral angle recess with the iris close to the trabecular meshwork. The PACS has iridotrabecular contact (ITC) in three or more quadrants, no peripheral anterior synechiae, and normal intraocular pressure (IOP), optic nerve, and visual field [3].
Even without a history of sub-acute or acute angle-closure attacks, this patient is at risk for future angle-closure. If it is not deemed necessary to treat the eyes at the time of initial diagnosis, the patient should be educated as to the potential for acute angle-closure, and the need for immediate care should symptoms occur. It may be wise to advise the patient to have an examination before any travel and/or against extended travel to locations where eye care is not readily available.
Primary Angle-Closure
In an acute primary angle-closure attack, there is a sudden obstruction of the anterior chamber (AC) angle with an acute IOP elevation to very high levels. This results in characteristic symptoms and ultimately blindness if left untreated. In PAC, the individual has iridotrabecular contact (ITC) in three or more quadrants, either elevated IOP and/or primary peripheral anterior synechiae (PAS), with normal optic nerve and normal visual field [3].
Primary Angle-Closure Glaucoma
In PACG, the individual has ITC in three or more quadrants, with evidence of glaucomatous damage to the optic nerve or visual field [3].
In chronic PACG, the angle usually closes gradually over time. Sustained ITC may result in a pathophysiologic cascade of trabecular dysfunction, PAS, increased IOP, optic nerve damage, and visual field loss. Because the pressure rises gradually, the patient may have no symptoms until there is advanced glaucomatous visual loss. In this regard, the natural history of asymptomatic progression to blindness may be analogous to POAG.
Mechanistic (Pathophysiologic) Classification
Ritch and Lowe [8] proposed a classification based on the angle anatomy and mechanisms of outflow obstruction. Primary and secondary angle-closures can also be classified mechanistically by the presence or absence of pupillary block and whether the iris is anatomically “pushed forward” or “pulled forward” to the trabecular meshwork (Table 14.2). Four successive anatomic levels of angle-closure have been described: iris (pupillary block), ciliary body (plateau iris), the lens (phacomorphic glaucoma), and vectors posterior to the lens (malignant glaucoma). Angle-closure may result from blockage of outflow with one or more of these anatomical changes, with one or more mechanisms involved. Pupillary block may occur to some degree in other forms of glaucoma and must be ruled out. A consensus statement from the 3rd Global Association of International Glaucoma Societies (AIGS) Consensus Meeting on Angle-Closure Glaucoma concluded the following: “Although the amount of pupillary block may vary among eyes with angle closure, all eyes with angle closure require treatment with iridotomy” [3].
Table 14.2
Mechanisms of angle-closure glaucoma
Underlying mechanisms of angle-closure glaucoma |
Posterior mechanisms – Iris pushed forward |
With pupillary block |
Pupillary block glaucoma (primary angle-closure) |
Lens-related mechanisms |
Subluxation of lens (partial zonular dehiscence) |
Mobile lens syndrome (weakened or lax zonules) |
Intumescent lens (phacomorphic) |
Implantable contact lens (ICL) |
Posterior synechiae |
Crystalline lens |
Pseudophakia |
Iris-vitreous block in aphakia |
Uveitis |
Without pupillary block |
Plateau iris configuration/syndrome (anteriorly located ciliary processes) |
Pseudoplateau iris (iris ciliary body cysts, tumors) |
Malignant glaucoma (aqueous misdirection syndrome, ciliary block) |
Lens-related mechanisms |
Subluxation of lens (partial zonular dehiscence) |
Mobile lens syndrome (weakened or lax zonules) |
Intumescent lens (phacomorphic) |
After cataract extraction (vitreous displaced forward) |
Uveal edema (after central retinal vein occlusion, after extensive panretinal photocoagulation (PRP), after scleral buckling procedures) |
Tumors |
Retinoblastoma |
Malignant melanoma |
Cysts |
Iris |
Ciliary body |
Retrolenticular tissue |
Persistent hyperplastic primary vitreous (PHPV) |
Retinopathy of prematurity (ROP) |
Anterior mechanisms – Iris pulled forward |
Membrane contraction |
Iridocorneal endothelial syndrome (ICE) – migration of corneal endothelium |
Posterior polymorphous dystrophy (PPMD) |
Penetrating and nonpenetrating trauma |
Neovascular glaucoma (NVG) |
Inflammatory precipitate contraction |
Inflammatory membrane |
Fuch’s heterochromic iridocyclitis |
Syphilitic interstitial keratitis |
The possibility to visualize the anterior segment structures with imaging techniques such as ultrasound biomicroscopy (UBM) and optical coherence tomography (OCT) has enhanced our ability to classify angle-closure glaucoma better anatomically, determine the mechanisms involved, and institute proper therapy. Imaging can often clarify nebulous situations, particularly if one clinician has found the angle to be open when another has not.
