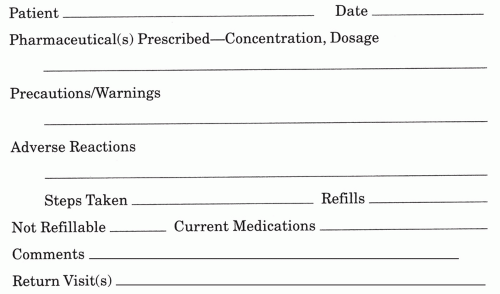
Do write legibly.
Do check drug spelling; many drug names look alike.
Do include dosage form, quantity, and strength of medication.
Do specify length of treatment on acute care prescriptions.
Do use the most economical size package on chronic care prescriptions.
Do make sure the patient understands all special instructions, for example
Refrigeration
Shake suspensions
Proper instillation techniques
Do specify the symptoms that require “as needed” treatment, for example
Itch
Pain
Redness
Do use only metric measurement. DO NOT use household or apothecary measurement systems.
Do not preprint your DEA number on your prescription pad.
Do use tamper resistant prescription pads where required by law.
Do not leave prescription pads unattended on counter tops.
reports an allergy to an anesthetic agent in one chemical class, an agent from the alternate class can be considered with caution. For example, if a patient reports an allergy to tetracaine (an ester compound), a compounding pharmacist can prepare topical lidocaine (an amide compound) for ocular use.
TABLE 5-1 Lati Abbreviations Commonly Used in Ophthalmic Prescription Writing | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-2 A “Minimum List” of Dangerous Abbreviations, Acronyms, and Symbols Has Been Approved by Joint Commission (Beginning January 1, 2004, the Following Items Must Be Included on Each Accredited Organization’s “Do Not Use” List) | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||
TABLE 5-3 Common Uses of Ophthalmic Dyes | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-4 Properties and Characteristics of Fluorescein | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 5-5 Characteristics of Fluorexon | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||
TABLE 5-6 Characteristics of Rose Bengal | |||||||
|---|---|---|---|---|---|---|---|
|
TABLE 5-7 Comparative Staining Characteristics of Fluorescein, Rose Bengal, and Fluorexon | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-8 Topical Products | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 5-9 Physiochemical and Pharmacologic Properties of the Injectable Local Anesthetics | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 5-10 Commonly Used Topical Ocular Anesthetics | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||
TABLE 5-11 Central Nervous System Toxicities of Local Anesthetics | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-12 Cardiovascular Symptoms of Toxicity from Local Anesthetics | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-13 Ocular Toxicity from Acute Administration of Topical Ocular Anesthetics | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-14 Indications for Dilation | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-15 Mydriatic Pharmaceuticals for Dilation | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 5-16 Van Herick Filtration Estimation Guide | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-17 Examination Procedures in Relation to the DFE | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-18 Example of the “Round-Robin” Approach to Patient Scheduling for Dilation | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||
TABLE 5-19 Indications for Cycloplegic Refraction | ||||||||
|---|---|---|---|---|---|---|---|---|
|
TABLE 5-20 Comparison of Cycloplegic Agents | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-21 Efficiency of Cycloplegics | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5-22 Ocular Side Effects of Cycloplegic Agents | |||
|---|---|---|---|
|
TABLE 5-23 Dose-related Systemic Side Effects of Atropine | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 5-24 Side Effects Associated with Topical Cholinesterase Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 5-25 Characteristics of Accommodative Esotropia | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||
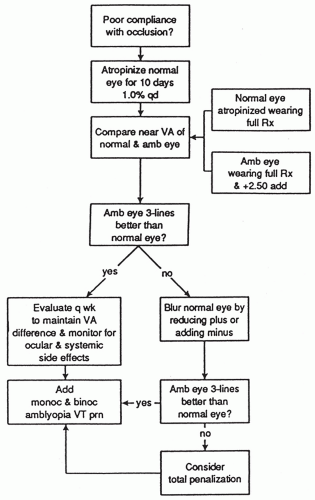 Figure 5-8. Management of amblyopia with penalization.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|