, Rudolf Leuwer2, Konrad Schwager3 and Sören Wenzel4
(1)
Otology and Neurotology Division, University of Arkansas for Medical Sciences, Little Rock, Arkansas, USA
(2)
Otorhinolaryngology Head and Neck Surgery, HELIOS Hospital, Krefeld, Germany
(3)
Otorhinolaryngology Head and Neck Surgery, Klinikum Fulda, Fulda, Germany
(4)
Otorhinolaryngology Head and Neck Surgery, Pinneberg, Germany
Eustachian tube (ET) function: ventilation, drainage and protection
Muscular compliance
Otitis media with effusion
Tympanostomy tubes
Cleft lip, alveolus and palate (CLAP)
ET function tests: sonotubometry and manometry according to Estève
2.1 Eustachian Tube Function
The Eustachian tube plays an important role in middle ear function. It provides ventilation, drainage and protection of the middle ear [53, 94, 113, 203]. The Eustachian tube is open for only very short periods [36], which usually occur every few minutes around 1,000 times per 24 h [140]. Under conditions of normal function, the lumen of the membrano-cartilaginous portion of the ET is usually closed because of the pressure in the surrounding tissue [36]. That is why its key function is to protect the middle ear against reflux, organisms, sound pressure and air pressure changes in the pharynx [90, 197]. The muscular coordination of the Eustachian tube depends in part on the activity of the tensor veli palatini muscle [53, 178]. The task of the tensor veli palatini muscle is to actively open and compress the Eustachian tube at the same time. The tensor veli palatini muscle consists of two portions, a superficial and a profound portion. The superficial portion is situated between the pterygoid process and the spine of the sphenoid bone, whereas the profound portion is attached to the lateral lamina of the tubal cartilage and the pterygoid hamulus [12, 26, 52]. The mechanical impairment of the muscle may lead to a poor tubal function [94, 174]. Contraction of the tensor veli palatini muscle occurs during swallowing, yawning or movements of the mandible [203]. From its origin and innervation, it appears to be a muscle of mastication that, at the end of the embryonic period, specializes in the movements of the soft palate and the auditory tube [27].
The muscular coordination is a complex function of the tensor veli palatini muscle: contraction of the profound portion of the tensor muscle only causes the opening of the cranial one-third of the tube (the so-called Rüdinger’s safety canal) [113, 161]. At the same time, contraction of the superficial portion of the muscle compresses the Ostmann’s fat pad and thus the lower two-thirds of the tube. Mucosal folds in the lower portion of the tube support a propulsion mechanism towards the nasopharynx during compression [134, 169]. Hence, the muscular coordination involves the simultaneous aeration and drainage of the middle ear by the contraction of the tensor veli palatini muscle.
To understand the mechanics of this muscle, one has to know the peculiarity of this muscle: due to its position between the skull base and the pterygoid process, with the exception of one small bursa on the lateral side of the pterygoid hamulus, it only has an isometric activity [27, 152]. That is why the tensor veli palatini muscle alone is not able to provide the muscular coordination of the Eustachian tube.
The labelling of the surrounding structures in an MRI study of the Eustachian tube (Fig. 2.1) reveals that the physiological tensor activity depends on certain anchors that change the direction of the muscle tension, so-called hypomochlia [93]. Three hypomochlia can be identified:
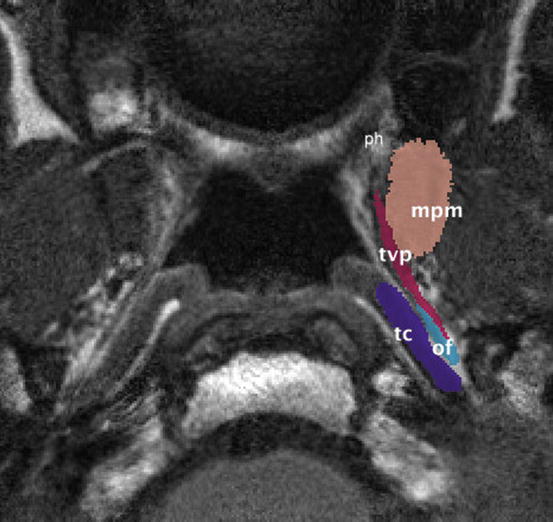

Fig. 2.1
Axial MRI study of the Eustachian tube. The critical structures are labelled. ph pterygoid hamulus, mpm medial pterygoid muscle, tvp tensor veli palatini muscle, tc tubal cartilage, of (lateral) Ostmann’s fat pad
These hypomochlia are acting as modulators of the isometric force vectors. However, in contrast to the pterygoid hamulus and Ostmann’s fat pad, the medial pterygoid muscle is mobile. Hence, its contraction and relaxation changes both the position and the tension of the tensor veli palatini muscle. This can be demonstrated by an in vivo 3D MRI model of the labelled structures from Fig. 2.1 in two different physiological states: (a) mouth closed (contraction of medial pterygoid muscle) and (b) mouth opened (relaxation) (Fig. 2.2). Relaxation of the medial pterygoid muscle rotates the tensor veli palatini muscle in an anterolateral direction off the lumen of the Eustachian tube.