Plateau Iris Configuration and Plateau Iris Syndrome
Plateau iris configuration and plateau iris syndrome are conditions that are often confused with pupillary block and PAC. Plateau iris configuration is an anatomically abnormal anterior segment configuration that has the potential to result in angle-closure. On gonioscopy in plateau iris, the root of the iris may be short and inserted more anteriorly on the ciliary face. This results in a plateau configuration with a shallow peripheral angle and a sharp drop-off of the peripheral iris. The anterior chamber may be of normal depth centrally and the iris only slightly convex.
Plateau iris syndrome may develop spontaneously or after pupillary dilation and may present with symptoms identical to an acute angle-closure (pupillary block) attack. In some cases, there may also be an additional element of relative pupillary block. Ultrasound biomicroscopy (UBM), when available, is especially useful in the assessment and differentiation of pupillary block and plateau iris configurations. Imaging under both light and dark conditions is helpful. In a recent study, Filho and associates compared the UBM findings between plateau iris configuration (PIC) and primary open-angle glaucoma with narrow angles (POAGNA) [9]. They found that PIC eyes are characterized by a more crowded anterior segment compared with POAGNA eyes. However, when the same parameters were evaluated between light and dark circumstances, no changes occurred.
The etiology of a glaucoma attack may not be appreciated without imaging. The episode is therefore often treated with laser iridotomy. An iridotomy may not open the angle or may only open it partially. An eye with an open laser iridotomy and plateau iris configuration may ultimately undergo an acute attack, confirming the diagnosis.
Laser peripheral iridoplasty can open the angle in plateau iris syndrome [10]. This technique is not readily embraced by all ophthalmologists – some voicing discomfort over the inflammatory changes created by the application of the laser to the iris. This technique employs argon laser burns of low power and large spot size to the peripheral iris adjacent to the angle to physically pull open the angle. The eye is pretreated with apraclonidine or brimonidine. The argon laser treatment parameters are set to produce contraction burns (500 μm spot, 0.5–0.7 s, 80–100 mW). The Abraham lens is used to place the burns as peripherally as possible. The laser power should be adjusted accordingly so that the spots produce contraction – not penetration. Failure to see any contraction is an indication of power being too low or that PAS is holding the iris to the sclera and preventing contraction. A full treatment requires from 20 to 24 spots over 360°. Overtreatment should be avoided. Approximately two spot diameters should be left between each spot, and radial iris vessels should be avoided. Postoperatively, the patient is treated with topical steroid drops with the pressure monitored as with any other glaucoma laser procedure [11]. A primary treatment of combined peripheral iridoplasty and laser iridotomy performed sequentially has also been proposed [12]. A patient with plateau iris configuration is susceptible to further shallowing of the anterior chamber with aging and may also develop pupillary block.
Primary Angle-Closure Glaucoma Versus Secondary Angle-Closure Glaucoma
Angle-closure glaucoma has two pathophysiologic etiologies. The final common mechanism of each type is appositional obstruction of the trabecular meshwork by peripheral iris. This results in an impairment of aqueous outflow accompanied by a sudden (acute) or gradual (chronic) IOP increases. Raised IOP and subsequent glaucomatous damage to the optic nerve result either from mechanisms that push the iris forward from behind or from mechanisms that pull the iris forward into contact with the trabecular meshwork. Degrees of closure range from ITC with normal IOP and eventual to total trabecular outflow occlusion with elevated pressure and eventual PAS.
Primary angle-closure is caused by pupillary block that results in appositional and/or synechial closure of the anterior chamber angle. Conditions that were previously classified as primary angle-closure glaucoma, such as plateau iris, are generally associated with anatomic abnormalities of the anterior chamber. Conditions classified as secondary angle-closure glaucoma are associated with other ocular or systemic abnormalities.
In cases of primary acute angle-closure attack or an acute secondary angle-closure, the IOP can rapidly elevate to a dangerous level. Prompt diagnosis and treatment are paramount as blindness may ensue quickly.
