Purpose
To characterize the frequency of and clinical indications for which experts treat retinopathy of prematurity (ROP) milder than type 1 disease, the recommended threshold for treatment from established consensus guidelines.
Design
Descriptive analysis.
Methods
setting : Multicenter. study population : A database of 1444 eyes generated prospectively from all babies screened for ROP at 1 of 6 major ROP centers whose parents provided informed consent. intervention : Retrospective review of the database and charts to identify all patients treated for ROP milder than type 1. main outcome measure : Indication(s) for treatment.
Results
A total of 137 eyes of 70 infants were treated for ROP. Of these 137 eyes, 13 (9.5%) were treated despite a clinical diagnosis milder than type 1 ROP. Indications for treatment included active ROP with the fellow eye being treated for type 1 ROP (2 eyes, 15.4%); concerning structural changes (9 eyes, 69.2%), including tangential traction with temporal vessel straightening concerning for macular dragging (8 eyes, 61.5%) and thick stage 3 membranes with anteroposterior traction concerning for progression to stage 4 ROP (3 eyes, 23.1%); persistent ROP at an advanced postmenstrual age (4 eyes, 30.8%); and/or vitreous hemorrhage (3 eyes, 23.1%).
Conclusions
Experts in this study occasionally recommended treatment in eyes with disease less than type 1 ROP. This study has important clinical implications and highlights the role of individual clinical judgment in situations not covered by evidence-based treatment guidelines.
Retinopathy of prematurity (ROP) is a disorder of retinal vascular development in premature and low-birthweight infants that is a leading cause of childhood blindness in the United States and throughout the world. However, timely screening and treatment can dramatically improve outcomes in patients with ROP and prevent blindness in children with treatment-requiring disease. Close adherence to published consensus ROP screening and treatment guidelines has been recommended by experts not only to ensure evidence-based ROP care and improve outcomes, but also to reduce medicolegal liability. ROP is classified using a standard international scheme describing the stage of pathology, the zone of retina affected, and the presence or absence of plus disease.
Based on the multicenter Early Treatment for Retinopathy of Prematurity (ET-ROP) study, ROP is now classified as mild ROP; type 2 ROP, defined as zone I, stage 1 or 2 without plus disease or zone II, stage 3 without plus disease; or type 1 ROP, defined as zone I, stage 3; zone I, any stage with plus disease; or zone II, stage 2 or 3, with plus disease. In 2013, a consensus policy statement by the American Academy of Pediatrics, American Academy of Ophthalmology, and American Association for Pediatric Ophthalmology and Strabismus recommended treatment for type 1 or worse ROP, based on the ET-ROP study findings.
Despite these well-defined criteria for treatment, it has been our anecdotal experience that clinicians and ROP experts may occasionally recommend treatment for patients with ROP milder than type 1. This has important implications for the role of clinical judgment in individual cases not covered by evidence-based treatment guidelines. The purpose of this study is to characterize the frequency and nature of cases of ROP that were treated for disease milder than type 1 by retrospectively evaluating a large, prospectively collected, multicenter database of neonates screened for ROP.
Methods
This is a descriptive analysis from a prospectively collected multicenter i-ROP (Imaging & Informatics in ROP research consortium) database of all neonates screened for ROP at any of 6 major ROP centers in the United States whose parents provided informed written consent for inclusion in the database and associated studies. The study was performed in accordance with protocols approved by the Institutional Review Board of all institutions involved, and all analyses were performed in a fashion compliant with the Health Insurance Portability and Accountability Act.
Participating centers included Oregon Health & Science University, Weill Cornell Medical College, Bascom Palmer Eye Institute (University of Miami), Children’s Hospital Los Angeles, William Beaumont Hospital, and Columbia University Medical Center. Study examinations were performed between April 4, 2006 and January 21, 2015.
All children who were screened for ROP and whose parents consented for inclusion in the database were included in this study. Screening criteria were birth weight ≤1500 grams, gestational age at birth ≤30 weeks, or unstable clinical course or high risk of ROP as determined by the pediatrician or neonatologist. Clinical examinations were performed at the bedside by a retina specialist or pediatric ophthalmologist using indirect ophthalmoscopy. Wide-angle retinal images were also captured using a commercially available camera (RetCam; Clarity Medical Systems, Pleasanton, California, USA) if they were felt by the examiner to contribute diagnostic value. Posterior, temporal, nasal, superior, inferior, and iris images of each eye were taken, along with additional images as needed.
The i-ROP database was reviewed for all infants treated for ROP with either laser photocoagulation or intravitreal anti–vascular endothelial growth factor (VEGF) agent. Infants who were treated based on a clinical diagnosis milder than type 1 ROP were identified. The treating center was documented and is presented here in a coded format. Medical records of these subjects, including all examination and operative notes, were reviewed by a coauthor (M.P.G.) to confirm the clinical diagnosis and determine the treating clinician’s reasoning for treatment. Each case was subsequently reviewed by the study authors (M.F.C., R.V.P.C., M.P.G.). Indications for treatment for each case were reviewed and determined to fall into 1 or more of the following categories: (1) active ROP with fellow eye being treated for type 1 ROP; (2) concerning structural changes; (3) persistent active ROP at an advanced postmenstrual age; (4) vitreous hemorrhage.
When available, fundus photographs from the examination prior to treatment were reviewed and subjected to a previously reported method for generating a “reference standard” diagnosis. Briefly, the “reference standard” diagnosis was derived by review of fundus photographs by 3 readers experienced in image-based ROP diagnosis and incorporation of the clinical diagnosis by the examining clinician and arbitration as necessary when discrepancies between these 4 diagnoses arose. The “reference standard” diagnosis was evaluated as a quality check to assess likelihood of accurate diagnosis by the treating clinician. However, because this study sought to determine the treating clinician’s rationale for treatment, indications for treatment were determined strictly from the medical documentation and review of the case with the treating clinician, rather than our retrospective review of the imaging or the “reference standard” diagnosis.
Results
Description of Study Population
During the study period, there were 722 infants in the i-ROP database (1444 eyes, 4795 eye examinations). Within this study database, 137 eyes (9.5%) of 70 infants were treated for ROP with laser photocoagulation or intravitreal anti-VEGF agents. Of these 70 infants, 3 underwent unilateral treatment and 67 underwent bilateral treatment.
Indications for Treatment of Retinopathy of Prematurity Milder Than Type 1
Of 137 eyes treated for ROP, 13 eyes (9.5%) of 9 infants were treated despite a clinical diagnosis of less than type 1 disease ( Table ). These 9 cases were from 4 of the 6 ROP centers included in the database. All of these eyes were treated with laser photocoagulation. Of the 137 eyes treated, 134 were cases of bilateral treatment and 3 were cases of unilateral treatment—all 3 of the unilaterally treated eyes (Subjects 6, 8, and 9 in the Table ) were outliers. Indications for treatment are described below. Only a few cases are described in detail below in order to describe a representative case for each treatment indication. Details for all cases studied are included in the Table . Of note, some eyes were treated for more than 1 indication.
| Subject: Eye | Center (Coded) | Gestational Age at Birth (Weeks) | Birth Weight (Grams) | Gestational Age at Time of Treatment (Weeks) | Clinical Diagnosis | “Reference Standard” Diagnosis (When Available) | Indication(s) for Treatment |
|---|---|---|---|---|---|---|---|
| 1: OS | A | 25 | 600 | 42 | Zone II, stage 2, pre-plus (mild ROP) | Zone II, stage 2, no plus (mild ROP) | ▪ Active ROP with fellow eye being treated for type 1 ROP |
| 2: OS | A | 25 | 740 | 39 | Zone II, stage 2, no plus (mild ROP) | Zone II, stage 2, pre-plus (mild ROP) | ▪ Active ROP with fellow eye being treated for type 1 ROP |
| 3: OD | B | 27 | 990 | 47 | Zone II, stage 3, no plus (type 2 ROP) | Zone II, stage 3, pre plus (type 2 ROP) | ▪ Concerning structural changes (tangential traction with temporal vessel straightening) |
| 3: OS | B | 27 | 990 | 47 | Zone II, stage 3, no plus (type 2 ROP) | Zone II, stage 3, pre-plus (type 2 ROP) | ▪ Concerning structural changes (tangential traction with temporal vessel straightening) |
| 4: OD | C | 23 | 534 | 38 | Zone II, stage 3, no plus (type 2 ROP) | N/A | ▪ Concerning structural changes (tangential traction with temporal vessel straightening) |
| 4: OS | C | 23 | 534 | 38 | Zone II, stage 3, no plus (type 2 ROP) | N/A | ▪ Concerning structural changes (tangential traction with temporal vessel straightening) |
| 5: OD | B | 23 | 510 | 40 | Zone II, stage 3, pre-plus (type 2 ROP) | N/A | ▪ Concerning structural changes (tangential traction with temporal vessel straightening) ▪ Vitreous hemorrhage |
| 5: OS | B | 23 | 510 | 40 | Zone II, stage 3, pre-plus (type 2 ROP) | N/A | ▪ Concerning structural changes (tangential traction with temporal vessel straightening) ▪ Vitreous hemorrhage |
| 6: OS | D | 26 | 950 | 42 | Zone II, stage 3, no plus (type 2 ROP) | Zone II, stage 3, no plus (type 2 ROP) | ▪ Concerning structural changes (thick stage 3 membrane with anteroposterior traction and concern for progression to stage 4) ▪ Vitreous hemorrhage |
| 7: OD | B | 25 | 840 | 41 | Zone II, stage 3, no plus (type 2 ROP) | Zone II, stage 3, pre-plus (type 2 ROP) | ▪ Concerning structural changes (thick stage 3 membrane with anteroposterior traction and concern for progression to stage 4) ▪ Concerning structural changes (tangential traction with temporal vessel straightening) ▪ Persistent active ROP at an advanced postmenstrual age (42 weeks) |
| 7: OS | B | 25 | 840 | 41 | Zone II, stage 3, no plus (type 2 ROP) | Zone II, stage 3, pre-plus (type 2 ROP) | ▪ Concerning structural changes (thick stage 3 membrane with anteroposterior traction and concern for progression to stage 4) ▪ Concerning structural changes (tangential traction with temporal vessel straightening) ▪ Persistent active ROP at an advanced postmenstrual age (42 weeks) |
| 8: OD | C | 26 | 1090 | 43 | Zone III, stage 3, no plus (mild ROP) | N/A | ▪ Persistent active ROP at an advanced postmenstrual age |
| 9: OS | B | 28 | 1077 | N/A (12 months of age) | Zone III, stage 1, no plus; peripheral ischemia on fluorescein angiography (mild ROP) | Zone II, stage 2, no plus (mild ROP) | ▪ Persistent retinal ischemia at old age (12 months) with treatment-requiring disease in the other eye |
Active Retinopathy of Prematurity With Fellow Eye Being Treated for Type 1 Retinopathy of Prematurity
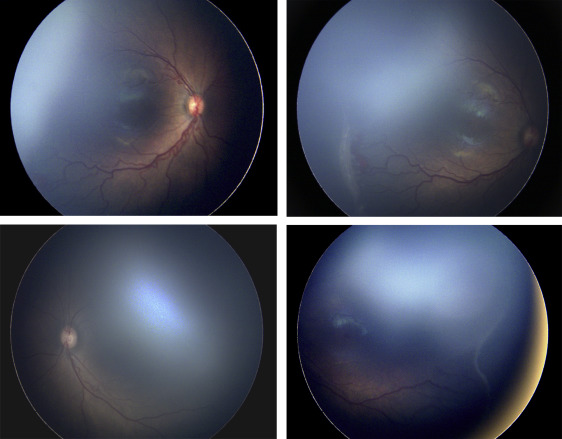
Two of the 13 eyes (15.4%) were treated for active type 2 or less ROP in the setting of the fellow eye being treated for type 1 ROP. Of the 137 treated eyes evaluated in this study, these 2 cases were the only ones in which only 1 of the 2 eyes met treatment guidelines, and both of these were treated in a single session, bilaterally. Subject 1, for example, was born at 25 weeks gestational age, with birth weight 600 grams. Examination at 42 weeks postmenstrual age revealed zone II, stage 3 with plus disease in the right eye (OD) ( Figure 1 , Top left and Top right) and zone II, stage 2 with pre-plus disease in the left eye (OS) ( Figure 1 , Bottom left and Bottom right). Given the presence of type 1 disease OD meeting guidelines for treatment, the option of treatment OS at the same time vs close observation OS was discussed with the parents. Informed consent was obtained, and laser photocoagulation was performed in both eyes (OU).
Concerning Structural Changes
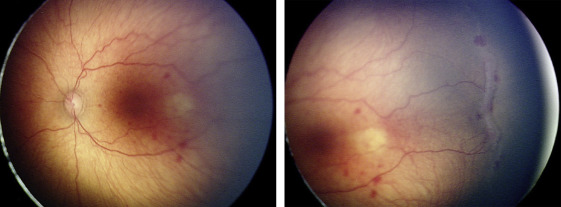
Nine of the 13 eyes (69.2%) were treated for concerning structural changes. Of these, 8 eyes (61.5%) were treated for tangential traction with temporal vessel straightening concerning for macular distortion and dragging. Subject 3, for example, was born at 27 weeks gestational age, with birth weight 990 grams. At 47 weeks postmenstrual age, examination revealed zone II, stage 3, no plus disease with temporal vascular straightening OU ( Figure 2 ) concerning for future macular distortion and dragging. The option of laser vs close observation was discussed with the parents. Informed consent was obtained, and laser photocoagulation was performed OU.
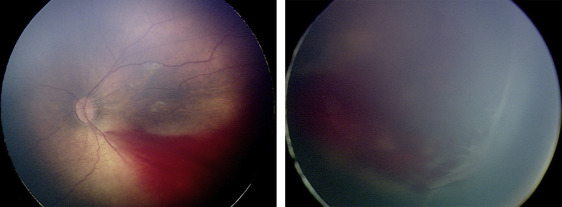
Three eyes (23.1%) were treated for thick stage 3 membranes with anteroposterior traction concerning for progression to stage 4 ROP. Subject 6, for example, was born at 26 weeks gestational age, with birth weight 950 grams. At 42 weeks postmenstrual age, examination revealed zone II, stage 3, no plus disease with a thick stage 3 fibrovascular membrane OS causing significant anteroposterior traction concerning for progression to stage 4, as well as vitreous hemorrhage ( Figure 3 ). Owing to these concerning structural changes, as well as concern for further vitreous hemorrhage limiting visualization and/or treatment in the future (see below), the option of laser vs observation OS was discussed with the patient’s parents. Informed consent was obtained, and laser photocoagulation was performed OS.
Persistent Active Retinopathy of Prematurity at an Advanced Postmenstrual Age
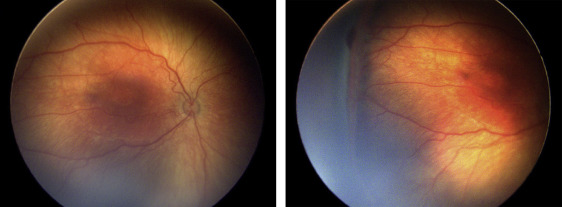
Four of the 13 eyes (30.8%) were treated for persistent active ROP at an advanced postmenstrual age. Of these, 3 eyes (23.1%) were noted to have persistent active stage 3 ROP at a postmenstrual age >41 weeks. Subject 7 was born at 25 weeks gestational age, with birth weight 840 grams. At 41 weeks postmenstrual age, the examination revealed zone II, stage 3 disease without plus OU ( Figure 4 ). There was concern for the degree of active stage 3 ROP, given the patient’s advanced postmenstrual age. Moreover, concerning structural changes were noted (as described above), including temporal vessel straightening and a thick neovascular ridge with significant anteroposterior vitreoretinal traction concerning for progression to stage 4 disease. Thus, the option of laser vs close observation was discussed with the parents. Informed consent was obtained, and laser photocoagulation was administered OU.
Subject 9 was born at 28 weeks gestational age weighting 1077 grams. At initial presentation at 5 months of age, examination revealed stage 5 ROP OD and zone II, stage 2 disease without plus disease OS. The patient underwent vitrectomy OD, but vision subsequently reached no light perception OD. At 12 months of age, there was persistent ROP with zone III, stage 1 ROP without plus disease OS and peripheral ischemia on fluorescein angiography. Given persistent ROP at such a late stage in this monocular patient with history of stage 5 ROP in the fellow eye, the option of laser vs close observation OS was discussed with the parents. Informed consent was obtained, and laser photocoagulation was applied OS.
Vitreous Hemorrhage
Three of the 13 eyes (23.1%) were treated because of vitreous hemorrhage. Of note, each of these eyes also demonstrated other concerning structural features that also contributed to the decision to treat ( Table ). As detailed above, 1 eye (Subject 6) also had thick stage 3 membrane OS with anteroposterior traction concerning for progression to stage 4 ( Figure 3 ), while 2 eyes (Subject 5) also exhibited temporal vessel dragging and tangential traction on the retina.
Comparison of Clinical Diagnosis to “Reference Standard” Diagnosis
Fundus photographs were available for 8 of 13 eyes that underwent treatment for clinical diagnosis of ROP milder than type 1 ROP. The “reference standard” diagnosis of all 8 eyes matched the clinical diagnosis with regard to overall disease category (type 1, type 2, or mild ROP). The “reference standard” diagnosis eyes matched the clinician’s diagnosis for zone, stage, and presence of plus disease in 7 of 8 eyes ( Table ).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


