Purpose
To report a simple novel technique to facilitate preparation of Descemet membrane grafts for Descemet membrane endothelial keratoplasty (DMEK).
Design
Laboratory investigation and retrospective, single-center, consecutive case series.
Methods
Preparation of the endothelial graft is performed on an artificial anterior chamber, endothelial side up. After an incomplete circular superficial trephination, we describe a simple technique using a 27 gauge cannula to detach the Descemet membrane (DM). Endothelial cell density (ECD) was measured before dissection on 12 human corneas for research and 3 days after storage in organ culture. Histologic and electron microscopy analysis were performed. A DMEK was performed in 50 patients with Fuchs dystrophy. Visual acuity and ECD were evaluated 2 and 6 months after surgery.
Results
ECD was 2765 ± 256 cells/mm 2 on corneas for research before dissection and 2651 ± 305 cells/mm 2 after 3 days in organ culture ( P < .01). Histologic and electronic sections confirm that the cleavage was between DM and posterior stroma. Clinically, preparation of 2 corneas from a single donor was unsuccessful; 48 corneas were clear at 2 months and 47 at 6 months. At 2 months 77% of the patients had recovered a visual acuity of at least 20/30. At 6 months, 91.5% of the patients had a visual acuity of at least 20/30. ECD was 2656 ± 28 cells/mm 2 (range: 2450-3100 cells/mm 2 ) preoperatively, 1797 ± 41 cells/mm 2 (range: 1100-2700 cells/mm 2 ) at 2 months, and 1658 ± 43 cells/mm 2 (range: 900-2600 cells/mm 2 ) at 6 months.
Conclusion
We report here a reliable and efficient technique for the preparation of pure Descemet membrane grafts.
The use of endothelial grafts has greatly increased over the last 10 years, gradually replacing penetrating keratoplasty in cases in which a corneal graft is necessary following Fuchs dystrophy, bullous keratopathy, or the failure of previous penetrating keratoplasty. Endothelial graft techniques have proved to be superior, because the introduction of such grafts via a small incision can reduce astigmatism, prevent fragility following circular trephination, and accelerate visual recovery. Several techniques for preparation of the posterior graft are currently available. The most popular is known as Descemet stripping automated endothelial keratoplasty (DSAEK). This technique has the advantage of being simple, in terms of both the cutting of the graft and its introduction into the anterior chamber. The second technique is Descemet membrane endothelial keratoplasty (DMEK), in which the posterior graft includes only the Descemet membrane and the endothelium. The graft is prepared manually in this case. It is clear from published articles reporting the outcome of endothelial grafts and from retrospective series comparing these techniques that visual recovery is better after DMEK than after DSAEK, probably because of the absence of residual stroma. However, DMEK techniques are not particularly simple to carry out, and there is always a risk of endothelial tearing during the graft preparation procedure.
We describe here a new technique for the preparation of pure Descemet membrane grafts that seems simpler than previously described techniques. We present a detailed description of this technique and a histologic analysis of the prepared graft. We then evaluate the possible endothelial trauma induced by this method, using eyes from an eye bank, and we report a clinical evaluation of the use of grafts of this type in a series of patients.
Patients and Methods
The study consisted of a laboratory investigation and retrospective, single-center, consecutive case series. We carried out a retrospective, single-center, consecutive case series at Charles Nicolle University Hospital (Rouen, France). The institutional review board (IRB; CPP Nord-Ouest I) declared that the type of retrospective study waived the need for IRB approval, in accordance with French law on human clinical trials. Informed consent was obtained from each patient. The research adhered to the tenets of the Declaration of Helsinki.
Description of the Technique for Descemet Membrane Graft Dissection
For this study, we used 12 corneal grafts destined for use in research. These were normal tissues procured after relatives had been informed, as authorized by French bioethics law. They were not suitable for transplantation owing to inconclusive donor serology, despite normal endothelial characteristics. In all cases, the grafts were stored in organ culture at 31°C, the prevalent method used in European eye banks, in CorneaMax medium (Eurobio, Les Ulis, France) for 2-3 weeks.
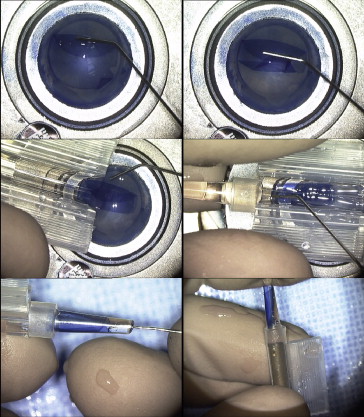
The technique is described in Figure 1 and Video 1 ( Supplemental Material , available at AJO.com ). A trephination blade with a diameter of 8-8.5 mm (Moria, Anthony, France) must first be broken to generate a fragment 3-4 mm long. The cornea to be dissected is positioned on the concave surface of an artificial anterior chamber (Moria) or any other concave material, with the endothelium uppermost. The blade is then held in Halstead forceps (Moria) and pressed against the corneal endothelium so as to section the Descemet membrane in a circular manner. Trephination of the Descemet membrane therefore occurs over 330 degrees, rather than 360 degrees. It is essential to curve the blade slightly toward the exterior after it has been broken to prevent the tearing of the Descemet membrane at the transition between the trephined section and the peripheral cornea (30 degrees). This ensures that any tearing occurs from the center to the periphery rather than in the opposite direction ( Figure 2 ). The depth of trephination is not important and can therefore just as easily be 50 μm as 500 μm. The essential point is the sectioning of the Descemet membrane over 330 degrees and the avoidance of perforating trephination. The circular trephination blade must be held in a vertical position and the graft must be well centered and flattened. No force should be applied to the rotation, to prevent membrane tearing. The artificial anterior chamber is then closed, with the endothelium still uppermost, and air enters the anterior chamber. This leads to an inversion of the cornea, with the endothelium bulging upwards. We used the Hanna 18095C artificial chamber (Moria), which was designed for the preparation of grafts for penetrating keratoplasty. However, any single-use artificial chamber can also be used. Air is injected into the anterior chamber at a sufficiently high pressure to turn the cornea over. This pressure was not measured, but should be sufficiently high for the cornea to be well stretched in the inverse position. An assistant may hold the air-filled syringe to maintain the pressure, or the tube supplying air may be clamped close to the anterior chamber. The surplus culture medium is then removed from the side with a microsponge and the endothelium is then stained with trypan blue (Eurobio). This is necessary to ensure perfect visualization of the zone of Descemet membrane trephination. The Descemet membrane must be sectioned over 330 degrees; if necessary, this can be achieved for nontrephined zones with a 15-degree knife.

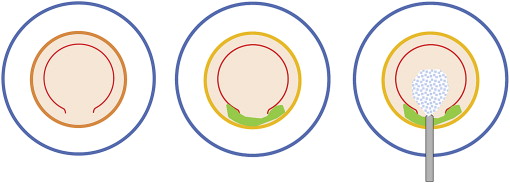
The graft is then rinsed with balanced salt solution (BSS) to remove excess trypan blue, and a cohesive viscoelastic droplet is placed on the endothelium to prevent its drying, particularly at the apex. It is then necessary to focus on the nontrephined zone (30 degrees). At this site, there is a zone of continuity between the central and peripheral endothelia. On either side of this zone of continuity, the peripheral endothelium can be detached very easily, in a single movement, with forceps. It is important to get hold of the peripheral Descemet membrane at the precise site of trephination. In this way there is no contact with the central endothelium, only with the peripheral zone, and, provided there is sufficient pressure in the anterior chamber, the Descemet membrane is easily detached. The peripheral endothelium is thus torn in the region of the zone of continuity in such a way as to create a small, easily lifted flap in this area. A small spatula or the jaws of a pair of Troutman forceps (Moria) should then be slipped into the opening to ensure the correct detachment of the Descemet membrane over a length of 2-3 mm. The rest of the dissection is then carried out with a 27 gauge cannula (FCI, Paris, France) mounted on a 2.5-mL syringe filled with culture medium or BSS. The 27 gauge cannula is slipped under the endothelium, towards the center of the cornea. The injection of the culture medium or BSS at this point readily detaches the endothelium by hydrodissection, in front of the cannula. Once the center of the endothelium is reached, it is straightforward to detach the Descemet membrane on either side, from right to left, up to the zone of trephination. It is then possible to extend the hydrodissection straight to the opposite zone of Descemet membrane trephination, and then to detach the endothelium on either side right up to the periphery. At the end of the dissection, the graft remains in contact with the underlying stroma because it is simply placed on top, not actually immersed. However, it is important to ensure that it does not slide too much to the side and to recenter it if necessary.
Analysis of Dissected Descemet Membrane Grafts
On the morning of dissection, for each graft, we evaluated endothelial density by the classical method: 2-3 drops of 0.4% trypan blue (Eurobio) were placed in the concavity of each cornea and incubated for 1 minute. The corneas were then immersed for 4 minutes in sterile 0.9% NaCl (packaged as single doses; Gilbert, Hérouville Saint-Clair, France). The trypan blue stained the nuclei of the dead cells and immersion in NaCl made it possible to determine cell density by dilating the intercellular spaces. The cornea was then placed under a light microscope and cell density was evaluated with a semi-automatic program (Image-Pro, Image Processing and Analysis Software; MediaCybernetics, Rockville, Maryland, USA). The percentage of dead cells was also determined by counting the cells with a blue-stained nucleus.
After this initial evaluation, the grafts were dissected as described above and then cultured for 3 days in Eurobio CorneaMax storage medium. For our evaluation of this technique with corneas not suitable for transplantation and in order to maintain Descemet membrane attached to the corneoscleral graft during storage, we left the Descemet membrane graft attached to the residual cornea at 2 small peripheral adhesion points at opposite ends, which we did not dissect. However, the detachment was clearly complete. Another endothelial evaluation was then carried out 3 days later.
Five corneas were fixed in 10% neutral buffered formalin and hematoxylin-eosin sections were examined for histology analysis. Five corneas were fixed in 1% glutaraldehyde/0.5% paraformaldehyde in 0.1 M mono Na/diK buffer (pH 7.4), postfixed in 1% osmium tetroxide in 0.1 M cacodylate buffer for 1 hour, dehydrated in a graded series of ethanol, and embedded in Epon resin. Ultrathin (90-nm) sections were cut and stained with uranyl acetate and lead citrate. Photographs of Descemet membrane and posterior stroma were taken with a transmission electron microscope (model H-800; Hitachi, Tokyo, Japan) equipped with a CCD camera (XR40; AMT, Danvers, Massachusetts, USA).
Patients
DMEK was performed by 1 surgeon (M.M.) on 50 consecutive eyes, in 50 patients, all suffering from primary Fuchs endothelial dystrophy with no other associated ophthalmologic diseases. The patient sample consisted of 28 women and 22 men, with a mean age of 65 ± 6 years (range: 45-78 years). Twenty-four patients underwent cataract surgery at the same time as DMEK, whereas the other 26 patients were already posterior chamber pseudophakic.
Snellen BCVA (best-corrected visual acuity), manifest refraction, and endothelial cell density were recorded 2 and 6 months after surgery. Eventual complications (graft dislocation, graft failure, graft rejection) were also recorded at 1 week, 2 weeks, 1 month, 2 months, and 6 months after surgery. Mean donor age was 64.4 years (range: 40-79 years). Corneas were stored for 18 days (range: 9-25 days) in tissue culture medium before dissection and graft.
In the postoperative period, all the patients were treated with eye drops containing a combination of dexamethasone and tobramycin 3 times daily for 2 months. They then received dexamethasone alone, in the form of eye drops administered twice daily for 2 months and then once daily for 1 year.
Postoperative cell density was measured with a Topcon SP-2000 specular microscope (Topcon, Saint-Denis, France), with the center’s method and the manufacturer’s calibration and software. Manual counts were averaged from 3 independent images.
Descriptive statistics for normally distributed variables are reported as means, standard deviations, and ranges. Differences between preoperative and postoperative values were analyzed in paired-difference t tests. Data were analyzed with GraphPad InStat software 3.0 (GraphPad Software, San Diego, California, USA). P values less than .05 were considered statistically significant.
Surgery
Descemet membrane implantation was possible in all cases. The endothelial graft, still stretched over the donor stroma, which was itself maintained on the artificial anterior chamber, was folded over itself, with the endothelium towards the interior, with the aid of the 27 gauge cannula, as indicated in Figure 3 and Video 2 ( Supplemental Material , available at AJO.com ). The Descemet membrane graft, rolled up with the endothelium on the inside, was guided into the chamber of an intraocular lens (IOL) injection cartridge designed for 2.2-mm implantation (Viscojet injector set 2.2; Medicel, Wolfhalen, Switzerland) as reported by Kruse and associates. A hook was then used to pull the graft close to the opening. Once the diseased endothelium had been removed from the patient via a 2.8-mm incision, the cartridge was fixed to the end of a cannula adapted to its internal diameter, which was then fixed onto a 2.5-mL syringe filled with BSS. The cartridge was introduced into the incision, rotating it through 180 degrees, and the endothelial graft was then injected into the anterior chamber by expelling the BSS from the syringe. At this point, the graft was placed in the anterior chamber with its stromal surface towards the cornea and the endothelial surface directed toward the iris. The graft was then progressively deployed by the injection of BSS, a process that may take some time. Ideally, the graft should be held in place with a 30 gauge needle mounted on a syringe containing BSS and introduced via puncture of the anterior chamber from above. Only when the graft appeared to be perfectly centered was an air bubble released to maintain the graft in the center of the cornea.