Purpose
To compare the outcomes of trabeculectomy surgery after Descemet stripping automated endothelial keratoplasty (DSAEK) to penetrating keratoplasty (PK).
Design
Retrospective case-control study.
Methods
Patients within an institutional setting who underwent trabeculectomy with mitomycin C (MMC) for uncontrolled elevated intraocular pressure (IOP) after corneal graft surgery were included. Patients with pre-existing glaucoma were excluded. Twenty patients with trabeculectomy after DSAEK and 41 patients with trabeculectomy after PK were analyzed. The main outcome measure was IOP control at 12 months. Secondary outcome measures were postoperative interventions including reinstatement of IOP-lowering medications, bleb needling with 5-fluorouracil (5FU) or further glaucoma surgery, and the incidence of complications related to trabeculectomy and/or corneal graft surgery.
Results
There was no difference in pre-trabeculectomy IOP between DSAEK vs PK group (35.5 ± 10.1 vs 32.9 ± 8.9, P = .31). At 12 months after trabeculectomy, mean IOP in the DSAEK group was lower compared to the PK controls (10.6 ± 3.2 vs 14.6 ± 8.5, P = .04). The proportion of patients who achieved an IOP less than 12 mm Hg was significantly higher in the DSAEK group (80.0% vs 48.6%, P = .03). The proportions of eyes that required intervention after trabeculectomy were comparable between the 2 groups (DSAEK vs PK, all interventions: 20.0% vs 39.5%, P = .15; IOP-lowering medications required: 15.0% vs 39.5%, P = .08; needling with 5FU: 20.0% vs 23.7%, P > .99; further glaucoma surgery: 0% vs 13.2%, P = .15). Corneal graft failure arising after trabeculectomy was seen in 10.0% of DSAEK cases and in 10.5% of PK controls ( P = 1.0).
Conclusion
Compared to trabeculectomy after PK, trabeculectomy after DSAEK achieved lower mean IOP at 12 months, and a larger proportion of DSAEK patients achieved an IOP of less than 12 mm Hg. There was no difference in the need for intervention after trabeculectomy, or incidence of other complications. Trabeculectomy is an effective surgical procedure for the management of postgraft ocular hypertension in DSAEK patients, and DSAEK may have an advantage in terms of success of trabeculectomy surgery over PK.
Development of glaucoma after corneal graft surgery from persistent and uncontrolled elevated intraocular pressure (IOP) remains as one of the most important complications after corneal transplantation, and can result in serious morbidity including corneal graft failure, glaucomatous optic neuropathy and associated visual field loss, and eventual irreversible loss of vision. Glaucoma after penetrating keratoplasty (PK) has been recognized as a problem since the 1960s. The incidence of post-PK glaucoma has been reported to range from 9% to 35%, and management remains difficult, with multiple treatment modalities often required to achieve satisfactory IOP control.
Descemet stripping with automated endothelial keratoplasty (DSAEK) is increasingly becoming the preferred surgical procedure for endothelial disorders over PK. The main advantages of DSAEK include a more rapid healing process, earlier visual rehabilitation, and a tectonically stronger eye. Compared to PK, there is less induced astigmatism and the ocular surface is more stable following DSAEK. Since less tissue is transplanted following DSAEK surgery, allograft rejection has been reported to be less frequent and milder compared to PK. However, whether this equates to a reduced duration and intensity of postoperative topical steroids has yet to be proven clinically in a randomized controlled study.
The management of secondary ocular hypertension after corneal transplantation involves medical treatment and, in recalcitrant cases, glaucoma surgery is often required. Surgical options for controlling elevated IOP post-PK include laser trabeculoplasty, trabeculectomy, glaucoma drainage devices, and cyclodestructive therapies. As DSAEK is a relatively new procedure, the incidence of de novo glaucoma after DSAEK is not clear, though some have reported it to range from 0% to 18%, with an average incidence of about 3%. The surgical management of elevated IOP post-DSAEK is not clearly defined at present. Previous authors have described outcomes of patients with trabeculectomy and tube shunts prior to DSAEK, in patients with preexisting glaucoma. Our study aims to describe the outcomes of trabeculectomy after DSAEK, and to compare this to trabeculectomy after conventional PK for secondary ocular hypertension.
