Purpose
To evaluate the morphologic features and viability of human retinal pigment epithelium (RPE) cells captured from the pigment epithelium detachment (PED) region outside the choroidal neovascular membrane lesion in eyes with hemorrhagic age-related macular degeneration.
Design
Prospective, observational case series.
Methods
Five specimens of the RPE sheet were obtained from the PED region after choroidal neovascular membrane excision in 5 eyes of 5 patients during RPE transplantation for hemorrhagic age-related macular degeneration. The specimens were stained with hematoxylin and eosin and with periodic acid–Schiff. Immunohistochemistry analysis for RPE-65 and zonula occludens-1 also was performed. RPE cells from the PED region were cultured and passaged 5 times. Scanning and transmission electron microscopy were performed to analyze the specimens.
Results
The RPE cells of the specimens contained brownish pigment and were autofluorescent in vitro. Periodic acid–Schiff staining revealed that the Bruch membrane below the RPE monolayer was not intact. The specimens demonstrated positive results for both zonula occludens-1 and RPE-65 staining. The RPE basement membrane in the specimen was observed by both scanning and transmission electron microscopy. Intercellular tight junctions among RPE cells of the specimen also were observed. RPE cells captured from the PED region were cultured successfully and were passaged 5 times.
Conclusions
The RPE sheet developed from the PED region outside the choroidal neovascular membrane lesion had tight intercellular junctions, a simple RPE basement membrane, and active cellular viability. This monolayer RPE sheet may be considered as a substitution for subfoveal RPE loss in eyes with hemorrhagic age-related macular degeneration.
Retinal pigment epithelium detachment (PED) is a prominent feature of many chorioretinal diseases, the most prevalent of which is exudative age-related macular degeneration (AMD). To date, however, no effective therapies for AMD with PED have been established.
Massive submacular hemorrhage and subfoveal retinal pigment epithelium (RPE) apoptosis play some role in the photoreceptor damage that occurs in hemorrhagic AMD with PED, causing subsequent severe vision loss. Modern vitreoretinal surgical techniques allow for safe subretinal hemorrhage debridement after a 180-degree peripheral retinotomy combined with flipping the temporal retina toward the nasal side or through a parafoveal incision. An autologous RPE cell transplantation to the subfoveal area theoretically can restore central vision in patients with hemorrhagic AMD who have submacular choroidal neovascular membrane (CNVM) and subfoveal RPE loss at presentation, and the results of subretinal injection of an autologous RPE cell suspension were encouraging at the 1-year follow-up. Disorganized piles of RPE cell deposits instead of the tightly formed RPE monolayer, however, limit the clinical applicability of this technique.
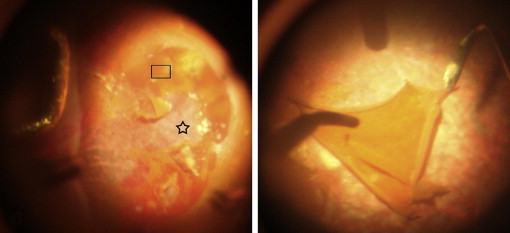
On many occasions, we have observed PED in hemorrhagic AMD eyes when performing the RPE–Bruch membrane complex transplantation procedure ( Figure 1 , Left). Meanwhile, an intact sheet could be formed in the PED region induced by hemorrhage and exudation originating from the adjacent CNVM or an underlying choroidovasculopathy ( Figure 1 , Right). More interestingly, the apparently intact RPE sheet obtained from the PED region can tolerate surgical manipulation. So, we further wondered (1) whether the detached RPE cells could be rescued and could function normally after they were reattached and (2) whether the detached RPE layer could be transplanted into the subfoveal RPE denuded region to restore vision in hemorrhagic AMD eyes. If so, such a procedure would be less traumatic and easier to manipulate than the previous procedures applied to treat hemorrhagic AMD. Considering the potential damage caused by sub-RPE fluid and hemorrhage, it is necessary to determine whether the morphologic features and viability of RPE cells are affected in PED regions where there is no CNVM before applying RPE monolayer graft transplantation in eyes with hemorrhagic AMD.
Methods
All patients and their family members provided written informed, special-designed consent regarding the possible removal of the RPE sheet and the use of the tissue in a research study, and they were informed as to the potential risks and possible poor outcomes of the surgery. This study was performed with the approval of the Institutional Review Board of Peking University Health and Science Center (reference no. IRB00001052-09070) and was conducted in accordance with the tenets of the Declaration of Helsinki.
Human Retinal Pigment Epithelium Sheet Capture
This prospective, observational single-center pilot study included 5 patients with hemorrhagic AMD. The first 4 patients underwent RPE–Bruch membrane complex transplantation, and the fifth patient underwent an RPE monolayer sheet transplantation. Five specimens of the RPE sheet were obtained from the PED region approximately 1 papilla diameter temporally away from the CNVM margin during the surgery.
The surgical procedure was described previously. After performing 180-degree peripheral retinectomy, the temporal retina was flipped toward the nasal side. Under direct observation, one of the authors (Z.M.) performed a complete choroidal neovascularization excision and debrided the subretinal and sub-RPE hemorrhage. The naturally produced PED region, which was next to the choroidal neovascularization lesion, clearly was exposed to the vitreous cavity ( Figure 1 , Left). An RPE sheet (3 × 2 mm) with no extra attachment of CNVM or blood clots was captured successfully from the PED region in the first 4 eyes, and a 1.5 × 1.5-mm specimen was captured from the PED region of Patient 5 ( Figure 1 , Right). All RPE sheet specimens, which were removed from the eyes through a 20-gauge suctioning tube, provided enough samples to be stained and cultured.
Tissue Preparation
Specimens 1 and 2 were separated into 2 pieces using microscissors while they were supported on a cellulose acetate membrane during surgery. One half of each of these 2 samples and all of specimen 5 were used for RPE cell culture. The other half of specimens 1 and 2 were examined first by a confocal laser scanning microscope (510 META; Carl Zeiss, Oberkochen, Germany) for in vitro autofluorescence using a 488-nm and 594-nm beam. While the samples were kept in a balanced salt solution, they were treated for different purposes: the other half of specimen 1 was examined for the presence of RPE-65; the left part of specimen 2 was used for zonula occludens (ZO)-1 staining. One half of specimens 3 and 4 was frozen in optimum cutting temperature compound and was cut in 7-μm thick sections using a cryostat (LEICA CM 1900; Leica, Bensheim, Germany) for hematoxylin and eosin staining and periodic acid–Schiff staining. The other half of specimens 3 and 4 was used for scanning and transmission electron microscopy, respectively.
Retinal Pigment Epithelium Cell Culture
Three RPE sheet specimens (specimens 1, 2, and 5) were placed in culture media at 37 C under a microscope. The RPE cells were dissociated from the sheets in a solution containing 0.25% trypsin and 0.02% ethylenediamine-tetraacetic acid (Life Technologies, Grand Island, New York, USA) at 37 C for 30 minutes. Afterward, the enzyme-treated tissues were dissociated into single RPE cells by gentle pipetting and centrifugation. The harvested RPE cells were resuspended in Dulbecco modified Eagle medium: Nutrient Mixture F-12 (DMEM/F-12; Life Technologies, Grand Island, New York, USA) supplemented with 10% fetal bovine serum (Life Technologies; Paisley, United Kingdom). The RPE cells were seeded into 1 well for each specimen in a 24-well culture plate (Corning, New York, USA) and were incubated at 37 C in a humidified atmosphere of 5% carbon dioxide and 95% air. The Dulbecco modified Eagle medium was exchanged with fresh medium every 2 days. After the cells grew to confluency, they were trypsinized and passaged. Some RPE cells were reseeded in a 24-well culture plate and were analyzed morphologically with phase-contrast microscopy. Other cells were reseeded on poly-L-lysine–coated cover glass (Corning) and were used for immunohistochemistry studies.
Immunohistochemistry Analysis
Some of the cultured RPE cells from specimens 1, 2, and 5 were fixed with cold methanol and acetone. They then were blocked with the appropriate normal serum in phosphate-buffered saline (PBS) for 1 hour at room temperature. They then were incubated at 4 C overnight with mouse anti–RPE-65 monoclonal antibody (1:100; Millipore Corporation, Temecula, California, USA). After being washed in PBS, the sections were incubated further with goat antimouse secondary antibody with tetramethylrhodamine isothiocyanate (Sigma-Aldrich, St. Louis, Missouri, USA) for 45 minutes at 37 C. Control sections were treated in the same way, but with no primary mouse anti–RPE-65 monoclonal antibody. Cell nuclei were counterstained with 4,′6′-diamino-2-phenylindole (Sigma-Aldrich).
The other half of specimens 1 and 2 was fixed with 4% paraformaldehyde. Then the 2 samples were tested for RPE-65 and ZO-1 staining, respectively. After being blocked with the appropriate normal serum in PBS for 1 hour at room temperature, specimen 1 was incubated at 4 C overnight with mouse anti–RPE-65 monoclonal antibody (1:100; Millipore, Billerica, Massachusetts, USA). After being washed in PBS, the sections were incubated further with horseradish peroxidase-labeled secondary antibody (Golden Bridge International, Inc, Mukilteo, Washington, USA) for 45 minutes and were developed with diaminobenzidine-tetrahydrochloride (Life Technologies, Paisley, United Kingdom). Specimen 2 was incubated at 4 C overnight with rabbit anti–ZO-1 polyclonal antibody (1:100; Santa Cruz Biotechnology, Santa Cruz, California, USA), and after being washed in PBS, the sections were incubated further with goat antirabbit secondary antibody with fluorescein isothiocyanate (Sigma-Aldrich) for 45 minutes at 37 C. Control sections were treated in the same way, but with no mouse anti–RPE-65 monoclonal or rabbit anti–ZO-1 polyclonal antibody.
Electron Microscopy Analysis
Scanning electron microscopy
Specimen 3 was fixed further in half-strength modified Karnovsky fixative. After rinsing in PBS, the specimen was dehydrated in a graded series of ethanol, critical point dried, and sputter-coated with 20 nm gold and palladium. The sample then was examined with a scanning electron microscope (JSM-5600LV; JEOL, Tokyo, Japan).
Transmission electron microscopy
Specimen 4 was fixed with 3% glutaraldehyde and postfixed with 1% osmium tetroxide. It was processed for ultrathin sectioning. The sections were stained with uranium acetate and lead citrate, and the ultrastructural observation of RPE cells and underlying attached membrane then were evaluated using a transmission electron microscope (JEM-1230; JEOL).
Results
Specimen Morphologic Features
Confocal microscopy revealed in vitro autofluorescence of the RPE cells on the specimens ( Figure 2 ). The RPE cells of the specimen contained brown pigment with transparent membrane attached below ( Figure 3 ). The sectioned specimen stained with hematoxylin and eosin showed that the RPE cells contained pigment and aligned regularly in a monolayer with no blood clot, CNVM, or choriocapillary attached below ( Figure 4 ), and no staining of the Bruch membrane underlying the monolayer of RPE cells was detected by periodic acid–Schiff staining ( Figure 5 ).
Retinal Pigment Epithelium-65 and Zonula Occludens-1 Expression
The RPE-65 expression was strongly positive on specimen 1. The RPE cells of the specimen appeared regular and contained brownish pigment ( Figure 6 ). The results for expression of ZO-1 were positive on specimen 2, and each hexagonal structure represented a single RPE cell ( Figure 7 ).
Electron Microscopy
Scanning electron microscopy
On the margin of specimen 3, a thin membrane underlying the RPE monolayer was observed on the RPE denuded region. Loosely arranged collagen fibrils were embedded in the membrane ( Figure 8 , Top).
Transmission electron microscopy
Specimen 4 had an approximately 0.3-μm thick RPE basement membrane underlying the RPE cell monolayer with fibrils attached and a large number of complex infoldings on the external RPE cell membranes. The specimen contained shedded rod outer segments embedded in the RPE microvilli ( Figure 8 , Middle). The apical dark region between adjacent RPE cells indicated the existence of tight junctions ( Figure 8 , Bottom).
Cultured Retinal Pigment Epithelium Cells
After 7 days in primary culture, RPE cells that appeared polygonal had proliferated to confluence and contained some pigment within the cells ( Figure 9 , Left). RPE-65 staining results were positive from primary RPE cells from specimens 1, 2, and 5 ( Figure 9 , Right). During the subsequent passages, the pigmentation of the RPE cells decreased gradually and the shape of the RPE cells became spindle-like. By the fifth passage, all the RPE cells were completely nonpigmented and demonstrated negative results for RPE-65 expression (data not shown). The major observational findings from the 5 specimens are listed in Table 1 .
| Specimen Number | Test | Observation Results |
|---|---|---|
| 1 | RPE cell culture | Primary cells were RPE-65 positive, whereas passaged cells were RPE-65 negative and appeared as spindle-like nonpigmented cells |
| Autofluorescence | Positive | |
| RPE-65 staining (cell) | Positive | |
| 2 | RPE cell culture | Primary cells were RPE-65 positive, whereas passaged cells were RPE-65 negative and appeared as spindle-like nonpigmented cells |
| Autofluorescence | Positive | |
| ZO-1 staining (cell) | Positive | |
| 3 | SEM observation | Thin membrane underlying RPE layer was detected |
| HE staining (graft) | RPE cells regularly aligned in a monolayer | |
| 4 | TEM observation | RPE basement membrane was positive; cell membrane infoldings and ROS were detected |
| PAS staining (graft) | Negative (no intact Bruch membrane was observed underlying the RPE layer) | |
| 5 | RPE cell culture | Primary cells were RPE-65 positive, whereas passaged cells were RPE-65 negative and appeared as spindle-like nonpigmented cells |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


