Purpose
To compare the late complications in the Ahmed Baerveldt Comparison Study during 5 years of follow-up.
Design
Multicenter, prospective randomized clinical trial.
Methods
setting : Sixteen international clinical centers. study population : Two hundred seventy-six subjects aged 18-85 years with previous intraocular surgery or refractory glaucoma with intraocular pressure of >18 mm Hg. interventions : Ahmed Glaucoma Valve FP7 or Baerveldt Glaucoma Implant BG 101-350. main outcome measures : Late postoperative complications (beyond 3 months), reoperations for complications, and decreased vision from complications.
Results
Late complications developed in 56 subjects (46.8 ± 4.8 5-year cumulative % ± SE) in the Ahmed Glaucoma Valve group and 67 (56.3 ± 4.7 5-year cumulative % ± SE) in the Baerveldt Glaucoma Implant group ( P = .082). The cumulative rates of serious complications were 15.9% and 24.7% in the Ahmed Glaucoma Valve and Baerveldt Glaucoma Implant groups, respectively ( P = .034), although this was largely driven by subjects who had tube occlusions in the 2 groups (0.8% in the Ahmed Glaucoma Valve group and 5.7% in the Baerveldt Glaucoma Implant group, P = .037). Both groups had a relatively high incidence of persistent diplopia (12%) and corneal edema (20%), although half of the corneal edema cases were likely due to pre-existing causes other than the aqueous shunt. The incidence of tube erosion was 1% and 3% in the Ahmed Glaucoma Valve and Baerveldt Glaucoma Implant groups, respectively ( P = .04).
Conclusions
Long-term rates of vision-threatening complications and complications resulting in reoperation were higher in the Baerveldt Glaucoma Implant than in the Ahmed Glaucoma Valve group over 5 years of follow-up.
The use of aqueous shunts for surgical glaucoma management has increased dramatically in the past 20 years. In a recent analysis of the US Medicare database, the number of aqueous shunt implantations performed annually has risen from 2356 in 1994 to 12,021 in 2012. There are numerous aqueous shunts in use but, at least in the US market, 2 models are used most commonly, the Ahmed Valve model FP7 (New World Medical, Rancho Cucamonga, California, USA) and the Baerveldt BG 101-350 (Abbott Medical Optics, Abbott Park, Illinois, USA). A 2008 survey of the American Glaucoma Society membership showed that approximately half of respondents favored the Ahmed Glaucoma Valve and half preferred the Baerveldt Glaucoma Implant when operating on patients with previous incisional eye surgery or refractory glaucoma. In that same year, the American Academy of Ophthalmology published a Technology Assessment article reviewing aqueous shunts and concluded that “Too few high-quality direct comparisons of various available shunts have been published to assess the relative efficacy or complication rates of specific devices….” While there have been several retrospective studies comparing the Ahmed and Baerveldt implants, these have been of relatively short duration and inconclusive as to the relative success rates and complications of these 2 implants. In addition, these earlier studies used older models with different materials and designs, which have been improved upon to address prior concerns. Lastly, these studies suffer from the selection bias inherent to all retrospective analyses of existing medical record data.
The Ahmed Baerveldt Comparison (ABC) Study was a prospective multicenter randomized surgical trial that compared the safety and efficacy of the Ahmed Glaucoma Valve FP7 and Baerveldt Glaucoma Implant BG 101-350 in patients with previous incisional eye surgery or refractory glaucoma. These 2 implants have markedly different design features. The Ahmed Glaucoma Valve FP7 is a valved implant with a 184 mm 2 endplate and the Baerveldt Glaucoma Implant BG 101-350 is a nonvalved implant with a 350 mm 2 endplate. At 5 years, the Baerveldt Glaucoma Implant group had an average intraocular pressure (IOP) that was 2 mm Hg lower than the Ahmed Glaucoma Valve FP7 group and, at most time points, subjects in the Baerveldt Glaucoma Implant BG 101-350 group were, on average, taking fewer medications than the Ahmed Glaucoma Valve FP7 group. Over the 5 years of follow-up, the 2 treatment groups experienced failures at the same rate, approximately 8% per year, but failures in the 2 groups occurred for different reasons. Subjects in the Ahmed Glaucoma Valve FP7 group experienced failures more often than the Baerveldt Glaucoma Implant BG 101-350 group owing to high IOP endpoints (persistently elevated IOP and reoperation for IOP elevation), while implants in the Baerveldt Glaucoma Implant BG 101-350 group failed more often than the Ahmed Glaucoma Valve FP7 group because of safety endpoints such as persistent hypotony or loss of light perception vision.