Methods
Our study was a retrospective case-control study. Demographic information was obtained from all patients who had undergone trabeculectomy with mitomycin C (MMC) from January 1, 2005 to December 31, 2009, indicated for persistently elevated IOP, uncontrollable on topical medication, of greater than 21 mm Hg (ocular hypertension) that developed after DSAEK or PK. Patients who were aged less than 21 years old, pregnant, or breastfeeding were excluded. Patients with preexisting ocular hypertension (IOP >21 mm Hg with no optic disc changes of glaucoma, and with normal visual field perimetry), with glaucoma (evidence of optic disc changes or visual field defects typical for glaucoma), or who had prior glaucoma surgery before cornea graft surgery were also excluded from the study. Transplantation surgery was performed by the 7 corneal surgeons in our center, while trabeculectomy surgery was performed by 5 glaucoma surgeons.
Case records were retrospectively reviewed and the following data were recorded. Demographic data included age at time of glaucoma surgery, sex, and race. Clinical data included indications for corneal graft, type of corneal graft (DSAEK or PK), time interval (months) between corneal graft and trabeculectomy, pre-trabeculectomy IOP, and post-trabeculectomy IOP at day 1 and months 1, 3, 6, and 12. IOP was measured using Goldmann applanation tonometry. Interventions at all the time points were also recorded, which included reinstatement of IOP-lowering medications, bleb needling with 5-fluorouracil (5FU), and repeated glaucoma surgery. Complications such as bleb failure and corneal graft failure were also recorded. Corneal graft failure was defined as irreversible edema and loss of graft clarity in a graft that was previously documented to be thin and clear.
The decision to select PK vs DSAEK was made primarily on the degree or severity of stromal involvement, based on surgeon discretion, with DSAEK selected for cases with minimal stromal haze or scarring. However, it should be noted that prior to initiation of DSAEK at our center in 2006, all cases underwent PK surgery.
Penetrating keratoplasty was defined as an allograft procedure involving full-thickness replacement of corneal tissue. A standard technique using a Hanna vacuum trephine system (Moria SA, Antony, France) was performed. The recipient cornea was excised with the Hanna trephine, after which a 0.25- to 0.50-mm oversized donor cornea was punched out and sutured onto the recipient with 10-0 nylon. Intraoperative astigmatic control was achieved using a microscope-mounted keratoscope. A bandage contact lens was placed at the end of the surgery, and subconjunctival steroids and antibiotics were injected, followed by a regimen of topical steroids and antibiotics postoperatively, according to the discretion of the surgeon.
The DSAEK procedure was performed as previously described. In summary, donor graft lamellar dissection was performed using an automated lamellar therapeutic keratoplasty microkeratome (Moria USA, Doylestown, Pennsylvania, USA). Trephination was performed using a standard Hanna punch trephine system with standard trephine sizes between 7.75 mm and 9 mm, at 0.25-mm increments. A 5-mm scleral tunnel incision was made in all cases. Stripping of the Descemet membrane and endothelium was performed with a reverse Sinskey hook under air followed by insertion of the donor graft by the taco-folded technique or the Sheets glide-insertion technique. An inferior surgical peripheral iridectomy was then performed. An air bubble was injected into the anterior chamber for tamponade and to enhance donor graft adhesion, while drainage of interface fluid was achieved through 4 paracentral vents. The air bubble was partially evacuated before the end of the procedure to leave an air bubble approximating the size of the graft. Postoperative topical steroids and antibiotics were subsequently given according to a regimen determined by the operating surgeon.
Postoperatively, the steroid regimen was similar in the DSAEK and PK group. The patients were given a combination of topical steroids (prednisolone acetate 1% or dexamethasone phosphate 0.1%), initially at 3-hour intervals, with topical antibiotics (tobramycin, levofloxacin, or moxifloxacin). Topical steroids were tapered gradually, reducing to 3 times daily by about 4 to 6 months and once daily by the end of 12 months, though in immunologically high-risk PK steroid application was maintained indefinitely at a higher dose ranging between twice and 3 times a day.
Trabeculectomy with mitomycin C was performed using a standard technique. A fornix-based conjunctival bleb and a partial-thickness scleral flap were created, after which MMC 0.4 mg/mL was applied subconjunctivally over a wide area around the scleral flap for 1.5 to 3 minutes using polyvinyl alcohol sponges. The sponges were then removed and copious irrigation with saline solution was performed. A full-thickness sclerostomy was then created, and the scleral flap and conjunctival flap were closed sequentially with 10/0 nylon sutures. All participants received a standardized regimen of postoperative topical antibiotics and steroids.
The main outcome measure was IOP control at 12 months after trabeculectomy. A successful outcome was defined as achievement of a certain IOP level (eg, IOP ≤18 mm Hg, IOP ≤15 mm Hg, and IOP ≤12 mm Hg) with or without antiglaucoma medications, and a failed outcome was defined as nonachievement of IOP at a certain level, or need for other interventions (eg, bleb needling with 5FU or further glaucoma surgery). Further glaucoma surgery was performed at the discretion of the glaucoma surgeon, when IOP was uncontrolled despite antiglaucoma medications and bleb needling with 5FU. Secondary outcome measures were the need for intervention after trabeculectomy and complications such as corneal graft failure.
Statistical analysis was performed using SPSS software version 13.0 (SPSS, Inc, Chicago, Illinois, USA). Univariate analysis was performed using the χ 2 test or t test as appropriate. Statistical significance was defined at P value <.05.
Results
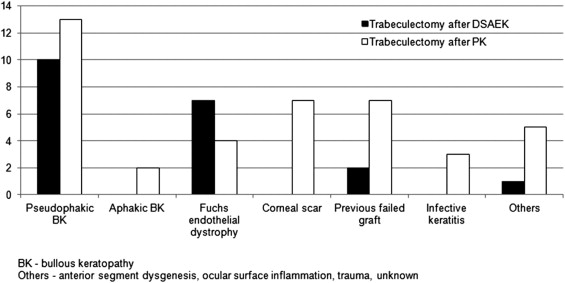
Baseline demographic data are shown in Table 1 . There were 13 male and 7 female subjects in the DSAEK group and 30 male and 11 female subjects in the PK group. There was no difference in mean age between both groups. The indications for corneal graft are shown ( Figure ); and most were indicated for pseudophakic bullous keratopathy (50.0% [10/20] in the DSAEK and 31.7% [13/41] in the PK group, P = .26) and Fuchs endothelial dystrophy (35.0% [7/20] and 9.8% [4/41] respectively, P = .03). The proportion with Fuchs endothelial dystrophy was significantly higher in the DSAEK group ( P = .03), while for the rest of the diagnoses they were comparable (all P > .05). After excluding those with Fuchs endothelial dystrophy and pseudophakic bullous keratopathy, the proportion with other corneal diagnoses (eg, corneal scar, infective keratitis) was lower in the DSAEK group (15.0% [3/20] vs 58.5% [24/41], P = .002). Mean interval (in months) between corneal graft and trabeculectomy appeared shorter in the DSAEK group, though this did not reach statistical significance. Mean duration of IOP-lowering medications used before trabeculectomy (in months) was comparable between both groups.
| Trabeculectomy After DSAEK | Trabeculectomy after PK | P Value | |
|---|---|---|---|
| Number of eyes | 20 | 41 | — |
| Sex (male:female) | 13:7 | 30:11 | — |
| Mean age, years (SD) | 60.8 (11.5) | 57.1 (16.2) | .362 |
| Mean interval between corneal graft and trabeculectomy, months (SD) | 8.0 (4.2) | 20.1 (27.5) | .056 |
There was no difference in pre-trabeculectomy IOP between DSAEK vs PK group. Post-trabeculectomy IOP at day 1 and at months 1, 3, and 6 were comparable between both groups. At 12 months after trabeculectomy, mean IOP in the DSAEK group was lower compared to the PK controls (10.6 ± 3.2 vs 14.6 ± 8.5, P = .04), as shown in Table 2 . The mean IOP reduction at 12 months after trabeculectomy compared to baseline was 70.1% in the DSAEK group and 55.6% in the PK group.
