Purpose
To report the results of oral mucosal graft for reconstruction of corneas with total limbal stem cell deficiency.
Design
Retrospective, interventional case series.
Methods
Seven patients (7 eyes) with total limbal stem cell deficiency caused by chemical burn (4 eyes), Stevens-Johnson syndrome / toxic epidermal necrolysis (1 eye), ocular cicatricial pemphigoid (1 eye), and multiple cryotherapies and application of mitomycin C for conjunctival melanoma (1 eye) were enrolled in this study. Oral mucosal graft was transplanted as a surrogate limbus together with amniotic membrane transplantation with a follow-up of at least 8 months. Symptomatic relief, restoration of a stable epithelium, corneal clarity, and the best-corrected visual acuity were assessed.
Results
Limbal stem cell deficiency was confirmed by impression cytology in all eyes, among which 6 were bilateral while 1 was unilateral. All 7 patients presented with severe loss of vision, photophobia, pain, chronic inflammation, and corneal vascularization and scarring. For 30 ± 19.8 months, pain and photophobia were resolved in all 7 eyes; 6 eyes exhibited a stable epithelium with regressed corneal vascularization and reduced chronic inflammation. Visual acuity was improved in all 7 eyes. One eye developed partial limbal stem cell deficiency due to exposure at 47 months follow-up and was reoperated. Five eyes had peripheral corneal vascularization.
Conclusions
Oral mucosal graft is a viable alternative for treating total limbal stem cell deficiency in eyes where transplantation of allogeneic limbal stem cells has failed or is not feasible.
The maintenance of a healthy corneal epithelium relies on stem cells located at the limbus. When limbal stem cells or their supporting stromal environment are destroyed, corneal blindness caused by limbal stem cell deficiency ensues. Histologically, corneas with limbal stem cell deficiency carry the hallmark of conjunctivalization associated with chronic inflammation, superficial neovascularization, scarring, and poor epithelial integrity. Consequently, patients with limbal stem cell deficiency experience severe photophobia and decreased vision and are not candidates for penetrating keratoplasty (PKP).
Several surgical reconstructive procedures have been proposed for treating limbal stem cell deficiency, all of which involve transplantation of either autologous or allogeneic limbal stem cells, depending on the laterality, the extent of involvement, and the patient’s compliance. For bilateral total limbal stem cell deficiency, transplantation of allogeneic limbal stem cells is achieved by either limbal conjunctival allograft from living, related donors or keratolimbal allograft from cadaveric donors, or both. However, transplantation of such allografts poses a high risk of rejection even in HLA-matched recipients, and its survival depends on long-term systemic combined immunosuppression.
To avoid the above disadvantages, clinical efficacy of cultivated autologous oral mucosal epithelial cell transplantation in treating limbal stem cell deficiency has been demonstrated in experimental and clinical studies. However, because of the easy harvesting of a free oral mucosal graft in a standard ophthalmic procedure, we wonder if direct transplantation of oral mucosal graft to the limbal region can also be an alternative of corneal surface reconstruction. In this study, we retrospectively reviewed the surgical outcome of 7 eyes of 7 patients undergoing oral mucosal graft for total limbal stem cell deficiency.
Patients and Methods
We retrospectively reviewed the medical records of 7 eyes of 7 patients (4 male and 3 female) with total limbal stem cell deficiency, all confirmed by impression cytology. The mean age was 41.9 ± 15 years (range, 24 to 65). The etiology of limbal stem cell deficiency included chemical burn (4 eyes), Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) (1 eye), ocular cicatricial pemphigoid (1 eye), and multiple cryotherapies and application of mitomycin C (MMC) for conjunctival melanoma (1 eye).
All 7 patients presented with severe loss of vision (from hand motion [HM] to 20/200), photophobia, pain, chronic inflammation, and corneal vascularization and scarring. Besides total limbal stem cell deficiency, other ocular surface deficits included severe symblepharon (4 eyes), aqueous tear deficiency confirmed by fluorescein clearance test (3 eyes), floppy eyelids (2 eyes), incomplete closure (2 eyes), infrequent blink (1 eye), infrequent blink with incomplete closure (1 eye), keratinization of the lid margin and the entire ocular surface (1 eye), and demodex mite infestation confirmed by microscopic examination (1 eye). One eye (Case 2) had undergone 3 keratolimbal allografts and 1 transplantation of ex vivo–expanded limbal stem cells, while another eye (Case 4) had received 2 keratolimbal allografts and 1 limbal conjunctival allograft from living, related donors. PKP was performed in 1 eye (Case 3) and deep anterior lamellar keratoplasty was performed in another eye (Case 4) to improve the visual acuity from HM to 20/200 and HM to count fingers (CF), respectively.
