Purpose
To describe the optical coherence tomography angiography (OCT-A) features of treatment-naïve quiescent choroidal neovascularization (CNV) secondary to age-related macular degeneration, and to estimate the detection rate for neovascularization by means of OCT-A.
Design
Diagnostic tool validity assessment.
Methods
Treatment-naïve quiescent CNV were identified from a pool of patients at 2 retina referral centers. Patients underwent a complete ophthalmologic examination including fluorescein angiography, indocyanine green angiography, spectral-domain optical coherence tomography, and OCT-A. Detection rates of CNV by means of OCT-A were estimated with a second cohort of patients without CNV (negative controls).
Results
Twenty-two eyes of 20 consecutive patients with quiescent CNV were included. In 4 out of 22 eyes it was not possible to classify the CNV “shape,” “core,” “margin,” and “location,” either because the vascular network was not clearly shown (3 cases) or because it was not visible at all (1 case). CNV shape on OCT-A was rated as circular in 8 eyes and irregular in 10 eyes. CNV core was visible in 2 eyes. CNV margin was considered as well defined in 15 eyes and poorly defined in 3 eyes. CNV margin showed small loops in 9 eyes and large loops in the other 6 eyes. CNV location was foveal-sparing in 12 eyes. Sensitivity and specificity of quiescent CNV detection by OCT-A turned out to be 81.8% and 100%, respectively.
Conclusions
OCT-A allows the clinician to noninvasively identify treatment-naïve quiescent CNV and may be considered as a useful tool to guide the frequency of return visits and, possibly, make treatment decisions.
Treatment-naïve quiescent choroidal neovascularization (CNV) is a recently described entity in the setting of age-related macular degeneration (AMD). In quiescent CNV, newly diagnosed sub–retinal pigment epithelium CNV (type 1 neovascularization) detected on fluorescein angiography (FA) and indocyanine green angiography (ICGA) shows absence of intraretinal/subretinal exudation on repeated spectral-domain optical coherence tomography (SD OCT) for at least 6 months.
FA and ICGA are the current gold standard to diagnose CNV. On FA, quiescent CNV appears as a late-phase ill-defined hyperfluorescent lesion, without late-phase leakage or pooling of dye, which defines typical type 1 neovascularization. Similarly to active CNV, ICGA allows the visualization of the hypercyanescent neovascular network of quiescent CNV in early to mid phases and the delineation of the fibrovascular plaque in the late phase. SD OCT reveals, at the site of quiescent CNV, an irregular elevation of the retinal pigment epithelium (RPE) with major axis in the horizontal plane, with moderately reflective material in the sub-RPE space, no intraretinal/subretinal hyporeflective fluid, and clear visualization of hyperreflective Bruch membrane.
OCT angiography (OCT-A) is a novel and noninvasive diagnostic technique used to visualize ocular blood flow in retinal and choroidal vasculature. It is a dyeless, fast, 3-dimensional method, unlike traditional angiography imaging. Utility of OCT-A in CNV detection in different ocular conditions has been extensively described in the literature. In this study, we investigated the features of treatment-naïve quiescent CNV in AMD and estimated the detection rate for neovascularization by means of OCT-A.
Methods
In this observational case series, treatment-naïve quiescent CNV were identified from a pool of AMD patients consecutively presenting between September 2015 and December 2015 at 2 high-volume referral centers (the Medical Retina & Imaging Unit of the Department of Ophthalmology, University Vita-Salute San Raffaele in Milan, Italy; and the Department of Ophthalmology of University Paris Est, in Creteil, France). The study was conducted in agreement with the Declaration of Helsinki for research involving human subjects and was approved by the local institutional review board at both sites. Included patients signed a written general consent to participate in observational studies. Inclusion criteria for the study group were diagnosis of treatment-naïve “quiescent” CNV, defined as an irregular elevation of the RPE with moderately reflective material in the sub-RPE space, no intraretinal/subretinal hyporeflective fluid on SD OCT, late-phase ill-defined hyperfluorescent lesion, without late-phase leakage or pooling of dye on FA, and hypercyanescent neovascular network in early to mid phases with the delineation of the fibrovascular plaque in the late phase of ICGA; sufficiently clear ocular media; and adequate pupillary dilation and fixation to permit high-quality OCT imaging. Ocular exclusion criteria consisted of any disease other than AMD (including retinal vascular diseases, vitreoretinal diseases, history of central serous retinopathy, or macular dystrophies) and any previous intervention for CNV, such as anti–vascular endothelial growth factor (VEGF) intravitreal injections, photodynamic therapy, laser photocoagulation, or vitrectomy, in the study eye.
Each enrolled patient underwent a comprehensive ophthalmologic examination, including measurement of best-corrected visual acuity (BCVA), dilated slit-lamp anterior segment and fundus biomicroscopy, FA, ICGA, SD OCT (Spectralis + HRA; Heidelberg Engineering, Heidelberg, Germany), and OCT-A. OCT-A was performed through AngioPlex CIRRUS HD-OCT model 5000 (Carl Zeiss Meditec, Inc, Dublin, California, USA) or using AngioVue RTVue XR Avanti (Optovue, Fremont, California, USA). OCT-A was performed in all patients with a scanning area of 3 × 3 mm, centered on the foveal area.
AngioPlex uses optical microangiography (OMAG), a recently developed imaging technique that produces 3D images of dynamic blood perfusion within microcirculatory tissue beds at an imaging depth up to 2.0 mm. AngioPlex CIRRUS HD-OCT model 5000 contains an A-scan rate of 68 000 scans per second, using a superluminescent diode (SLD) centered on 840 nm. The resultant 3 × 3 angio cube contains 245 B-scan slices repeated up to 4 times at each B-scan position. Each B-scan is made up of 245 A-scans; each A-scan is 1024 pixels deep.
AngioVue relies on a split-spectrum amplitude-decorrelation angiography (SSADA) algorithm. This instrument has an A-scan rate of 70 000 scans per second, using a light source centered on 840 nm and a bandwidth of 50 nm. Each OCT-A volume contains 304 × 304 A-scans with 2 consecutive B-scans captured at each fixed position before proceeding to the next sampling location. Each A-scan is 2–3 mm deep. SSADA was used to extract the OCT angiography information. Each OCT-A volume is acquired in about 3 seconds and 2 orthogonal OCT-A volumes were acquired in order to perform motion correction to minimize motion artifacts arising from microsaccades and fixation changes.
Technical specifications of different devices are listed in Table 1 .
| Device | Algorithm | OCT SS | OCT AID | 3 × 3 AIV (B-scan) | AIS Retina |
|---|---|---|---|---|---|
| AngoPlex | SSADA | 68 000 scans/s | 2 mm | 245 A-scans | 3 × 3, 6 × 6 |
| AngioVue | OMAG | 70 000 scans/s | 2–3 mm | 304 × 304 A-scans | 2 × 2, 3 × 3, 6 × 6 |
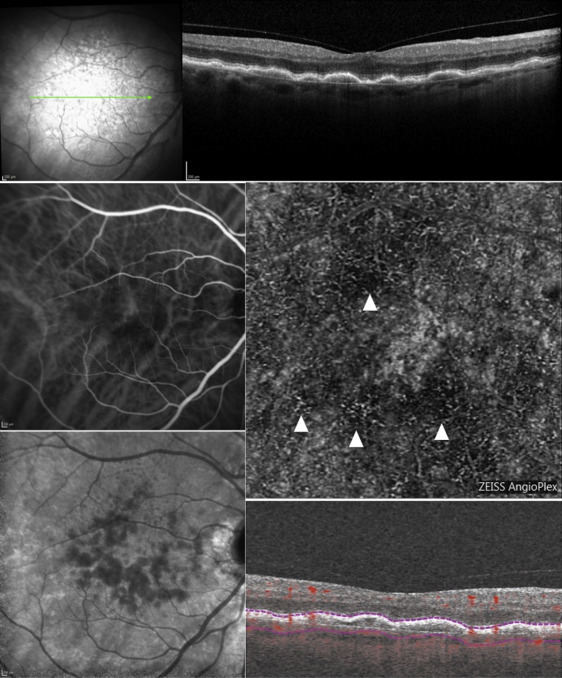
The automatic segmentation provided by the OCT-A software was manually adjusted by 2 expert retina specialists (G.Q. and F.C.) for correct visualization of the capillary plexus, outer retinal layers, and choriocapillaris, in order to better identify the CNV plane. The OCT-A images and corresponding OCT B-scans were assessed for CNV shape, CNV core, CNV margin and margin-loops, and CNV location. CNV shape was classified as “circular” or “irregular.” CNV core has been defined as a vessel of greater caliber or “trunk vessel” from which other, smaller vessels branch off. CNV core was classified as “visible” or “not visible”; if the core was visible, CNV was classified as “central core” or “eccentric core.” CNV margin on OCT-A was classified as “well defined” or “poorly defined” on the basis of its appearance and its borders; moreover, CNV margin was classified as “large loops” or “small loops” when the margin was “well defined.” CNV location was classified as “foveal involving” if the lesion involved the foveal center or “foveal sparing” if CNV lesion spared the foveal center.
To estimate sensitivity and specificity of OCT-A for neovascular detection (ie, diagnostic tool validity assessment), an additional cohort of 22 eyes of 22 patients with drusenoid pigment epithelium detachments (PED) derived from adjacent confluent soft drusen, as visualized by structural SD OCT with no evidence of any vascular network at ICGA, were used as negative controls. All these patients had been diagnosed with early non-neovascular AMD ( Figure 1 ). For these patients also, the study was conducted in agreement with the Declaration of Helsinki for research involving human subjects and was approved by the local institutional review board at both sites. Included patients signed a written general consent to participate in observational studies. Ocular exclusion criteria included any disease other than AMD (including retinal vascular diseases, vitreoretinal diseases, history of central serous retinopathy, or macular dystrophies), and anti-VEGF intravitreal injections, photodynamic therapy, laser photocoagulation, or vitrectomy in the study eye. OCT-A of the 2 cohorts were randomly presented to and independently evaluated by 2 expert readers (A.C. and M.V.C.) to confirm the presence/absence of CNV. If an eye was determined to have a CNV on ICGA, an OCT-A showing an abnormal neovascular network was considered to be a true positive; if a CNV was not visualized on OCT-A, the examination was considered to be a false negative. If the ICGA did not demonstrate a CNV (negative control group), an OCT-A with no evidence of a CNV was considered to be a true negative; if a CNV was detected, the examination was considered to be a false positive.
Statistical analysis was performed using SPSS software 21 (SPSS, Inc, Chicago, Illinois, USA). All data were expressed as mean ± standard deviation; continuous variables were assessed with a Student t test, while categorical variables were analyzed using χ 2 test. P values < .05 were considered to be statistically significant.
Results
Study Population and Choroidal Neovascularization Features
Twenty-two eyes of 20 consecutive patients (11 female/9 male; mean age 76.5 ± 6.9 years, range 58–88 years) with quiescent CNV secondary to AMD were enrolled (study group). Twelve eyes were enrolled at University Vita-Salute San Raffaele, Milan, Italy, and 9 eyes were enrolled at University Paris Est, Creteil, France. In all eyes, treatment-naïve quiescent CNV were defined as an irregular elevation of the RPE with moderately reflective material in the sub-RPE space; no intraretinal/subretinal hyporeflective fluid on SD OCT; late-phase ill-defined hyperfluorescent lesion, without late-phase leakage or pooling of dye on FA; and hypercyanescent neovascular network in early to mid phases with the delineation of the fibrovascular plaque in the late phase of ICGA. Of the 20 patients, 2 patients presented with bilateral quiescent CNV, 12 had received anti-VEGF injections (ranibizumab) for actively leaking CNV in the fellow eye, and 6 presented with nonexudative AMD (with no evidence of plaques at ICGA) in fellow eyes. Demographic data and angiographic features of the study group are listed in Table 2 .
| Patient No. | Age, y | Sex | Eye | Shape | Core | Margin | Margin Loops | Location | Device |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 80 | M | OD | IRR | NV | WD | LL | FS | AngioVue |
| 2 | 72 | F | OS | CIR | NV | WD | SL | FI | AngioVue |
| 3 | 79 | F | OS | CIR | NV | WD | SL | FI | AngioVue |
| 4 | 77 | F | OS | IRR | NV | WD | LL | FS | AngioVue |
| 5 | 81 | F | OD | IRR | NV | WD | LL | FS | AngioVue |
| 6 | 86 | F | OD | IRR | NV | PD | N.C. | FS | AngioVue |
| 7 | 77 | M | OD | IRR | NV | PD | N.C. | FS | AngioVue |
| 8 | 88 | M | OD | CIR | NV | WD | SL | FI | AngioVue |
| 9 | 72 | F | OD | IRR | NV | PD | N.C. | FI | AngioVue |
| 10 | 83 | F | OD | CIR | NV | WD | LL | FS | AngioPlex |
| 11 | 86 | M | OD | CIR | NV | WD | SL | FS | AngioPlex |
| 12 | 75 | M | OD | IRR | NV | WD | SL | FS | AngioPlex |
| 13 | 73 | F | OD | IRR | NV | WD | SL | FS | AngioPlex |
| 13 | OS | IRR | V | WD | LL | FI | AngioPlex | ||
| 14 | 75 | M | OD | CIR | V | WD | SL | FI | AngioPlex |
| 15 | 73 | F | OD | CIR | NV | WD | SL | FS | AngioPlex |
| 15 | OS | CIR | NV | WD | SL | FS | AngioPlex | ||
| 16 | 58 | F | OS | N.C. | N.C. | N.C. | N.C. | N.C. | AngioPlex |
| 17 | 75 | M | OS | IRR | NV | WD | LL | FS | AngioPlex |
| 18 | 73 | F | OD | N.C. | N.C | N.C | N.C | N.C. | AngioPlex |
| 19 | 80 | M | OS | N.C. | N.C. | N.C. | N.C. | N.C. | Both |
| 20 | 67 | M | OS | N.C. | N.C. | N.C. | N.C. | N.C. | AngioPlex |
In 12 eyes OCT-A examinations were acquired with AngioPlex, in 9 eyes with AngioVue, and in 1 eye with both devices. In 4 out of 22 eyes it was not possible to classify the CNV “shape,” “core,” “margin,” and “location,” either because the vascular networks were not clearly shown (3 cases) or because they were not visible at all (1 case; Figure 2 ); therefore, these eyes were excluded from the analysis. Thus, it was possible to analyze the OCT-A features in 18 out of 22 eyes.