Gonioscopy
Gonioscopy is essential to identifying the underlying anatomy and pathophysiology of angle-closure in order to properly classify the type of glaucoma and initiate treatment. Gonioscopy as a technique to visualize the anterior chamber angle was devised by Trantas in the 1880s [13]. His method employed indenting the sclera and viewing with a direct ophthalmoscope. Koeppe gonioscopy, a method rarely used today both because of the elaborate setup it requires and because it is time-consuming to perform, is an excellent method of “direct” gonioscopy. A dome-shaped Koeppe lens is applied to each eye with the patient in the exam chair in the recumbent position. The directly through the lens with a handheld source of magnification and illumination (Fig. 14.1a, b). The advantages of this method are that there is no distortion of the anatomy and that corresponding area of the angle in each eye can easily be compared by placing a lens on each eye and then viewing back and forth. The disadvantages of Koeppe lenses are that they require extra diagnostic equipment, cannot be used at the slit lamp, and do not allow for compression gonioscopy that would allow dynamic manipulation of the anterior chamber angle. Because the lens remains on the eye without any pressure from the examiner’s hands, there is no possibility that the act of holding the lens on the eye can deform the angle. Deformation of the angle is a common problem by novice gonioscopists.
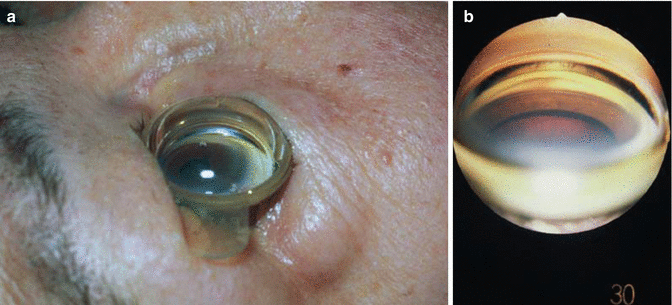

Fig. 14.1
(a) Koeppe lens placed on eye in the recumbent position for direct gonioscopy. View through Koeppe lens of open, (b) heavily pigmented angle
Current commonly employed methods of gonioscopy are termed “indirect” because the lenses are mirrored and the portion of the angle viewed is 180° away, as seen through the mirror. The widespread use of gonioscopy following the introduction of the Goldmann lens in 1938 greatly improved the understanding of anterior chamber anatomy and the pathogenesis of the angle-closure glaucoma. The three-mirror Goldmann lens (Fig. 14.2) is the most versatile Goldmann-type lens because in addition to the mirror for examining the angle, the other portions of the lens can also be used to view the posterior pole and more peripheral retina. The two-mirror Goldmann and single-mirror Goldmann lenses are smaller and easier to manipulate at the slit lamp. The advantage of the Goldmann type of lens is that it does not distort the appearance of the angle. The disadvantage is that viscous gonio solution must be employed as a coupling agent between the lens and the patient’s corneal surface.
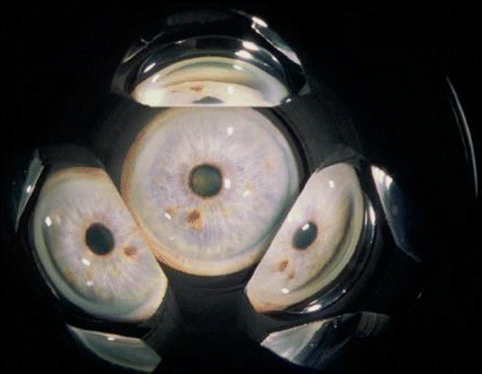

Fig. 14.2
View through Goldmann three-mirror lens. The top mirror (angle mirror) shows an open angle
The original Zeiss lens is a pyramidal, silvered glass, four-mirror lens with a shallow corneal curvature on its ocular surface. Non-silvered versions with high-index glass, which afford an excellent view, are now available. The traditional lens is clipped into a retaining fork on a metal handle that is held in the clinician’s fingertips (Fig. 14.3a, b). One advantage of this lens is that it is convenient to use during a routine slit lamp examination, as it is placed directly on the tear film and does not require a sticky gonio solution. At the conclusion of the gonio exam, the cornea remains clean; there is nothing to rinse off from the eye. Another major advantage is that the Zeiss lens may be employed to perform “compression gonioscopy.” Compression gonioscopy is the intentional depression of the cornea with the gonioscopy lens to force fluid into the peripheral angle open in an attempt to widen or open it (Fig. 14.4). This technique is useful to determine if the narrow angle has appositional closure, synechial closure, or both [14]. The findings from this “dynamic gonioscopy” can aid the clinician in determining whether the patient would best benefit from laser iridotomy, trabeculectomy, or other glaucoma procedure. The disadvantage of the Zeiss lens is that the clinician must be wary not to unintentionally compress the cornea when initially viewing the angle structures and thus erroneously overestimate the depth of the angle or its susceptibility for closure. The Zeiss lens (and its derivatives) requires more skill by the examiner, than does the Goldmann lens, to get an adequate view of angle structures.
