Posterior Lamellar Endothelial Keratoplasty
Nicoletta Fynn-Thompson
Dimitri T. Azar
Posterior lamellar endothelial keratoplasty refers to the removal of the diseased posterior corneal layers and their replacement by partial-thickness donor tissue. A clear advantage of this form of lamellar keratoplasty over penetrating keratoplasty is that it replaces only the diseased portion of the cornea, leaving the uninvolved anatomic layers of the recipient cornea intact.
Anterior lamellar keratoplasty is discussed in another chapter. It is used for tectonic support of the globe in patients with corneal thinning disorders (such as peripheral ulcerative keratitis, Mooren’s ulceration, and Terrien’s marginal degeneration) and for replacement of anterior stromal tissue, caused by such conditions as keratoconus, corneal stromal dystrophies, and stromal scarring after resolved corneal ulcers. The recipient’s Descemet’s membrane and endothelium are in retained anterior lamellar keratoplasty (1).
Posterior lamellar keratoplasty is performed when the corneal endothelium is not functioning properly, such as in cases of Fuchs’ endothelial dystrophy and aphakic and pseudophakic bullous keratopathy. Theoretically, lamellar keratoplasty reduces the amount of corneal resection and provides greater visual benefit.
Compared with full-thickness penetrating keratoplasty, there are several theoretical advantages to performing posterior lamellar keratoplasty. These include lower risk of intraoperative complications and astigmatism, faster visual recovery, less frequent follow-up visits for suture adjustment, elimination of suture-induced corneal neovascularization, less risk of wound dehiscence, and fewer side effects of topical steroid therapy. In addition, donor tissue may be used more efficiently, for example by using the remaining tissue for the management of complicated photorefractive keratectomy (2).
There are drawbacks to posterior lamellar keratoplasty. The surgical techniques may be more demanding than penetrating keratoplasty. Meticulous dissection of the corneal lamellae is required. Also, perforation may occur. In this situation, the procedure may have to be converted to penetrating keratoplasty. In addition, patients with postoperative astigmatism cannot benefit from suture adjustment, as used in penetrating keratoplasty. There is also a risk of interface haze, which occurs in varying degrees (3).
SURGICAL TECHNIQUES
Posterior lamellar endothelial keratoplasty is used for corneal decompensation secondary to endothelial dysfunction. There are currently two surgical approaches: the scleral incision approach and the microkeratome flap approach. The first involves posterior corneal disk transplantation by scleral incision and trephination using an intrastromal trephine. The second approach uses an automated microkeratome to create a hinged anterior stromal flap and trephination of the diseased posterior stroma using a standard trephine (4).
Scleral Incision Approach
Early reports and the surgical technique for this approach for posterior lamellar keratoplasty were described in patients with Fuchs’ dystrophy or deep stromal opacities (5,6). This surgical technique was slightly altered by Melles et al. (7) for transplantation of posterior corneal tissue through a scleral incision and deep stromal pocket. It was initially performed on cadaveric human eyes and in vivo animal models. Melles and colleagues used samesize recipient cornea and donor disk in a sutureless technique. Postoperative examination revealed well-positioned posterior implants, and complete apposition of host and donor stromal edges in the cadaveric specimens. They later reported treating a total of eight patients with sighted eyes suffering from Fuchs’ endothelial dystrophy or bullous keratopathy (pseudophakic or aphakic) (8,9). Terry et al. introduced the Melles technique to the United States and described the results in the first two
U.S. patients who had endothelial dysfunction from Fuchs’ endothelial dystrophy (10).
U.S. patients who had endothelial dysfunction from Fuchs’ endothelial dystrophy (10).
Melles’ Original Surgical Technique
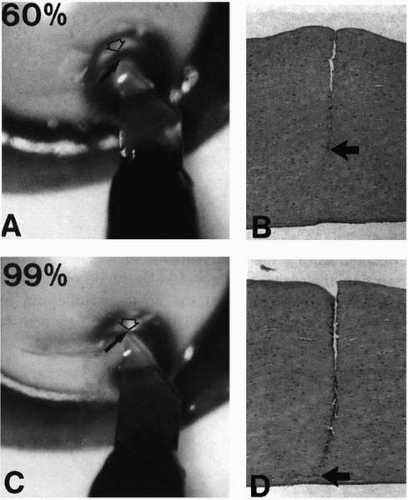
The donor eye is mounted on an eye holder (8). Through a paracentesis site, the anterior chamber is completely filled with air. A 4-mm peripheral corneal incision is made. A stromal pocket is dissected with a custom-made spatula blade (D.O.R.C. International, Zuidland, the Netherlands) across the cornea at 60% stromal depth using the air-to-endothelial interface as a guide for dissection depth (11). In the mirror effect, the air bubble in the anterior chamber acts as a convex mirror, such that a blade held at the anterior corneal surface is reflected at the posterior corneal surface (Fig. 63-1). The indentation effect describes the specular light reflex created near the tip of the blade by the indentation of the tissue. The amount of nonincised tissue between the light reflex and blade is seen as a dark band (Fig. 63-2). The folding effect describes the increased number in small folds that develop in the posterior corneal tissue as the blade approaches the posterior surface (Fig. 63-3). Once the stromal pocket is dissected, a plastic strip is inserted into the
pocket and the corneoscleral rim is excised and mounted endothelial side up on a punch block (Medical Workshop, De Meern, the Netherlands). A full-thickness corneal button is excised with a 7- to 7.5-mm punch trephine (Ophtec, Groningen, the Netherlands). The donor button is placed endothelial side down onto a custom-made spoon-shaped glide (D.O.R.C. International). It is covered with viscoelastic substance [hydroxypropylmethylcellulose (Ocucoat); Storz, Clearwater, FL]. The anterior lamella and plastic glide are then removed (Fig. 63-4).