| Trabeculectomy After DSAEK | Trabeculectomy After PK | P Value | |
|---|---|---|---|
| Pre-trabeculectomy IOP, mm Hg (SD) | 35.5 (10.1) | 32.9 (8.9) | .313 |
| Mean IOP at 12 months, mm Hg (SD) | 10.6 (3.2) | 14.6 (8.5) | .043 a |
| Proportion achieving IOP ≤18, % | 100 | 83.8 | .081 |
| Proportion achieving IOP ≤15, % | 90.0 | 64.9 | .059 |
| Proportion achieving IOP ≤12, % | 80.0 | 48.6 | .026 a |
| Without intervention | 87.5 | 77.8 | .660 |
| With intervention | 12.5 | 22.2 | — |
We compared the proportion of patients who had achieved IOP ≤18 mm Hg, IOP ≤15 mm Hg, and IOP ≤12 mm Hg in both surgical groups ( Table 2 ). The proportion of patients with IOP ≤12 mm Hg was significantly higher in the DSAEK compared to PK group (80.0% [16/20] vs 43.9% [18/41], P = .03). Importantly, of these eyes, 87.5% (14/16) did not require additional postoperative interventions such as reinstatement of topical antiglaucoma medications, needling with 5FU, or repeat glaucoma surgery to achieve an IOP ≤12 mm Hg, compared to 77.8% (14/18) in the PK group ( P = .66). Comparing the DSAEK to PK group, the proportion of eyes with IOP ≤18 mm Hg was 100% (20/20) vs 75.6% (31/41) ( P = 0.08), and that achieving IOP ≤15 mm Hg was 90.0% (18/20) vs 58.5% (24/41) ( P = .06) respectively.
Subgroup analysis by diagnosis of pseudophakic bullous keratopathy and Fuchs endothelial dystrophy was performed ( Table 3 ). In the pseudophakic bullous keratopathy group, IOP at 12 months was significantly lower in the DSAEK group compared to the PK group (10.2 ± 2.5 mm Hg vs 16.1 ± 6.8 mm Hg, P = .01), while the proportion achieving IOP ≤12 mm Hg was significantly higher in the DSAEK group (90.0% vs 30.8%, P = .01). In the group with Fuchs endothelial dystrophy, there was no difference in IOP at 12 months, nor in the proportion of patients achieving IOP ≤12 mm Hg, between the DSAEK and PK groups.
| Trabeculectomy After DSAEK | Trabeculectomy After PK | P Value | |
|---|---|---|---|
| Pseudophakic bullous keratopathy (n) | 10 | 13 | |
| Preoperative mean IOP, mm Hg (SD) | 32.9 (7.5) | 32.5 (11.0) | .92 |
| Postoperative 12-month mean IOP, mm Hg (SD) | 10.2 (2.5) | 16.1 (6.8) | .01 a |
| Proportion achieving IOP ≤12 mm Hg (n) | 90.0% (9) | 30.8% (4) | .01 a |
| Fuchs endothelial dystrophy (n) | 7 | 4 | |
| Preoperative mean IOP, mm Hg (SD) | 33.3 (10.1) | 33.8 (5.3) | .92 |
| Postoperative 12-month mean IOP, mm Hg (SD) | 10.1 (3.7) | 13.8 (5.1) | .27 |
| Proportion achieving IOP ≤12 mm Hg (n) | 71.4% (5) | 50.0% (2) | .58 |
The proportion of eyes that required postoperative intervention after trabeculectomy was considerably lower in the DSAEK group, but this difference did not attain statistical significance (20.0% [4/20] vs 36.6% [15/41], P = .15), Table 4 . Subgroup analyses did not show any difference. The proportions requiring IOP-lowering medications were 15.0% (3/20) DSAEK and 36.6% (15/41) PK ( P = .08), those requiring needling with 5FU were 20.0% (4/20) and 22.0% (9/41) respectively ( P > .99), and those requiring further glaucoma surgery were 0% (0/20) and 12.2% (5/41) respectively ( P = .15). Corneal graft failure arising after trabeculectomy was seen in 2 eyes (2/20, 10.0%) of DSAEK cases and in 4 eyes (4/41; 9.8%) of PK controls ( P > .99). No patients developed glaucoma during the course of study.
| Intervention Required at 12 Months, % | Trabeculectomy After DSAEK | Trabeculectomy After PK | P Value |
|---|---|---|---|
| All interventions | 20.0 | 39.5 | .155 |
| Reinstatement of IOP-lowering medications | 15.0 | 39.5 | .076 |
| Needling with 5FU | 20.0 | 23.7 | > .999 |
| Repeat glaucoma surgery | 0.0 | 13.2 | .153 |
| Graft failure | 10.0 | 10.5 | > .999 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