However, in any surgical comparison study, it is important to weigh the relative efficacy against the relative risks of each procedure. The intraoperative and early postoperative complications (within 3 months of surgery) in the ABC Study were reported along with the 1-year outcomes. The purpose of the current study was to examine the long-term (5-year) complications of the Ahmed FP7 and Baerveldt BG 101-350 implants in subjects with prior incisional surgery or refractory glaucoma.
Methods
This prospective randomized clinical trial was approved at the Institutional Review Boards at 16 clinical centers and each patient gave informed consent. The study was registered at www.clinicaltrials.gov (identifier NCT00376363 ). The design and methods of the ABC Study are described in detail in the baseline methodology paper, and are summarized as follows.
Randomization, Eligibility, and Treatment
Subjects aged 18-85 years with refractory glaucoma and IOPs greater than or equal to 18 mm Hg in whom aqueous shunt surgery was planned were enrolled in the study. Subjects with primary glaucomas with a previous failed trabeculectomy or who had previous intraocular surgery were included. Also, subjects without previous intraocular surgery were eligible if they had secondary glaucomas known to be refractory to trabeculectomy, such as neovascular glaucoma (NVG), uveitic glaucoma, or glaucoma associated with iridocorneal endothelialization (ICE) syndrome.
Individuals enrolled in the study were randomized in a 1:1 ratio to placement of an Ahmed Glaucoma Valve FP7 or Baerveldt Glaucoma Implant BG 101-350 according to a permuted variable block randomization scheme, stratified by surgeon within 1 of 16 clinical centers and type of glaucoma. Subjects were allocated to 1 of 4 strata according to their type of glaucoma, as follows: (1) primary glaucomas with previous intraocular surgery; (2) secondary glaucomas (excluding uveitic glaucoma and NVG); (3) NVG; and (4) uveitic glaucoma. Subjects were excluded if they lacked light perception vision, were unwilling or unable to give informed consent, lived out of the area and were expected to be unavailable for follow-up visits, underwent a previous cyclodestructive procedure or previous aqueous shunt implanted in the same eye, underwent a prior scleral buckling procedure or other external impediment to superotemporal device implantation, had silicone oil in the eye, had vitreous in the anterior chamber sufficient to require a vitrectomy, had uveitis associated with a systemic condition like juvenile rheumatoid arthritis, had nanophthalmos, had Sturge-Weber syndrome or other conditions associated with elevated episcleral venous pressure, or needed aqueous shunt surgery combined with other ocular procedures. For subjects in whom both eyes were eligible for enrollment, only the first eligible eye to be implanted was enrolled. Neither the subject nor the investigator was masked to the randomization assignment. Details of the surgical procedures for Ahmed Glaucoma Valve and Baerveldt Glaucoma Implant BG 101-350 implantation used in this study are described in the baseline paper.
Patient Visits
Follow-up visits were scheduled 1 day, 1 week, 1 month, 3 months, 6 months, 1 year, 18 months, 2 years, 3 years, 4 years, and 5 years postoperatively. Detailed information about data obtained at baseline and follow-up visits is contained in the baseline paper.
Postoperative Interventions
At each follow-up visit, investigators were asked about interventions performed since the subject’s last visit. There were specific questions about whether anterior chamber reformation or intravitreal injections had been performed, as well as an open-ended “Other Interventions” category. Postoperative interventions were counted as such in the analysis if the intervention was deemed related to the original surgery, needed for further IOP lowering but not incisional surgery (such as laser trabeculoplasty), or needed for a complication of the surgery.
Definition of Complications
The current analysis only includes reoperations or loss of vision if they were attributable to the aqueous shunt surgery. Early complications were those that were recorded by the 3-month follow-up visit. These were reported in the 1-year outcomes paper. Late complications were those that were experienced after the 3-month follow-up visit. A serious complication was defined as any complication, early or late, that was associated with a 2-line Snellen acuity decrease or a return to the operating room for a surgical procedure to manage the complication. A revision in the operating room to manage an occluded tube was considered a reoperation for a complication. The Snellen visual acuity (VA) decrease was assessed at the 5-year visit. If the patient did not have a 5-year visit, then the patient’s complication could not be categorized as serious by vision loss, but could by virtue of reoperation.