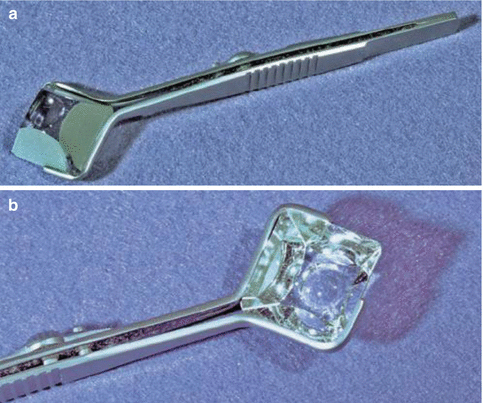
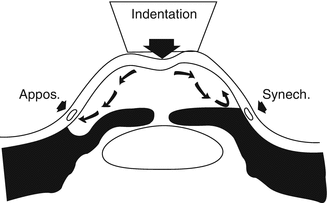

Fig. 14.3
Zeiss lens. (a) Undersurface of Zeiss four-mirror lens shows the concave face of the lens which is applied to the tear film of the cornea. (b) Close-up of the clinician’s (slit lamp) view of the four mirrors

Fig. 14.4
Compression gonioscopy. Compression with the four-mirror lens is employed to indent the cornea and force fluid into the peripheral angle. When the angle-closure is appositional (iridotrabecular contact), the iris is pushed away allowing access of aqueous to the trabecular meshwork. When the angle-closure is synechial the angle cannot be opened by compression
The Posner lens (Fig. 14.5) is a variation of the Zeiss-type lens with the four-mirror lens mounted on a wand-shaped handle. The Sussman lens (Fig. 14.6a, b) is a similar four-mirror lens mounted in a retaining ring and is directly held in the fingertips as is the Goldmann lens. The Posner and Sussman lenses are used in a similar fashion as the Zeiss lens to view the anterior chamber angle and to perform compression. In some patients, the corneal ocular surface curvature of these types of lenses does not well match that of the patient’s cornea, and it is difficult to get the tear film to fill the interface appropriately. In these instances, a single drop of a thick artificial tear solution can often remedy this problem. With experience, most ophthalmologists easily master the technique of using these gonio lenses that use only tears as a coupling medium.



Fig. 14.5
Posner four-mirror indirect gonioscopy lens

Fig. 14.6
(a) Sussman four-mirror indirect gonioscopy lens side view. (b) Sussman four-mirror indirect gonioscopy lens mirror view. The central concave surface is applied to the tear film of the cornea, and the angle is viewed through each of the mirrors
Various grading systems have been proposed to describe the appearance of the AC angle and its probability of closure [15].
The Scheie system grades the angle from “0 wide open” to “4 closed.”
The Shaffer system, the most widely used, grades the angle from “0 closed” to grade “4 wide open.” A grade 1 angle would be considered possibly to probably occludable.
The Spaeth system is more descriptive and specifies the iris insertion, amount of pigment, and the configuration of the iris.
The findings of the gonioscopy exam may be diagramed on a chart of concentric rings (Fig. 14.7a) with pathology such as closure or synechiae charted (Fig. 14.7b, c).
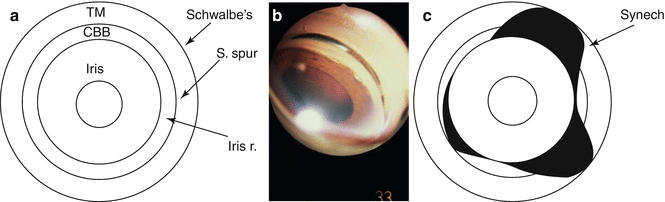

Fig. 14.7
Diagramming the angle (a) Blank chart for diagramming the angle with the landmarks of Schwalbe’s line, scleral spur, and iris root indicated by lines and trabecular meshwork (TM), ciliary body band (CBB), and iris indicated by concentric rings. (b) Gonioscopic photographic (through Koeppe lens) of synechial angle-closure. (c) Angle diagram of synechial angle-closure depicting synechia completely covering the TM band and lower synechia up to the inferior border of the TM
Gonioscopy lenses have therapeutic as well as diagnostic uses. In cases of acute angle-closure attacks, dynamic gonioscopy (with Zeiss, Posner, or Sussman lenses) can sometimes be used to interrupt the attack by compressing the cornea to relieve apposition and forcibly opening the peripheral angle after medical therapy has been initiated. The flat, nonsilvered portion of these lenses (between the mirrors) may also be applied to a filtering bleb after trabeculectomy surgery to compress the conjunctiva and visualize the underlying sutures to perform laser suture lysis when a lens dedicated for this purpose is not available. Gonioscopy is discussed in greater detail in Chap. 10.