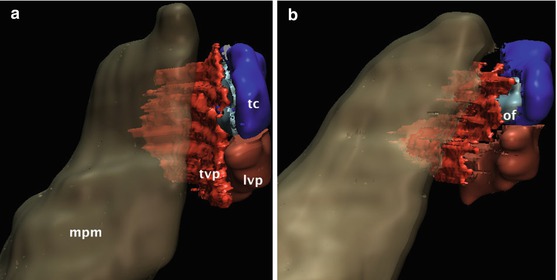

Fig. 2.2
In vivo 3D MRI model of the labelled structures from Fig. 2.1 in two different physiological states: (a) mouth closed (contraction of medial pterygoid muscle) and (b) mouth opened (relaxation). Relaxation of the medial pterygoid muscle rotates the tensor veli palatini muscle in an anterolateral direction off the lumen of the Eustachian tube. mpm medial pterygoid muscle, tvp tensor veli palatini muscle, tc tubal cartilage, of (lateral) Ostmann’s fat pad
The discussion about the differential role of the various auditory tube-associated muscles in tubal function remains controversial [61]. Most researchers would agree that the tensor veli palatini muscle is the primary dilator of the auditory tube [12]. Although Hecht et al. for the first time mentioned that the medial pterygoid muscle might play a role in auditory tube function, this opinion was denied by others [35]. However, comparative anatomical findings suggest that the upper portion of the medial pterygoid muscle stiffens the anterior portion of the tube [18, 47].
Figure 2.3 is a model of the main force vectors determined by the muscular coordination of the Eustachian tube. It shows the anterolateral rotation of the medial pterygoid muscle when relaxing, the pressure forces of the lateral layer of the tensor veli palatini muscle on the Ostmann’s fat pad and the laterocaudal traction of the medial layer on the lateral lamina of the cartilage. Due to the position of the lateral suspensory ligament, there probably is a mediocranial rotation of the medial lamina of the cartilage. The latter vector could indicate the role of the salpingopharyngeal muscle as an anchor chain for the cartilage.
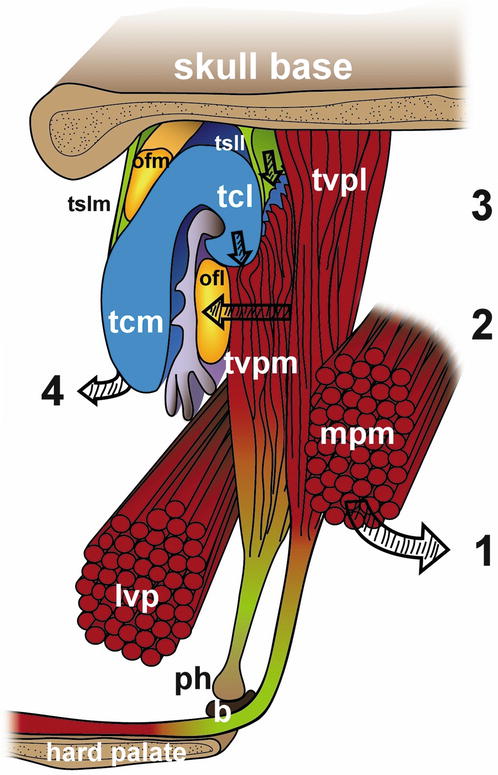

Fig. 2.3
Main force vectors determined by the muscular coordination of the Eustachian tube: (1) Anterolateral rotation of the medial pterygoid muscle during relaxation; (2) pressure forces of the lateral layer of the tensor veli palatini muscle on the Ostmann’s fat pad; (3) laterocaudal traction of the medial layer on the lateral lamina of the cartilage; (4) mediocranial rotation of the medial lamina of the cartilage. tvp m tensor veli palatini muscle, medial layer, tvp l lateral layer, ph pterygoid hamulus, b bursa, lvp levator veli palatini muscle, mpm medial pterygoid muscle, tcm tubal cartilage, medial lamina, tcl lateral lamina, ofm medial Ostmann’s fat pad, ofl lateral Ostmann’s fat pad, tslm medial tubal suspensory ligament, tsll lateral tubal suspensory ligament (Graphical model by Daniela Beyer, MA, Hannover University Medical School)
There are typical clinical examples of an impairment of these hypomochlia. One is the loss of Ostmann’s fatty tissue, which has been described as one factor for the development of a patulous Eustachian tube [137]. This topic is part of Sect. 2.3.