Statistical Analysis
Univariate comparisons between treatment groups were made using the 2-sided Student t test, χ 2 test, or Fisher exact test. Subjects’ data were analyzed in the group to which they were assigned during randomization (intent-to-treat analysis). A P value of .05 or less was considered statistically significant in our analyses.
Results
A total of 276 subjects were enrolled between October 2006 and April 2008. One hundred forty-three subjects (52%) were randomly assigned to placement of an Ahmed Glaucoma Valve FP7 and 133 (48%) to a Baerveldt Glaucoma Implant BG 101-350. The disposition of subjects is summarized in the Consort Flow Diagram ( Supplemental Figure available at AJO.com ). Intraoperative complications, early postoperative complications, and visual acuity results have been described in detail previously.
Postoperative Interventions
Table 1 lists postoperative interventions that occurred over 5 years of follow-up, excluding those included in the1-year report. The total number of subjects requiring interventions beyond 1 year was 16 in the Ahmed Glaucoma Valve FP7 group and 25 in the Baerveldt Glaucoma Implant BG 101-350 group, a difference that was not statistically significant ( P = .21, Fisher exact test). Excluding cataract extraction (detailed below), there were only 7 surgical interventions needed between 3 and 5 years of follow-up.
| Intervention | Ahmed (N = 143) | Baerveldt (N = 133) |
|---|---|---|
| Anterior chamber reformation | 0 | 1 (1%) |
| Intravitreal injection | 0 | 2 (2%) |
| Needling at slit lamp | 1 (1%) | 0 |
| Macular laser for CME | 1 (1) | 0 |
| Laser trabeculoplasty | 0 | 1 (1%) |
| Corneal scraping for band keratopathy | 0 | 1 (1%) |
| Total number of patients with interventions | 2 (1.4%) | 5 (3.8%) |
Late Postoperative Complications
Table 2 details the cumulative 5-year incidence of late (after 3 months) complications by randomized treatment group. Late complications developed in 56 subjects (46.8 ± 4.8 5-year cumulative % ± standard error [SE]) in the Ahmed Glaucoma Valve group and 67 subjects (56.3 ± 4.7 5-year cumulative % ± SE) in the Baerveldt Glaucoma Implant BG 101-350 group during 5 years of follow-up ( P = .082). The overall incidence of late postoperative complications was similar between treatment groups.
| Complication | Year 5 N, Cumulative Proportion (SE) | Log-Rank P Value | Risk Ratio a | 95% CI | |
|---|---|---|---|---|---|
| Ahmed | Baerveldt | ||||
| Tube occlusion | 1, 0.8% (0.8%) | 6, 5.7% (2.3%) | .037 | 6.93 | 0.83, 57.5 |
| Choroidal effusion | 0, 0.0% NA | 2, 1.8% (1.2%) | .16 | NA | NA |
| Endophthalmitis | 0, 0.0% NA | 2, 2.2% (1.6%) | .16 | NA | NA |
| Cystoid macular edema | 6, 6.2% (2.5%) | 7, 7.2% (2.7%) | .81 | 1.14 | 0.38, 3.40 |
| Shallow anterior chamber | 2, 2.2% (1.5%) | 3, 3.7% (2.1%) | .64 | 1.53 | 0.26, 9.16 |
| Hypotony maculopathy | 0, 0.0% NA | 1, 0.8% (0.8%) | .30 | NA | NA |
| Diplopia | 16, 12.7% (3.0%) | 14, 11.8% (3.0%) | .81 | 0.92 | 0.45, 1.88 |
| Corneal edema-all | 18, 20.1% (4.4%) | 18, 20.4% (4.3%) | .71 | 1.13 | 0.59, 2.18 |
| Corneal edema-likely attributable to implant | 9, 11.9% (3.8%) | 9, 11.7% (3.7%) | .82 | 1.10 | 0.49, 2.44 |
| Tube-corneal touch | 4, 3.5% (1.7%) | 4, 3.7% (1.8%) | .91 | 1.08 | 0.27, 4.34 |
| Corneal graft rejection | 8, 7.1 (2.4%) | 8, 7.0% (2.4%) | .96 | 0.98 b | 0.37, 2.62 |
| Band keratopathy | 1, 1.2% (1.2%) | 2, 2.0% (1.4%) | .57 | 1.99 | 0.18, 21.9 |
| Corneal neovascularization | 0, 0.0% NA | 1, 1.0% (1.0%) | .33 | NA | NA |
| Tube erosion | 3, 2.9% (1.7%) | 1, 1.0% (1.0%) | .33 | 0.34 | 0.04, 3.31 |
| Encysted bleb | 1, 0.9% (0.9%) | 0, 0.0% NA | .32 | NA | NA |
| Recurrent or persistent iritis | 7, 6.2% (2.3%) | 6 5.5% (2.2%) | .83 | 0.89 | 0.30, 2.63 |
| Phthisis bulbi | 1, 0.8% NA | 6, 5.7% (2.3%) | .037 | 6.93 | 0.83, 57.5 |
| Hyphema | 2, 1.5% (1.1%) | 2, 1.6% (1.1%) | .97 | 1.04 | 0.15, 7.40 |
| Vitreous hemorrhage | 3, 2.7% (1.5%) | 3, 2.5% (1.4%) | .96 | 1.04 | 0.21, 5.17 |
| Pupillary membrane | 1, 0.8% (0.8%) | 0, 0.0% NA | .33 | NA | NA |
| Epiretinal membrane | 0, 0.0% NA | 1, 0.8% (0.8) | .31 | NA | NA |
| Retinal detachment | 2, 1.6% (1.1%) | 2, 1.7% (1.2%) | .98 | 1.02 | 0.14, 7.25 |
| Corneal blood staining | 0, 0.0% NA | 2, 1.6% (1.1%) | .15 | NA | NA |
a Ahmed is the reference group.
b Adjusted for previous PKP prior to enrollment and implantation of study GDI, which was itself highly significantly related to corneal graft failure ( P < .001).
Tube occlusion ( P = .037, Fisher exact test) and phthisis bulbi ( P = .037, Fisher exact test) were late postoperative complications that occurred with significantly greater frequency in the Baerveldt Glaucoma Implant BG 101-350 group than in the Ahmed Glaucoma Valve group. The issue of tube occlusion was discussed in the 1-year outcomes paper and there were no additional tube occlusions beyond the 1 year time point. Phthisis bulbi was found in 6 subjects in the Baerveldt Glaucoma Implant BG 101-350 group compared to 1 subject in the Ahmed Glaucoma Valve FP7 group. These same subjects were counted as failures owing to persistent hypotony attributable to severe vision loss in all of these subjects.
Persistent corneal edema was found in 18 of 87 (20.1%) and 18 of 87 (20.4%) of subjects in the Ahmed Glaucoma Valve FP7and Baerveldt Glaucoma Implant BG 101-350 groups, respectively, although this was attributed to nonimplant causes in 50% of these, so the percentage of subjects with persistent corneal edema attributable to the aqueous shunt was closer to 12%. Diplopia was found in 12.7% and 11.8% of subjects in the Ahmed Glaucoma Valve FP7 and Baerveldt Glaucoma Implant BG 101-350 groups, respectively, and there was no difference between the 2 groups in the incidence of diplopia. Cystoid macular edema was the third most frequent complication, occurring in 6.2% of subjects in the Ahmed Glaucoma Valve group and 7.2% of subjects in the Baerveldt Glaucoma Implant BG 101-350 group. It was difficult to determine whether this was surgically related or caused by underlying conditions such as diabetic retinopathy, uveitis, or neovascular glaucoma, all of which were common in this cohort and are also associated with cystoid macular edema.
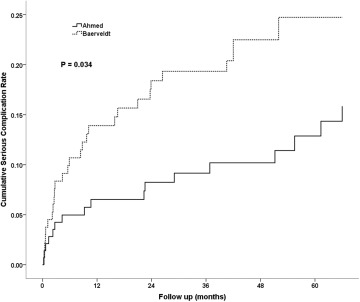
Serious Complications
Table 3 shows serious complications resulting in reoperation and/or vision loss. Complications were classified as serious either if they were associated with reoperation for complication or if they were associated with a Snellen acuity loss of 2 or more lines at their last study visit that could be attributed to a complication of tube implantation. The incidence of serious complications was higher in the Baerveldt Glaucoma Implant BG 101-350 group. Serious complications were observed in 17 (15.9%) subjects in the Ahmed Glaucoma Valve group and 29 (24.7%) subjects in the Baerveldt Glaucoma Implant BG 101-350 group ( P = .034, log-rank test adjusted for stratum). Figure 1 shows the cumulative rates of serious complications for each group. Persistent corneal edema was the most common cause for both reoperation for a complication and loss of 2 or more lines of Snellen VA in both groups ( Table 3 ). As reported previously, approximately 40% of subjects in both groups lost 2 or more lines of Snellen VA after 5 years, but the vast majority of these were attributable to their underlying ocular disease rather than to the aqueous shunt surgery. Sixteen subjects in the Ahmed Glaucoma Valve group and 24 subjects in the Baerveldt Glaucoma Implant BG 101-350 group underwent reoperations for complications. A total of 22 eyes experienced complications during follow-up resulting in the loss of 2 or more lines of Snellen VA. The study principal investigator (D.L.B.) reviewed all of these, masked to randomized treatment assignment and the first 3 months of follow-up (during which eyes implanted with Baerveldt tubes could be expected to have high IOP), and attributed the acuity loss to glaucoma progression (n = 4; Ahmed Glaucoma Valve FP7:3, Baerveldt Glaucoma Implant BG 101-350:1), progressive retinal disease (n = 4; Ahmed Glaucoma Valve FP7:2, Baerveldt Glaucoma Implant BG 101-350:2), and other causes (n = 7; Ahmed Glaucoma Valve FP7:2, Baerveldt Glaucoma Implant BG 101-350:5; in 1 case the cause could not be determined as follow-up information was obtained from a non-study ophthalmologist). The other causes of acuity loss not attributed to the GDI implantation included corneal decomposition secondary to ICE (n = 2), corneal epitheliopathy secondary to dry eye (n = 1), pre-existing corneal disease (n = 2), posterior corneal opacification (n = 1), and aphakia (n = 1).
| Ahmed Group (N = 143) | Baerveldt Group (N = 133) | |
|---|---|---|
| Reoperation for complications | 16 (14.3%) | 24 (19.5%) |
| Vision loss of ≥2 Snellen lines | ||
| Persistent corneal edema | 1 | 1 |
| Persistent corneal edema + hypotony maculopathy | 0 | 1 |
| Persistent corneal edema + tube-corneal touch | 0 | 2 |
| Cystoid macular edema | 0 | 1 |
| Total number of subjects with serious complications a | 17 (15.9%) | 29 (24.7%) |
a P = .034 for the difference in 5-year cumulative serious complication rates between treatment groups from Kaplan-Meier analysis (log-rank test adjusted for stratum).
Reoperation for Complications
At year 5, the cumulative proportion (SE) of subjects with complication-related reoperations, including explantations, in the Ahmed Glaucoma Valve group was 14.3% (3.5%), n = 16, compared to 19.5% (3.6%), n = 24, in the Baerveldt Glaucoma Implant BG 101-350 group ( P = .109, log-rank test). Figure 2 shows the cumulative proportion of subjects in each group who required reoperation for a complication throughout 5 years of follow-up. The risk ratio was 0.60 with 95% confidence interval 0.32–1.13 (Cox proportional hazard regression), so a clinically significant difference in favor of Ahmed Glaucoma Valve implantation having a risk that was one-third that of Baerveldt Glaucoma Implant BG 101-350 implantation with respect to complication reoperations cannot be excluded. There was no significant difference by stratum ( P = .98, Cox regression accounting for randomized treatment group), nor was there a significant interaction between randomized treatment group and stratum ( P = .38, cox regression). This implies that diagnostic stratum, particularly neovascular glaucoma, did not affect the likelihood of having a reoperation for complications. After 5 years of follow-up there were no significant differences (all P > .1) in rates of late-onset complications between clinical centers or between surgeons more experienced with the randomized aqueous shunt (≥20 prior implantations) compared to those with less experience.