All patients were seen and managed at the Ocular Surface Center (Miami, Florida, USA) with a minimum postoperative follow-up of 8 months. As a routine practice at the Ocular Surface Center, all patients received photographic documentation of all preoperative and postoperative visits and digital recordings of their surgical procedures. Corneal epithelial integrity was evaluated by fluorescein staining, while corneal clarity was examined by slit-lamp examination and graded by a reported method. Chronic ocular inflammation was graded as mild (+), moderate (++), or severe (+++) according to a reported grading system, while corneal neovascularization was graded as 1 if limited to the periphery, 2 if extending to mid-periphery, 3 if involving the entire cornea, and 4 if massively involving the entire cornea. We retrieved the patients’ clinical data and summarized it in the Table .
| Case/Eye | Age/Sex | Cause | Prior Surgeries (No. of times) | Risk Factors | Surgical Procedures | Vision | Vascularization | Ocular Inflammation | FU (M) | Note | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chronic Inflammation | Sicca | Exposure | Symble-pharon | Simultaneous | Subsequent | Pre-op | Post-op | Change of Lines | Pre-op | Post-op | |||||||
| 1/OS | 65/F | OCP (OU) | N | ++ | N | Floppy eyelids | Y | OMG for fornix and tarsorrhaphy | N | CF/2 ft | 20/200 | 2 | 3 | 0 | − | 13 | Cataract |
| 2/OS | 54/M | CB (OU) | AMT, KLAL (3), ex vivo expansion of limbal stem cells | ++ | Y | Infrequent blink | N | N | PKP+ECCE+IOL | HM | CF/5 ft | 1 | 4 | 1 | − | 55 | N |
| 3/OS | 38/M | CB (OU) | LKP (2), PKP+Phaco+IOL, GDD | +++ | N | Infrequent blink | N | N | PKP; OMG for limbus | HM/1 ft | 20/200 | 3 | 2 | 1 | + | 49 | Demodex blepharitis; chronic active hepatitis |
| 4/OD | 32/M | CB (OU) | KLAL (2), DALK (2), lr-LCAL | ++ | Y | N | N | OMG for fornix and PKP | N | CF/1 ft | 20/70 | 5 | 4 | 1 | − | 20 | N |
| 5/OS | 24/M | CB (OU) | AMT, Sym+FR+AMT+ MMC, SK+AMT | ++ | N | Floppy eyelids | Y | N | N | 20/200 | 20/80 | 2 | 3 | 1 | − | 48 | N |
| 6/OS | 51/F | CM, MMC application and multiple cryotherapies (OS) | Excision of CM, application of MMC (10), cryotherapy (2) | ++ | N | Incomplete closure | Y | Tarsorrhaphy | N | CF/1ft | 20/200 | 2 | 4 | 2 | − | 8 | N |
| 7/OD | 29/F | SJS/TEN (OU) | N | + | Y | Infrequent blink, incomplete closure | Y | OMG for lids | N | HM | 20/400 | 2 | 4 | 0 | − | 17 | N |
Surgical Procedures
Prior to surgery, written informed consent regarding the surgical procedures, alternatives, risks, and benefits was obtained from each patient. Patients were also informed of the advantage and disadvantage of the off-label use of fibrin glue in ophthalmology. All surgeries were performed by the same surgeon (S.C.G.T.). The surgery was carried out under general anesthesia with the endotracheal tube secured to one side of the mouth so as to allow adequate exposure of the oral mucosa on the other side. Lid speculum or 4-0 black silk suture placed on the lid margin as a traction suture was used to open the eye. For all eyes, epinephrine (1:1000) (Hospira, Inc, Lake Forest, Illinois, USA) was applied for hemostasis control, while a 7-0 Vicryl (Ethicon, Somerville, New Jersey, USA) traction suture was placed at the superior and inferior limbal sclera to achieve adequate exposure. A 360-degree peritomy was followed by removal of pannus by blunt superficial keratectomy ( Figure 1 , Top row). For eyes with additional symblepharon, dissection was carried out to release it from the limbus along the bare sclera plane and recess it to the fornix. Fornix reconstruction was performed as described using 4-0 black silk mattress sutures. If the resultant corneal surface was still irregular, it was polished with a dental bur. The bare sclera was covered by a sheet of cryopreserved amniotic membrane (AM) (Bio-Tissue, Miami, Florida, USA) using fibrin glue (Tisseel; Baxter, Irvine, California, USA) ( Figure 1 , Middle row). If the corneal surface was not smooth