Anterior Segment Imaging
Ultrasound Biomicroscopy, Optical Coherence Tomography, Scanning Peripheral Anterior Chamber Depth Analyzer, and Darkroom Infrared Gonioscopy
Imaging techniques such as ultrasound biomicroscopy (UBM) and optical coherence tomography (OCT), although not usually available to the community-based physician, have proven to be a useful adjunct to gonioscopy. Compared to gonioscopy, they are more objective, not as dependent on patient cooperation, and more precise in determining the anterior segment anatomy [16–18]. The role of UBM in the differential diagnosis of angle-closure glaucoma has been extensively reviewed by Tello et al. [19]. The standard UBM images the anterior segment with ultrasound in the low-frequency range. It is useful to view the angle, iris, lens, postlenticular space, and ciliary body (Fig. 14.8a–c). The newer higher-frequency UBM can visualize Schlemm’s canal and the angle and can be used for imaging in the operating room. UBM can be used to elucidate pathophysiologic mechanisms in narrow-angle and angle-closure eyes. It is especially helpful in the assessment of pupillary block (Fig. 14.9a, b) and plateau iris configurations (Fig. 14.10). UBM is valuable in evaluating the persistence of appositional angle-closure after laser iridotomy in the fellow eyes after acute primary angle-closure [20].
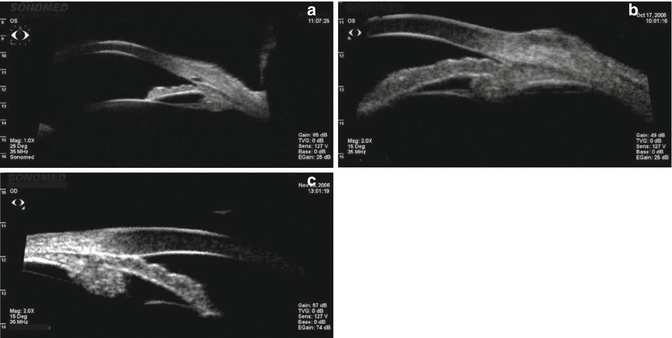
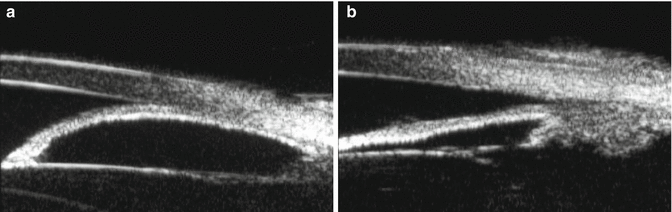
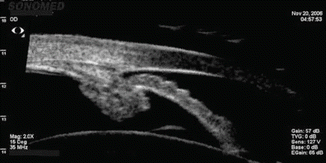

Fig. 14.8
(a) UBM open-angle. The anterior chamber is deep with the iris well away from the TM. The lucency internal to the TM is Schlemm’s canal. The ciliary sulcus is imaged as the larger lucent area posterior to the iris. (b) UBM narrow angle. The iris is convex, and the peripheral anterior chamber is shallow but the angle is not closed. (c) UBM closed angle. The angle is closed with iridotrabecular contact (Images courtesy of Sonomed, Inc., Lake Success, NY)

Fig. 14.9
(a) Pupillary block. The convex iris is bowed forward occluding the angle. (b) Pupillary block broken with angle opened by laser iridotomy (Images courtesy of Fred Kapetansky, M.D.)

Fig. 14.10
Plateau iris. There is a shallow peripheral angle with a sharp drop-off of the peripheral iris (Image courtesy of Sonomed, Inc., Lake Success, NY)
Optical coherence tomography images the anterior segment with light rather than ultrasound (Fig. 14.11a, b). It does not require direct ocular contact and is more comfortable for the patient. OCT is excellent at imaging structures anterior to the iris, but is not good at penetrating the pigmentation of the iris.
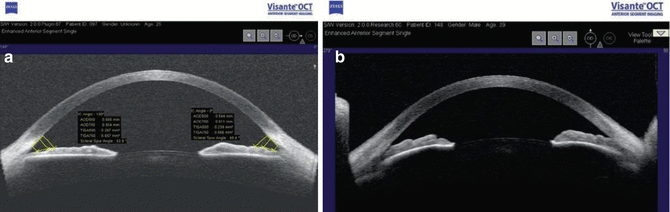

Fig. 14.11
(a) Anterior segment OCT. OCT of open angle with anterior segment measurements. (b) OCT of narrow angle with convex iris (Images courtesy of Zeiss-Humphrey Meditec)
The scanning peripheral anterior chamber depth analyzer (SPAC) obtains rapid slit photographs of the central and peripheral anterior chamber and creates an anterior iris surface contour. This measure can be compared to sample databases [21].