Another clinical example is the sequelae of hamulotomy after cleft palate repair (see below).
2.2 Epidemiology of the Diseased Eustachian Tube
2.2.1 Eustachian Tube Dysfunction in Children
Otitis media with effusion is a major paediatric healthcare issue. It is defined as a middle ear effusion without signs or symptoms of an acute infection [98]. By the age of 2, about 91 % of children will have had at least one episode of middle ear effusion [159]. The pathogenesis and duration of middle ear effusions are multifactorial. Viral upper respiratory tract infection, an immature immune system, exposure to tobacco smoke in the household and Eustachian tube dysfunction seem to play the most important roles [24, 34].
Eustachian tube dysfunction is a major problem in the young child due to the Eustachian tube’s horizontal direction, its small calibre and its shortness [36]. But, when morphologically assessing the Eustachian tube alone, there is no explanation as to why some children suffer from otitis media with effusion and some do not [24, 194]. A failure of the tubal opening function has been regarded as the major problem in otitis media with effusion and retractions of the tympanic membrane. However, a tubal closing failure may contribute to the same condition [15]. In addition, the function of the Eustachian tube is impaired by the adenoids, which can mechanically obstruct the nasopharyngeal orifice of the Eustachian tube [43], provide the bacterial focus for otitis media [126, 138] or hinder velopharyngeal competence, especially with cleft lip, alveolus and palate [72, 183]. The clinical impact of each of these alternative pathophysiological mechanisms has not yet been conclusively answered.
2.2.2 Eustachian Tube Dysfunction in Cleft Palate Patients
Due to aberrant anatomy of the palate and the nasopharyngeal space [153], the cleft palate may serve as a model for understanding Eustachian tube dysfunction and its impairment.
Cleft lip, alveolus and hard and soft palate (CLAP) is one of the most frequent inborn deformities, with about 2 cases per 1,000 births [170]. The incidence of otitis media with effusion (OME) in cleft palate children is about 97 % [55]. It is important to note that the incidence of OME in children without a cleft is not much smaller. Consequently if we consider the role of cleft palate repair on Eustachian tube function [45], we should not only have a look at the early need for tympanostomy tubes [82]. Tube insertion does not necessarily have an influence on the risk of cholesteatoma, atelectasis or tympanic membrane perforations [154]. In fact it does not have an impact on Eustachian tube function at all [204]. Thus, for practical purposes, we should completely delineate otitis media with effusion in cleft children and chronic middle ear disease in cleft adults. It is still unclear whether the extent of middle ear disease depends on the type of cleft [59]. Although the velum may obstruct the nasopharyngeal orifice of the Eustachian tube before palate repair [11], the prevalence of OME after veloplasty still remains about 70 % [158], and a very early closure does not significantly change the need for ventilation tubes [127]. According to Flores et al. [45] follow-up of patients after cleft palate repair should last at least 4–7 years in order to appreciate its effects on Eustachian tube function. In addition the neuromuscular development of the tensor veli palatini muscle generally is unfinished before the age of 7 years [59]. All surgical procedures on the growing bone and soft tissue of the midface have an impact upon the development of the facial bones [170]. Thus, it is very difficult to separate between those factors that are independent of the technique of cleft palate repair and those factors occurring by or after closure of the soft palate and which often persist during adulthood [45]. It is obvious that during veloplasty the integrity of the hamulus as well as the tensor veli palatini muscle is at risk. Thus, the muscular coordination of the Eustachian tube can be immediately impaired by surgery [9, 92]. Sheer et al. [178] used histological specimens of the peritubal space from infants with cleft palates in order to develop a finite element model of the forces involved in Eustachian tube opening. They could demonstrate two different things: the tensor veli palatini muscle force is the only direct predictor of Eustachian tube opening during muscle-assisted lumen dilations and that zero tensor veli palatini muscle force would result in Eustachian tube dysfunction. Thus, surgical division of the tensor tendon or resection of the hamulus itself, which can be simulated as a reduction in tensor force, was predicted to have negative effects on Eustachian tube function. These experimental observations were clinically confirmed by Flores et al. [45], who showed that tensor tenopexy and the abandonment of tendon transection would decrease the need for tympanostomy tubes. Our group [174] examined 15 adult cleft palate patients, 7 of whom had chronic ear disease. Eleven patients had a unilateral and four patients a bilateral CLAP. The patients were examined using ear microscopy and MRI. For different reasons ear microscopy is the only suitable method to reliably assess chronic middle ear disease [182, 211]. The patients without middle ear pathology had an intact hamulus and tensor veli palatini muscle. In all patients with chronic middle ear disease, the tensor muscle had no continuity in any of the cases as shown by MRI. In four of the latter cases, the pterygoid hamulus was absent. Thus, the mechanical impairment of the tensor and/or the pterygoid hamulus may lead to an impaired function of the tube and loss of middle ear integrity [94] (Fig. 2.4a, b). If we consider that OME in cleft children and chronic middle ear diseases in adult patients are different entities and that the effects of cleft palate surgery upon middle ear function are visible after at least 4–7 years, it is easy to interpret a study published by Kane et al. [77]. They observed 161 children for 1 year after palatoplasty using brainstem-evoked audiometry and speech assessment. They did not perform ear microscopy. About half of the patients received hamulotomy. At the age of 2, they did not find a significant difference concerning perioperative morbidity, hearing test results and preliminary speech results in both groups. Considering the method (brainstem-evoked audiometry), the short period of observation (1 year) and the fact that most of the patients received tympanostomy tubes, this result is not amazing. Major drawbacks of many of these studies are the time frame of observation, the examination method, different kinds of palatoplasty performed and the fact that most of the children receive tympanostomy tubes [177].
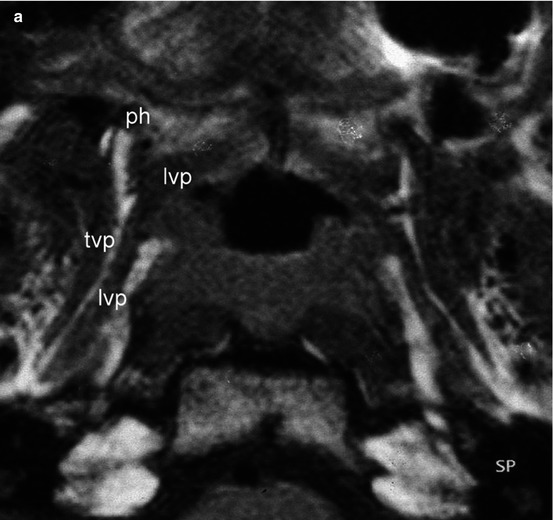
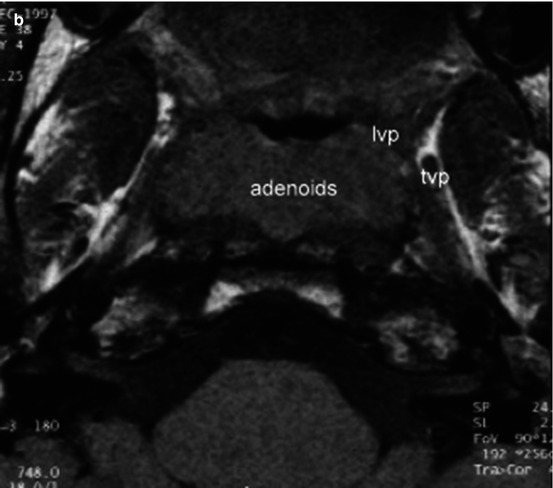


Fig. 2.4
(a) Axial MRI study of the Eustachian tube with a CLAP patient. Note the position of the levator veli palatini muscle after cleft palate repair. The tensor and hamulus are intact. Both middle ears are normal. ph pterygoid hamulus, tvp tensor veli palatini muscle, lvp levator veli palatini muscle. (b) Axial MRI study of the Eustachian tube with another CLAP patient. The tensor and hamulus are not intact. Both middle ears are atelectatic. tvp tensor veli palatini muscle, lvp levator veli palatini muscle
The question as to whether the adenoid pad in the nasopharynx should be maintained in order to prevent postoperative velopharyngeal insufficiency still remains unanswered. According to Hubbard et al. [72], only those cases should undergo adenoidectomy in which the adenoids do not contribute to the velopharyngeal closure.
Because OME and its sequelae are a common problem of children with CLAP, the rational concept of an interdisciplinary follow-up with an ENT specialist is a crucial task [127]. If we reconsider the idea that OME of the cleft child and chronic middle ear disease of the cleft adult are different entities, this follow-up should include not only the young children before and immediately after cleft surgery, but also young adults. Tympanostomy tubes are a reliable tool to restore hearing with OME, but their general value for a long-term hearing improvement is controversial [98]. There are several studies in favour of a more aggressive or a more conservative use of tympanostomy tubes [82, 181 vs. 172]. The assessment of cleft palate preschool and school children by the otolaryngologist should take place at least twice a year. Bilateral OME for 6 months should be treated with tympanostomy tubes [97]. Children with CLAP have a need for long-term interdisciplinary prospective studies in order to characterize those who would benefit most from tympanostomy tubes. The focus of these studies should be identifying the treatment that will be adequate to prevent persistent hearing loss due to atelectasis or cholesteatoma.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


