Purpose
To describe myofibroblastic metaplasia of corneal endothelial cells in 2 cases with impaired visual function despite complete graft adherence after Descemet membrane endothelial keratoplasty (DMEK).
Design
Interventional case series.
Methods
In 2 of 90 consecutive DMEK surgeries, the cornea failed to clear up to 6 months postoperatively despite complete graft attachment. After secondary penetrating keratoplasty, both corneal buttons were examined using histopathologic analysis and transmission electron microscopy.
Results
Light microscopy revealed distinct corneal endothelial cell attenuation with the presence of an abnormal posterior collagenous layer in both cases. Most of the remaining endothelial cells had an elongated fibroblast-like appearance with immunopositivity for α-smooth muscle actin indicative of myofibroblast metaplasia. Transmission electron microscopy showed a slightly thickened Descemet membrane with an abnormal posterior fibrillar collagenous layer and a myofibroblast-like transformation of the remaining endothelial cells. Descemet membrane grafts closely adjoined the collagen lamellae of the host corneal stroma similar to the Descemet membrane–stroma interface of a normal cornea.
Conclusion
Myofibroblastic metaplasia of attenuated corneal endothelial cells with formation of an abnormal posterior collagenous layer may contribute to an impaired visual function despite complete graft adherence after Descemet membrane endothelial keratoplasty (DMEK).
Posterior lamellar keratoplasty is currently replacing penetrating keratoplasty (PK) for corneal endothelial disorders because it offers faster visual rehabilitation, improved surface topography, and reduced postoperative astigmatism as well as preservation of the integrity of the stroma. Among the techniques described so far, Descemet stripping automated endothelial keratoplasty (DSAEK) and Descemet membrane endothelial keratoplasty (DMEK) offer the greatest potential. In DMEK a thin graft consisting of Descemet membrane and endothelium is transplanted after stripping the recipient Descemet membrane. In theory, DMEK offers functional advantages over DSAEK because it leads neither to an increase of corneal thickness nor to a change of the posterior corneal curvature. Despite recent improvements, DMEK is technically demanding and the surgical procedure by itself has a significant impact on postoperative function. At present, the influence of key elements of the surgery (eg, graft preparation, manipulation in the anterior chamber, and air injection) on endothelial function are unknown. Furthermore, the mechanism of graft adhesion to the denuded corneal stroma is not understood.
While histopathology of failed DSAEK grafts suggests that donor endothelial cell loss causes postoperative edema and possibly graft dehiscence, the underlying cause of primary graft failure after DMEK is still obscure. Investigating Descemet membrane grafts adhering to the corneal stroma of recipients, we hereby report that myofibroblastic metaplasia of endothelial cells can occur after DMEK, which offers therapeutic implications for the future. Furthermore, our data suggest that late graft adhesion in DMEK seems to depend not only on endothelial function.
Methods
Ninety consecutive DMEK surgeries were performed over a period of 12 months starting in June 2009. While 85 grafts stayed clear with marked improvement of visual acuity, 5 grafts were classified as early graft failure. Descemet membrane was chronically detached and graft failure was attributed to this fact in 3 of the 5 patients, while slit-lamp optical coherence tomography (SL-OCT, Heidelberg Engineering, Lübeck, Germany) confirmed complete graft adherence in the 2 patients who are presented here ( Figure 1 ). Out of the 5 patients, 3 opted for PK instead of repeat DMEK or DSAEK.
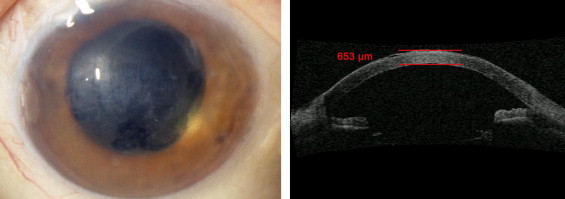
Both patients (a 70-year-old woman and a 55-year-old man) had suffered from pseudophakic bullous keratopathy. They had received donor tissue (donor age 61 and 68 years, postmortem time 5 and 7 hours, endothelial cell density 2454 and 2753 cells/mm 2 ) that had been preserved at 4C for 264 and 312 hours. Both surgeries were performed, as previously described, in the beginning of our series and uneventful surgery was characterized by prolonged manipulation in the anterior chamber. One single air injection 3 weeks after surgery was required because of peripheral graft detachment in 1 of the patients. At 6 months best spectacle-corrected visual acuity (BSCVA) failed to exceed 6/60 and 6/38, respectively; SL-OCT confirmed graft adherence over the entire area of the graft in both patients; central corneal thickness measured by Pentacam (Oculus, Wetzlar, Germany) decreased from 904 and 731 μm before surgery to 707 and 695 μm 5 days after DMEK and varied afterwards between 669 and 683 μm as well as 645 and 662 μm up to 6 months follow-up; and endothelial cell density was not measurable during the entire follow-up using specular microscopy (SeaEagle; Hightec American Industrial Laboratories, Lexington, Massachusetts, USA) in both patients because of a cloudy cornea. Both patients underwent secondary PK 6 months after DMEK surgery.
Corneal buttons were cut into halves and processed for histopathologic analysis and transmission electron microscopy as previously described.
Results
Light microscopy revealed distinct corneal endothelial cell attenuation. Mean endothelial cell count was 4.2 ± 0.8 cells (range, 3-5) and 5.0 ± 2.5 cells (range, 1-7) (normal range: >120 cells) measured in 8 consecutive high-power fields (magnification ×400) per slide in 5 consecutive serial sections ( Figure 2 , Left). Most of the remaining endothelial cells had an elongated fibroblast-like appearance ( Figure 2 , Top right). Immunostaining revealed the presence of α-smooth muscle actin (α-SMA) indicative of myofibroblast metaplasia ( Figure 2 , Bottom right). An abnormal posterior collagenous layer was present in both cases. Because of tissue preparation and processing in the histology laboratory, Descemet membrane was partly detached and the host corneal stroma disclosed moderate edematous changes.
Transmission electron microscopy showed a slightly thickened Descemet membrane (15-18 μm) consisting of a normal anterior banded (2.8-3.2 μm) and a normal posterior nonbanded (9.8-12.6 μm) layer as well as an abnormal posterior fibrillar collagenous layer (2.5-4.0 μm). The posterior collagenous layer was primarily composed of randomly arranged collagen fibrils interspersed among amorphous basement membrane material ( Figure 3 ). Remaining endothelial cells displayed myofibroblast-like transformation ( Figure 3 , Bottom left and right). Descemet membrane grafts closely adjoined the collagen lamellae of the host corneal stroma ( Figure 3 , Top right). The Descemet membrane–stroma interface appeared similar to that of a normal cornea ( Figure 4 ).