or polished to be smooth, AM was extended to cover the entire cornea as a basement membrane substitute using fibrin glue. A free oral mucosal graft was then harvested from the inferior labial surface of the mouth cavity ( Figure 1 , Bottom left). After submucosal injection of 2% lidocaine with 1:100 000 epinephrine (Hospira, Inc), a freehand excision of the mucosal tissue 20 × 6 mm in size was obtained by demarcating the border with a super blade followed by scissor dissection. The donor site was covered by Gelfoam (Pharmacia & Upjohn, Kalamazoo, Michigan, USA) soaked with 1000 U/mL thrombin (Gentrac, Inc, Middleton, Wisconsin, USA). The excess fat was trimmed off from the stromal surface before the oral mucosal graft was divided in half to an L shape and was secured by 10-0 nylon interrupted sutures to be stretched to encircle the entire limbus ( Figure 1 , Bottom middle). Fibrin glue was used to further reinforce the attachment of the graft to the limbus. For eyes with lid margin keratinization, scarring, and distichiasis/trichiasis, these pathologies were removed and the anterior lamella was recessed, and the defect was covered by the remainder of a larger size of oral mucosal graft by a continuous 8-0 Vicryl suture (Ethicon). The finished ocular surface was covered by another sheet of AM as a patch by 10-0 nylon suture in a continuous purse-string fashion at 3 mm from the limbus or by placement of ProKera (Bio-Tissue) ( Figure 1 , Bottom right). In eyes with the problem of exposure attributable to poor blink or incomplete closure, tarsorrhaphy was performed by 4-0 black silk suture that was secured by a bolster made of 25-gauge intravenous tubing. Finally, the eye was patched with an ointment containing 0.3% tobramycin and 0.1% dexamethasone (Alcon Laboratories, Fort Worth, Texas, USA).
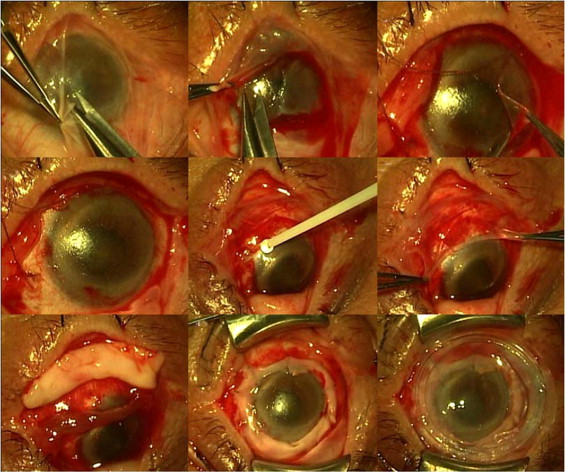
Postoperative Visits
After surgery, all patients received topical 0.3% ofloxacin (Allergan, Irvine, California, USA) 3 times a day and 1% prednisolone acetate (Allergan) or 0.1% unpreserved dexamethasone (prepared by the Ocular Surface Center under a state license for eyes with sensitivity to preservatives) 4 times daily. The former was discontinued when epithelialization was completed. At that time, the latter was tapered off over a course of 1 to 2 months. ProKera was removed in 2 weeks and replaced with a bandage contact lens with high Dk (Diffusionskonstante) to protect the healing of corneal epithelium. Sutures on the skin and the remaining sutures were removed at the 2-weeks-postoperative and 1-month-postoperative visit, respectively. A plastic goggle was recommended to protect the eyelids from compression during sleep for patients with floppy eyelid syndrome; lid scrub with 50% tea tree oil was prescribed as previously reported for patients diagnosed with ocular demodex infestation; and punctal occlusion either by plugs or thermal cauterization was performed in patients with aqueous tear deficiency. Patients with infrequent blink were encouraged to blink every 5 seconds with the aid of a timer. During each visit, patient’s symptoms including photophobia were recorded.
Outcome Measures
Outcome measures were determined by comparing the preoperative and postoperative data regarding any relief of subjective symptoms, resolution of chronic ocular inflammation and corneal neovascularization, restoration of corneal epithelial integrity and corneal clarity, and improvement of the best-corrected visual acuity.
Results
Limbal stem cell deficiency was confirmed by impression cytology in all eyes, among which 6 were bilateral while 1 was unilateral. Complete conjunctivalization of the entire limbus was observed with the presence of goblet cells in the central cornea. Chronic ocular inflammation ( Figure 2 , Pre-op) was graded as severe in 1 eye, moderate in 5 eyes, and mild in 1 eye. Corneal vascularization ( Figure 2 , Pre-op) was detected in all 7 eyes with grade 2 in 1 eye, grade 3 in 2 eyes, and grade 4 in 4 eyes.