pocket and the corneoscleral rim is excised and mounted endothelial side up on a punch block (Medical Workshop, De Meern, the Netherlands). A full-thickness corneal button is excised with a 7- to 7.5-mm punch trephine (Ophtec, Groningen, the Netherlands). The donor button is placed endothelial side down onto a custom-made spoon-shaped glide (D.O.R.C. International). It is covered with viscoelastic substance [hydroxypropylmethylcellulose (Ocucoat); Storz, Clearwater, FL]. The anterior lamella and plastic glide are then removed (Fig. 63-4).
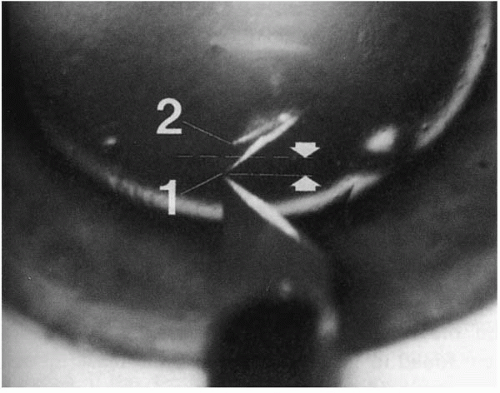 FIGURE 63-1. Mirror effect. In an eye that has the anterior chamber completely filled with air, two mirror images of the blade are visible, one a reflection from the anterior corneal surface [1] and one from the posterior surface [2]. The dotted line indicates the approximate location of the plane of reflection (i.e., the posterior surface). Thus, the estimated corneal thickness (arrows) is half the distance between the blade tip and its reflection from the posterior corneal surface [2]. (From Melles GRJ, Rietveld FJR, Beekhuis WH, et al. A technique to visualize corneal incision and lamellar dissection depth during surgery. Cornea 1999;18:80-86.) |
 FIGURE 63-3. Folding effect. Small folds (arrows) in the posterior corneal tissue are visible adjacent to the tip of a dissection blade that approaches the posterior corneal surface. (From Melles GRJ, Rietveld FJR, Beekhuis WH, et al. A technique to visualize corneal incision and lamellar dissection depth during surgery. Cornea 1999;18:80-86.)(see color image) |
The recipient eye’s anterior chamber is completely filled with air through a paracentesis site. A 9-mm partial-thickness scleral incision is made after the superior conjunctiva is opened. A stromal pocket is dissected across the cornea at 80% depth with a dissection spatula, using the air-to-endothelium interface (11). A custom-made 7- to 7.5-mm diameter flat trephine (D.O.R.C. International) is inserted into the stromal pocket and the posterior lamellar disk is excised. Remaining attachments of the corneal tissue may be removed with custom-made microscissors (D.O.R.C. International; Fig. 63-5). The spoon-shaped glide containing the “same-size” donor posterior disk is introduced into the recipient stromal pocket and positioned into the recipient’s posterior opening. The scleral incision is closed with 10-0 monofilament nylon sutures (Alcon, Gorinchem, the Netherlands; Fig. 63-5).
Deep Lamellar Endothelial Keratoplasty
Terry et al. (12) described his technique of deep lamellar endothelial keratoplasty for the treatment of endothelial dysfunction. This procedure is based on the original technique of Melles et al. Refinements on this technique continue to evolve including the small incision approach and the use of the femtosecond laser. Hence we describe the standard technique of DLEK (12). A 9-mm limbal-scleral incision is made by a trifaceted guarded diamond knife 1 mm posterior to the superior limbus. The knife is set at a depth of 350 μm. An angled crescent tunnel blade (Wilson Ophthalmics, Mustang, OK) is used to make the deep lamellar dissection from the incision into clear cornea, approximately 3 mm centrally. A special semisharp dissector spatula (Bausch and Lomb, St. Louis, MO) extends the lamellar pocket over the entire cornea (Fig. 63-6). A paracentesis is made at the 2 o’clock position with a supersharp blade (Katena, Denville, NJ) at the limbus, and air is injected in the anterior chamber with a 30-gauge cannula. The air bubble is maintained at all times. A low-profile intralamellar trephine (Terry Trephine; Bausch and Lomb) with a cutting diameter of 7 mm, along with an external knurled circular handle, is used to trephinate the posterior lamellar recipient tissue (Fig. 63-7). When the anterior chamber is entered, air escapes into the deep lamellar plane. The remaining stromal fibers are cut with intralamellar corneal scissors and the stromal disk is grasped with angled Kelman forceps (Storz, St. Louis, MO) and removed. The anterior chamber depth is maintained with repeated injections of air.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree