Percutaneous Tracheostomy
Karen M. Kost
INTRODUCTION
Tracheostomy has a long and colorful history. The first reference to the procedure can be found in the sacred book of Hindu medicine, the Rig Veda, dated to approximately 2000 BC. There are many interesting early references to tracheostomy being performed as a lifesaving procedure in both animals and humans. One of the first animal descriptions involves a farmer inserting a hollow reed in the neck of one of his sheep in an effort to save the animal from upper airway obstruction. Alexander the Great is reported to have performed a tracheostomy in the fourth century BC when he “punctured the trachea of a soldier with the point of his sword when he saw a man choking from a bone lodged in his throat.” In the early 1600s, Nicholas Habicot of Paris described a successful tracheostomy in a 14-year-old boy who developed upper airway obstruction following an attempt at swallowing a bag of gold coins he had stolen.
For each of these success stories, however, there were many descriptions of tracheostomies associated with high morbidity and mortality rates. Fabricius ab Aquapendente aptly wrote: “The terrified surgeons of our times have not dared to exercise this surgery and I also have never performed it. Even the mention of this operation terrifies the surgeons; hence it is called the scandal of surgery.” In 1799, when Dr. Elisha Dick recommended that a tracheostomy be performed for a high-profile patient in airway distress, two colleagues vocally opposed him, and on December 14, 1799, President George Washington died of an acute upper airway obstruction. It was to be the work of Armand Trousseau and later Chevalier Jackson that would successfully bring tracheostomy into the realm of the surgeon’s armamentarium. Jackson standardized the technique and indications for the procedure and showed that with attention to a few key technical details, the morbidity and mortality could be dramatically reduced to acceptable levels. As tracheostomy became widely accepted as a useful surgical tool, indications for its use gradually expanded to include upper airway obstruction from croup and diphtheria, trauma, tuberculosis, and syphilis. It was even briefly used as a means of securing the airway during the administration of general anesthesia in the early 20th century.
The first descriptions of percutaneous tracheostomy date back to the 1950s following Seldinger’s report of needle replacement over a guidewire for arterial catheterization. The first attempt at percutaneous tracheostomy was reported in 1955 by Sheldon as a means of providing rapid airway access in emergency situations. The technique never gained popularity, however, and the single publication on the procedure reports a case in which the trachea was perforated with the device. Over the next several years and indeed decades, several other percutaneous techniques with various modifications were introduced, but almost all were quickly abandoned because of high complication rates.
Interest in the area persisted, however, and in 1985 when Ciaglia described a bedside percutaneous dilatational tracheostomy technique, the procedure was rapidly adopted in many intensive care units (ICUs), principally by critical care physicians and anesthesiologists. The technique is based on progressive dilatation of an initial tracheal puncture and, as originally described, is performed blindly. An early publication in the otolaryngology literature reported several instances of severe morbidity with the procedure including creation of a false passage, pneumothorax, pneumomediastinum, and even one death. All of these complications were the result of poor patient selection and the blind aspect of the technique. The addition of endoscopic guidance in the early
1990s addressed the blind aspect of the procedure and added significantly to its safety. Several publications of percutaneous dilatational tracheostomy in the last 20 years have shown that with continuous endoscopic guidance and attention to technical detail, percutaneous dilatational tracheostomy is as safe as traditional open surgical tracheostomy in the operating room with similar or lower complication rates.
1990s addressed the blind aspect of the procedure and added significantly to its safety. Several publications of percutaneous dilatational tracheostomy in the last 20 years have shown that with continuous endoscopic guidance and attention to technical detail, percutaneous dilatational tracheostomy is as safe as traditional open surgical tracheostomy in the operating room with similar or lower complication rates.
HISTORY
Critically ill patients often suffer from multisystem disease and require extremely complex care. Tracheostomy is a frequently performed procedure in intubated ICU patients and not surprisingly is associated with a higher risk in this particular subset. Well over half of currently performed tracheostomies are on critically ill patients requiring prolonged mechanical ventilation. Other indications include pulmonary toilet, sleep apnea, chronic lung disease, and conditions requiring home mechanical ventilation. The decision to proceed with a tracheostomy should be based on a thorough assessment of the patient in conjunction with the treating team of the ICU. Several factors may influence the decision to perform the procedure, and these include the facilities at the particular institution, the skills of the personnel, and the particular features of the patient’s airway and respiratory physiology as well as the patient’s prognosis.