A new imaging method for evaluating the angle is darkroom infrared gonioscopy (DIG). Gonioscopy is performed with infrared light eliminating the bright light of the slit lamp. This makes it possible to view the dynamics of the angle in the dark as well as light or accommodative stimulation. Although there is no commercially available instrument available at present, DIG can be performed by modifying currently available video instruments [22].
Pathogenesis and Pathophysiology of the Angle-Closure Glaucoma
Angle-closure results from appositional obstruction of the trabecular meshwork by the peripheral iris. This can be a consequence of an abnormal relationship between the size and position of the structures of the anterior segment and the relative pressure differential between the anterior and posterior chambers. Blockage of the meshwork may occur by forces acting at four successive anatomic levels, as proposed by Ritch et al. [8]. The first anatomic level described is at the iris. Mechanistically, this is the site of pupillary block. The next anatomic level is at the ciliary body, the level at which plateau iris syndrome occurs. The third level is the lens, which is involved in phacogenic mechanisms. The fourth anatomic level relates to forces posterior to the lens. This level has therefore been described as retinovitreal or uveal. Mechanistically this level is the site of malignant glaucoma and secondary angle-closure syndromes. Furthermore, each level of block can have some component of block of the level preceding it, creating a situation in which multiple mechanisms should be considered and treated. Anterior segment abnormalities – such as inflammatory, fibrotic, or fibrovascular conditions – can pull the peripheral iris forward to cause appositional and eventual synechial closure of the angle. Pupillary block, plateau iris, aqueous misdirection, or posterior segment abnormalities – such as choroidal swelling, choroidal hemorrhage, choroidal detachment, tumor, or space occupying lesions – can push the iris forward from behind to cause appositional and eventual synechial closure of the angle (Table 14.2). Of these various mechanisms, pupillary block is by far the most common. Further, the mechanisms are not mutually exclusive, and relative pupillary block may be present.
Pupillary Block Mechanism
Pupillary block is the most frequent etiology of angle-closure [23, 24] and the underlying mechanism of most cases of PACG. Flow through the pupil is compromised, and the peripheral iris bows forward against the trabecular meshwork. Pupillary block is maximum at the point of mid-dilation (Fig. 14.12). Outflow is impeded, and the pressure gradient between the posterior and anterior chambers increases until eventually there is no outflow. Eyes with preexisting shallow anterior chambers are predisposed to pupillary block. (This is why gonioscopy should be performed on all patients with eyes that appear to have shallow anterior chambers by slit lamp examination.) Apposition of the pupil to anterior lens capsule is the greatest in the mid-dilated position. This apposition can be relieved by constriction of the iris or dilation of the pupil away from the mid-position. Surgical iridectomy or laser iridotomy will permanently eliminate angle-closure from pupillary block (but not cure plateau iris configuration as described later).
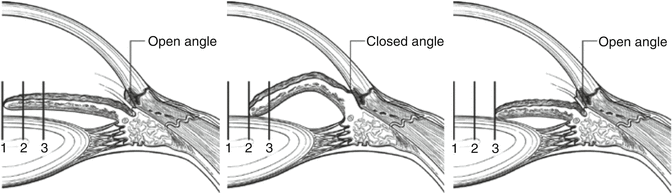

Fig. 14.12
Pupillary block mechanism. (1) The pupil is constricted and the angle is open. (2) The pupil is in the mid-dilated position. Pupillary block is maximal in this position, and as a result the iris is bowed anteriorly and the angle narrows. (3) The pupil is more completely dilated, and the relative pupillary block is diminished, with a return to a flatter iris configuration. If full-blown angle-closure occurs, the iris may stay in the mid-dilated position until the angle-closure attack is broken
Another form of a more chronic pupillary block can occur when the pupil is completely adherent to the lens capsule (secluded) by posterior synechia. This is often the result of an inflammatory process. The pressure gradient balloons the iris forward resulting in iris bombé (Fig. 14.13).
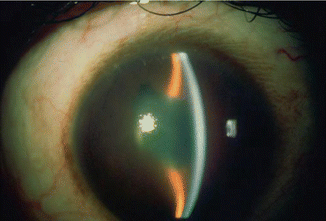

Fig. 14.13
Iris bombé with secluded pupil secondary to inflammatory glaucoma
It is essential that all cases of pupillary block be interrupted in order that anatomic appositional closure does not result in permanent synechial closure. If that occurs, relieving the pupillary block may not restore normal intraocular pressure, because access to the trabecular meshwork is prevented, and additional surgical measures may become necessary to lower IOP to safe levels.
Primary Angle-Closure
Primary angle-closure glaucoma is associated with relative pupillary block in most of the cases [23, 24]. It must be differentiated from plateau iris configuration and lens-induced secondary pupillary block. Risk factors for developing primary angle-closure glaucoma are well established and are discussed in the following sections.
Risk Factors for Developing Primary Angle-Closure
The major risk factors for angle-closure are advancing age, female gender, and Asian ethnicity [25–31]. Women have shallower anterior chambers than men. The anterior chamber shallows with age [32, 33]. Biometric risk factors include small anterior segment dimensions and refractive error with axial and limbal anterior chamber depth the most strongly correlated to PAC and PACG [34, 35].
Age
PACG is generally a disease of an aging population. It is uncommon to see angle-closure in individuals less than 40 years of age, after which time the incidence advances by the decade. The depth and volume of the anterior chamber decrease with age. The increased incidence of angle-closure with age can be attributed to the normal increase in thickness of the lens with age, akin to the increasing rings and thickness of a tree trunk with age. As cataracts mature, the preexisting shallow anterior chamber and forward movement of the lens can result in increasing pupillary block. In young patients, PACG is typically associated with other anatomical abnormalities [36]. In a young angle-closure patient, consideration should be given to plateau iris mechanism.
Gender
Independent of race, primary angle-closure has a two to four times greater prevalence for women than men. This increase cannot be accounted for by women’s somewhat smaller anterior chamber depth (ACD) and axial lengths [37].
Race
The prevalence of PACG in patients older than age 40 varies greatly depending on race (Table 14.3). The difference in some of the biometric parameters (anterior chamber depth, axial length, which will be discussed) accounts for some of the variation in white and Inuit populations. However, biometric findings alone cannot justify the increased incidence in the Chinese and East Asian populations. In addition, acute forms are more prevalent in whites, whereas Africans and Asians are more likely to suffer from asymptomatic chronic disease. The burden of angle-closure glaucoma is greater in Asian countries. There has been controversy regarding the explanation of the greater rate of angle-closure in this demographic population [37, 43, 47, 54–59].
Table 14.3
Reported prevalence of angle-closure glaucoma among select populations
Angle-closure prevalence | ||
|---|---|---|
Population | Location or subpopulation | Reported prevalence (%) |
2.65–3.8 (age >40) | ||
Inuit | East Greenlandic Inuit [40] | 2.5 (age ≥40) |
Taiwan [41] | 3.0 (age ≥40) | |
Mongolia [27] | 1.4 (age ≥40) | |
Singapore Chinese [42] | 1.25 (age ≥60) | |
Southern India [43] | 1.08 (age ≥40) | |
Asian | Japan [26] | 0.34 (age ≥40) |
Hispanic | Arizona, USA [44] | 0.1 (age >40) |
African and African derived | Baltimore, USA [45] | 0.9 (age ≥40) |
Tanzania, East Africa [46] | 0.58 (age ≥40) | |
South African Zulus [47] | 0.5 (age ≥40) | |
Italy [48] | 0.6 (age ≥40) | |
Baltimore, USA [45] | 0.4 (age ≥40) | |
Bedford, United Kingdom [49] | 0.17 (age >40) | |
Sweden [50] | 0.1 (age 55–69) | |
European and European derived | United Kingdom [51] | 0.09 (age ≥40) |
Beaver Dam, USA [52] | 0.04 (age ≥43) | |
Ireland [53] | 0.009 (age >50) | |
The blinding consequences of PAC and PACG have been well studied and emphasized in East Asian populations, especially the Chinese. Foster reported that an estimated 28 million Chinese have narrow angles, predisposing them to PACG. Of these nine million are estimated to have peripheral anterior synechia or increased intraocular pressure indicative of significant angle-closure. It is postulated that PACG is the cause of 91 % of bilateral blindness from glaucoma in the Chinese [4].
Lin and associates have studied the characteristics of glaucoma in Chinese Americans, Vietnamese Americans, Filipino Americans, and Japanese Americans extensively. In a retrospective, clinic-based study within the San Francisco Chinatown district, 163 Chinese Americans (61 % women, mean age 68; 39 % men, mean age 65) were evaluated. In the entire group 57 % had narrow angles by gonioscopy. In those 60 years or older, 73 % were found to have narrow angles [60]. In a large, prospective cross-sectional study of mainland Chinese, Chinese Americans, and white Americans, all of the Chinese had similar biometric parameters, which were smaller or narrower than the white Americans, even after accounting for axial length and refractive error [61]. Vietnamese Americans were also found to have a high incidence of PACG in a retrospective, cross-sectional study of 2,247 patients. The overall rate of glaucoma was 14 % of which 53 % was POAG, 31 % PACG, 10 % combined mechanism glaucoma, and 5 % secondary glaucoma [62]. In a study of 1,112 Filipino American versus white American patients, the Filipino Americans had a higher rate of glaucoma, 15 % versus 9 %; a higher proportion that was classified as angle-closure, 8 % versus 0 %; and a higher proportion that was normal tension glaucoma, 47 % versus 27 % [63]. With regard to Japanese Americans, in a clinic-based study of 1732 (1,321 40 years or older, 64 % women, 36 % men) patients, the prevalence of glaucoma was 6.4 % and glaucoma suspects was 10.5 %. In contrast to the preceding Asian groups, the distribution of glaucoma types was skewed towards normal tension glaucoma (NTG) 69 %, angle-closure glaucoma 3 %, POAG 17 %, secondary glaucoma 9 %, MMG 2 % [64].
Genetic Predisposition
It has been reported that angle-closure glaucoma patients and their siblings, but not their offspring, have less than normal axial lengths [65]. In population studies, first-degree relatives (parents, siblings, and offspring) of primary angle-closure glaucoma patients are found to be at greater risk. Indeed, it may be prudent to examine the first-degree relatives of a patient who presents with angle-closure glaucoma. In whites, the prevalence increases 1–12 %. In the Inuit population, the risk is 3.5-fold, and, in the Chinese, the risk is six-fold greater than in the general population [37]. Nanophthalmos, a secondary angle-closure, has been associated with chromosomal abnormalities.
Biometrics
Anterior chamber dimensions may be measured by optical pachymetry, OCT, UBM, Scheimpflug photography, and SPAC. The OCT can measure central anterior chamber depth, corneal curvature, and axial length by a combination of infrared partial coherence biometry and optical method. The Pentacam uses other methods to measure central ACD by rotating Scheimpflug photography to yield more insight into angle structures. Small anterior segments and short axial lengths are typical in patients who develop primary angle-closure. The primary findings predisposing to angle-closure are a shallow anterior chamber depth, a thicker lens, increased anterior curvature of the lens, a shorter axial length, a small corneal diameter, and radius of corneal curvature. An anterior chamber depth (ACD) of less than 2.5 mm increases the chances of primary angle-closure, whereas most patients with primary angle-closure have an ACD of less than 2.1 mm. Advances in biometry techniques have demonstrated a definite association between ACD and peripheral anterior synechia (PAS). An ACD of greater than 2.4 mm is not typically associated with primary PAS. There is, however, a strong correlation of increasing PAS formation with an ACD shallower than 2.4 mm. The previous notwithstanding, angle-closure still occurs with deep anterior chambers in some cases [35, 37, 58, 59, 66].
New Concepts of Angle-Closure
Quigley and associates have studied the change of iris cross-sectional (CS) area with pupillary dilation as a risk factor in angle-closure. This concept likens the iris to a sponge that is wrung out when the pupil dilates. The iris in open-angle eyes routinely loses half of its area when the pupil dilates; however, in angle-closure, the iris maintains its volume by retaining water and increases the possibility that it will block the angle.
They employed anterior segment OCT in angle-closure and open-angle glaucoma eyes and evaluated the change in CS with pupil dilation. In addition to iris CS area, they considered iris CS area/mm of pupil diameter change. They found that the iris loses almost half of its volume as the pupil increases from 3 to 7 mm in diameter. This was presumed to be due to loss of extracellular fluid. Smaller iris CS area change with physiologic pupil dilation was considered to be a possible risk factor for angle-closure. It was postulated that dynamic iris change deserves testing as a prospective indicator of angle-closure [67, 68]. Aptel has further shown that there is an increase in iris volume in the fellow eyes of patients who had a previous episode of acute angle-closure while the iris in open-angle control subjects lost a substantial amount of volume [69]. Quigley has also put forth a theory on the possible contribution of inappropriate choroidal expansion in eyes at risk for angle-closure. In an acute glaucoma attack, the IOP would immediately elevate with acute choroidal expansion. As aqueous leaves via the trabecular meshwork, the normal posterior chamber to anterior chamber pressure differential would significantly elevate. In susceptible eyes the iris could push forward and close the angle [70].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