PHYSICAL EXAMINATION
The importance of a comprehensive preoperative patient assessment with particular attention to the examination of the neck cannot be overemphasized. Examination of the neck is very helpful in determining whether a patient is a candidate for endoscopic percutaneous dilatational tracheostomy and will help to avoid any unpleasant surprises during the procedure. Intubated patients in the ICU are often first observed in bed with their head flexed and turned to the side. It is virtually impossible to appropriately examine the neck in this position. It is well worth investing the effort to properly position the patient with the head extended as would be done for open tracheostomy. In very agitated patients, it may sometimes be necessary to administer a small amount of sedation in order to be able to do this. Once the patient is properly positioned, the first step is to palpate the critical landmarks of the neck, which include the cricoid cartilage and sternal notch. The cricoid may lie low in the neck, but with careful palpation, it can usually be identified. The presence of a midline neck mass, a high innominate artery, and a scarred fibrotic neck such as is the case in patients with previous surgery or radiotherapy to the area should all raise red flags. In these instances, percutaneous dilatational tracheostomy may not be appropriate and an open surgical technique in the operating room is likely to be safer. Patients with anatomical deformities such as cervical osteoarthritis and kyphoscoliosis where the neck cannot be extended and the cricoid cartilage is not palpable are not suitable candidates for endoscopic percutaneous dilatational tracheostomy. Obese patients with thick pretracheal soft tissues are at increased risk of accidental decannulation owing to the short proximal length of “standard” tracheostomy tubes. In these patients, placement of a proximally extended tracheostomy tube should be routine.
INDICATIONS
Endoscopic percutaneous tracheostomy should only be considered in adult intubated ICU patients. Since well over half of all of tracheostomies are performed on this patient subset, endoscopic percutaneous tracheostomy is an attractive option for the following reasons: the procedure is well suited for the bedside, the required instruments are simple, and only ambient light is required. This obviates the need for transporting a full instrument kit, headlight, and possible cautery unit, all of which would be required for a standard open bedside procedure.
CONTRAINDICATIONS
Appropriate patient selection is key in reducing complications of endoscopic percutaneous tracheostomy. Contraindications include the following (Table 34.1):
1. Unprotected airway—Attempting endoscopic percutaneous tracheostomy on any patient with an unprotected airway is an invitation to disaster and indeed accounts for some of the early reports of morbidity and mortality. The procedure requires an airway that is secured with an endotracheal tube (ETT) or laryngeal mask airway; a bronchoscope can be passed through either of these for direct endoscopic visualization of the entire procedure.
2. Children—The anatomy of the pediatric airway differs significantly from that of the adult. The trachea is much smaller and more pliable with the carotid arteries in very close proximity. It is therefore difficult to maintain adequate ventilation through a necessarily small ETT that is largely occupied by a bronchoscope. Furthermore, the airway is extremely collapsible, and attempted dilatation is likely to result in anterior/posterior collapse with possible damage to the posterior wall. For these reasons, percutaneous
dilatational tracheostomy should never be attempted in children. Eligibility for percutaneous dilatational tracheostomy is not so much a function of age but more of physical maturity.
dilatational tracheostomy should never be attempted in children. Eligibility for percutaneous dilatational tracheostomy is not so much a function of age but more of physical maturity.
TABLE 34.1 Contraindications to Percutaneous Tracheostomy | |
|---|---|
|
3. Inability to palpate the cricoid cartilage—The cricoid cartilage and sternal notch are key anatomical landmarks, which must be clearly palpable prior to considering endoscopic percutaneous dilatational tracheostomy. Palpating the cricoid cartilage in obese patients requires extra effort. This means placing the head in maximum extension and sometimes requires pulling the skin up over the neck with the left hand to allow the right hand to palpate the cricoid cartilage clearly. Patients with severe anatomic abnormalities such as kyphoscoliosis are clearly unsuitable for percutaneous dilatational tracheostomy.
4. High innominate artery—Careful preoperative examination of the patient allows for an easy identification of a high innominate artery. Resting the index and third finger just above the sternal notch reveals a strong pulsation immediately below. It is important to distinguish a high innominate artery from a transmitted pulsation from the chest.
5. Midline neck mass—A midline neck mass lying over the inferior aspect of the cricoid and the first three tracheal rings is a contraindication to percutaneous dilatational tracheostomy.
6. High positive end-expiratory pressure (PEEP)—Patients who require a very high PEEP of fifteen (15) or more are at risk for complications such as subcutaneous emphysema and pneumothorax. As a result, endoscopic percutaneous dilatational tracheostomy is unsuitable in this group.
7. Coagulopathy—A large number of patients in the ICU have some sort of coagulopathy at the time of consultation for tracheostomy. These abnormalities should be corrected as much as is medically possible preoperatively. Ideally, the number of functioning platelets should be >50,000, and the International Normalized Ratio (INR) should be corrected to a value of <1.5. This being said, many patients are on both aspirin and clopidogrel bisulphate (Plavix) and are unable to stop one or both of these agents for medical reasons. Although the risk of perioperative bleeding is increased in these cases, endoscopic percutaneous dilatational tracheostomy may still be performed and indeed may be the preferred option because of the reduced dissection and risk of bleeding. Ideally, patients on warfarin should stop the drug 5 days prior to tracheostomy or receive fresh frozen plasma. Again, achieving these goals may not be possible or realistic, and in a patient with a persistent low level coagulopathy, endoscopic percutaneous dilatational tracheostomy is preferred to open surgical tracheostomy because of the reduced dissection and incidence of bleeding.
It has been said in the literature that obese patients or those having had a previous tracheostomy are unsuitable candidates for endoscopic percutaneous tracheostomy. Both of these statements are incorrect. Patients having had a previous tracheostomy can certainly have an endoscopic percutaneous dilatational tracheostomy if they have no other contraindications. A small incision should be placed in the previous scar, and the key is carrying the incision down through the scar tissue. The site of the previous tracheostomy can often be identified endoscopically by a small irregularity in the anterior tracheal wall, and this site may be chosen for entry into the trachea. Obese patients are at higher risk of complications whether they undergo endoscopic percutaneous dilatational tracheostomy or open surgical tracheostomy in the operating room. There is no evidence to suggest that obese patients undergoing percutaneous dilatational tracheostomy in the ICU are at an even higher risk of complications. Because of the increased pretracheal soft tissue thickness in this population, it is extremely important to use a proximally extended tracheostomy tube in order to reduce the risk of accidental decannulation. It has been shown that pretracheal soft tissue thickness (Table 34.2) can be reliably predicted within 4 mm in obese patients as a function of neck and arm circumference. Using Table 34.2, it can be seen that a patient with a neck circumference of 55 cm and an arm circumference of 45 cm would have a pretracheal soft tissue thickness of 2.8 cm. A proximally extended tracheostomy tube would be required in this patient since “standard” tubes have too short a proximal length.
PREOPERATIVE PLANNING
As with any surgical procedure, the patient’s comorbidities should be optimized prior to percutaneous dilatational tracheostomy. Preoperative testing is minimal and includes a recent chest radiograph as well as serum determination of hemoglobin, prothrombin time, partial thromboplastin time, INR, and platelets. Because of the minimal bleeding associated with the procedure, cross-matching is not necessary even in the presence of low hemoglobin levels. A fully equipped intubation cart should be available in the event of accidental extubation
during the procedure. Obese patients or those with a short, thick neck and excessive subcutaneous tissues are at particular risk for accidental decannulation. This potential problem can be anticipated and avoided by using a proximally extended tracheostomy tube.
during the procedure. Obese patients or those with a short, thick neck and excessive subcutaneous tissues are at particular risk for accidental decannulation. This potential problem can be anticipated and avoided by using a proximally extended tracheostomy tube.
TABLE 34.2 Predicted Neck Thickness in Centimeters (cm) as a Function of Neck Circumference (NC in cm) and Arm Circumference (cm) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
SURGICAL TECHNIQUE
The Bedside Team
Four people are needed for bedside endoscopic percutaneous therapy (PDT), each with a clearly defined role. (1) The surgeon stands at the patient’s right. (2) The endoscopist stands at, or close to the head of, the bed and is charged with providing a good view of the intratracheal portion of the procedure, adjusting the position of the ETT, and making sure the airway is protected by securely holding the ETT. Any physician with endoscopic skills may fulfill this role, including a resident, respirologist, anesthetist, or critical care physician. (3) The respiratory therapist at the patient’s left is responsible for adjusting ventilator settings. (4) A nurse is required to administer medications during the procedure. The positions of the surgeon and respiratory therapist are reversed for left-handed surgeons.
Anesthesia
The type of anesthesia delivered depends to a great extent on the institution, and the personnel involved. The presence of an anesthetist is optional and again depends on hospital policy. The goal is for the patient to be comfortable. Options include the equivalent of general anesthesia at the bedside or local anesthesia with intravenous (IV) sedation. I typically use the latter, with a combination of midazolam, propofol, and fentanyl. Immediately preceding the procedure, 2 to 3 mg of midazolam is given IV, and 1 to 2 mL of topical lidocaine is sprayed into the ETT to decrease coughing. The incision site is generously injected with lidocaine containing 1:100,000 epinephrine. A bolus of propofol is then given IV based on the patient’s weight, followed by a continuous infusion which is titrated according to degree of sedation and vital signs. Fentanyl is given in measured doses for pain throughout the procedure. Tracheostomy is a highly stimulating procedure, and once the tracheostomy tube is in place, the stimulation stops abruptly. It is important to stop the propofol immediately at the end of the procedure to avoid a rapid drop in blood pressure.
Technique
Prior to beginning the procedure, the surgeon should optimally position the patient, prepare the medications, place the instruments, check the bronchoscope, and “troubleshoot” any potential problems in advance with the team. This effort translates into a quick and smooth procedure and reduces the chance of complications.
Although several techniques for performing percutaneous tracheostomy have been developed, the most commonly used in North America is the endoscopic percutaneous dilatational technique. Currently, there are a number of similar, commercially available kits. Periodically, modifications are introduced in an effort to further simplify/streamline an already simple procedure. The different kits have subtle differences, but the common
denominator in all of them is dilatation of an initial tracheal puncture with a single, sharply tapered dilator. More recently, a kit with an inflatable balloon, instead of the single dilator, was introduced to dilate the tracheal puncture: purported advantages include evenly distributed radial pressure and dilatation of the trachea, thus avoiding the depression of the anterosuperior tracheal wall seen with the single dilator. Because there is no evidence that any one kit is superior, the choice becomes a matter of personal preference.
denominator in all of them is dilatation of an initial tracheal puncture with a single, sharply tapered dilator. More recently, a kit with an inflatable balloon, instead of the single dilator, was introduced to dilate the tracheal puncture: purported advantages include evenly distributed radial pressure and dilatation of the trachea, thus avoiding the depression of the anterosuperior tracheal wall seen with the single dilator. Because there is no evidence that any one kit is superior, the choice becomes a matter of personal preference.
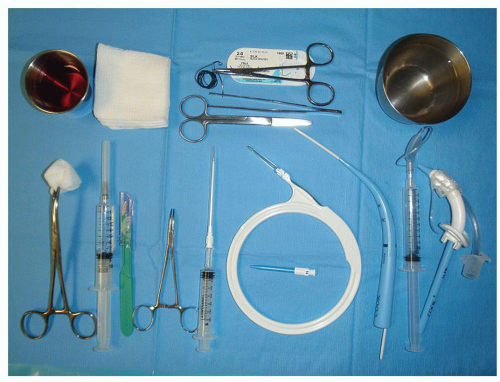
One of the most thoroughly evaluated kits in North America is the Ciaglia Blue Rhino percutaneous introducer kit (Cook Critical Care Inc., Bloomington, IN). The basic kit contains the following: a 15 blade scalpel, a 5-mL syringe, an introducer needle, a J-wire guide, an introducer dilator, a white guiding catheter, a sharply tapered single blue dilator with a hydrophilic coating, as well as 26-French and 28-French loading dilators.
Other required instruments include a curved hemostat, straight scissors, a needle driver, forceps, nonresorbable sutures, water-based lubricant, and sterile saline to activate the hydrophilic coating of the single dilator, two 10-mL syringes, and an appropriately sized tracheostomy tube.
The instruments are placed on an instrument stand over the patient’s bed in the order in which they are to be used (Fig. 34.1




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree