Purpose
To investigate the morphologic characteristics of macular complications of dome-shaped maculas using swept-source optical coherence tomography (OCT).
Design
Retrospective observational case series.
Methods
Axial length measurements and swept-source OCT were performed in 49 highly myopic eyes (in 5 male and 30 female subjects) with dome-shaped maculas. We classified the dome patterns and measured the central retinal thickness, central choroidal thickness, central scleral thickness, and the macular bulge height, and assessed the associations of these parameters with macular complications.
Results
The central scleral thickness was significantly negatively correlated with age and the axial length. We classified the eyes into 3 groups: 6 with choroidal neovascularization (CNV group), 8 with retinal pigment epithelial detachment (PED group; 5 with serous retinal detachment), and 35 with no complications (no complications group). Nine eyes had a round dome and 40 had horizontally oriented oval-shaped domes. There were no significant differences in the frequency of macular complications between these patterns. The CNV group was significantly older and had a longer axial length than the other groups. The PED group had significantly larger values for both the central scleral thickness and bulge height than the other groups. The central choroidal thickness was significantly thinner in the CNV group than in the no complications group.
Conclusion
A dome-shaped macula results from relative thickening of the macular sclera, and this may lead to PED. Thinning of the sclera owing to long-term changes and elongation of the axis may develop CNV and cause visual impairment.
A dome-shaped macula was first reported by Gaucher and associates as a finding of highly myopic eyes, characterized by a forward bulge of the macula within the posterior staphyloma detected by optical coherence tomography (OCT). They suggested that the macular bulge was caused by the thickening of the choroid. Subsequently, Imamura and associates, using enhanced depth imaging (EDI)-OCT, reported that the macular bulge was the result of the thickening of macular sclera. Later, Ellabban and associates, through a 3-dimensional (3-D) analysis of prototype swept-source OCT, reported a finding of 2 types of morphology: the dome shape (17.6%) and the horizontal band shape (82.4%). Recently, Caillaux and associates classified the dome shape into 3 subtypes using EDI-OCT: the round dome (20.8%), the horizontally oriented oval-shaped dome (62.5%), and the vertically oriented oval-shaped dome (16.7%). In addition, complications such as serous retinal detachment (SRD) and choroidal neovascularization (CNV) were observed in the macular region of the dome-shaped macula, which caused visual impairment.
Recently, the development of OCT has made remarkable progress. In particular, it has made it possible to properly penetrate the retinal pigment epithelium by swept-source OCT, in contrast to the spectral-domain OCT, and to observe the choroid and sclera using a light source at 1 μm wavelength. In the present study, we investigated the morphologic characteristics of dome-shaped macula and the correlations between these characteristics and the macular complications using a swept-source OCT (DRI OCT-1; Topcon Corp, Tokyo, Japan) that recently became available.
Methods
A retrospective study was conducted on 49 eyes in 35 patients (5 male, 30 female) with dome-shaped macula based on OCT images. Using our database, we selected 528 highly myopic eyes of 276 Japanese patients with refraction of −6 diopters or more, or those with an axial length of 26 mm or more, from the patients seen at Tsukazaki Hospital from the beginning of September 2012 to the end of April 2013. This research adhered to the Declaration of Helsinki and was approved by the ethics committee of Tsukazaki Hospital. All of these patients had undergone a complete ophthalmologic examination, including assessments of the best-corrected visual acuity, refractive errors, axial length measurement using ocular biometry (IOLMaster; Carl Zeiss Meditec, Jena, Germany), intraocular pressure, color fundus photography, and swept-source OCT. Except for 1 patient with a history of allergy, the eyes with the complications of SRD and CNV were evaluated with fluorescein angiography and indocyanine green angiography, and the diagnoses were thus confirmed.
All layers of the choroid and sclera were scanned using swept-source OCT at a speed of 100 000 A-scans/second. Thirty-two images, where each image consisted of 1024 A-scans, were averaged to obtain the vertical and horizontal raster scans of 12 mm through the fovea. In addition, the 6 × 6-mm region centered on the fovea was scanned by a fast raster scanning protocol to obtain the 3-D imaging data sets. The A-scan density was 512 lines (horizontal) × 256 lines (vertical).
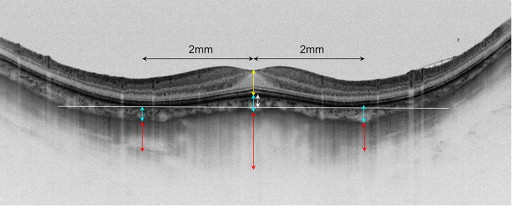
The dome-shaped macula diagnosis was based on the findings of a forward bulge of the macular region by 3-D OCT. The OCT images of vertical and horizontal sections were used to measure the height of the inward bulge of the retinal pigment epithelium (RPE) above the tangent line of the RPE of 2 outward concavities at the bottom of the posterior staphyloma ( Figure 1 ). Some studies have reported that bulges are easy to locate, especially on the vertical section; however, Caillaux and associates recommended an observation of both vertical sections and cross-sections, owing to the possible presence of a vertically oriented oval-shaped dome. Accordingly, we measured the bulge height based on the vertical and horizontal sections, and included the patients who had at least 1 section showing a bulge of 50 μm or more in our study, which was the same criterion used by Ellabban and associates. In addition, we measured the central retinal thickness, defined as the distance from the inner limiting membrane to the RPE; the central choroidal thickness, defined as the distance from the RPE to the choroid–sclera interface; and the central scleral thickness, defined as the distance from the choroid–sclera boundary to the outer scleral border. We also measured the choroidal and scleral thicknesses in the surrounding parafoveal regions 2 mm nasal, temporal, superior, and inferior to the fovea.
Statistical Analysis
The statistical analyses, including the Kruskal-Wallis 1-way analysis of variance, Mann-Whitney U test, and a single regression analysis, were performed using a commercially available software program (JMP, version 10.0; SAS Institute, Inc, Cary, North Carolina, USA). A P value <.05 was considered to indicate a significant difference.
Results
The mean age of the patients was 63.7 ± 11.5 years, the mean refractive error was −11.2 ± 4.4 diopters, the mean axial length was 28.5 ± 2.1 mm, and the mean intraocular pressure was 13.5 ± 2.8 mm Hg. The mean logMAR best-corrected visual acuity (BCVA) was 0.083 ± 0.28. The mean choroidal thickness was 98.8 ± 56.9 μm at the fovea, 56.3 ± 37.2 μm nasally, 97.8 ± 56.0 μm temporally, 102.9 ± 65.3 μm superiorly, and 89.8 ± 60.4 μm inferiorly. The choroidal thickness at the nasal was significantly thinner than that at the fovea ( P = .0002). The mean scleral thickness was 502.2 ± 152.8 μm at the fovea, 345.0 ± 103.5 μm nasally, 282.9 ± 115.4 μm temporally, 307.6 ± 125.3 μm superiorly, and 284.7 ± 107.2 μm inferiorly. The scleral thickness at the center was significantly greater than that at parafoveal areas in all quadrants ( P < .0001, respectively). The mean bulge height was 123.2 ± 82.5 μm and was correlated with the difference between the thickness of the fovea and the average thickness of the surrounding area (mean bulge height = 40.3 + 0.42 × difference between the thickness of the fovea and the average thickness of the surrounding area, R 2 = 0.30; P < .0001, Figure 2 ) The central scleral thickness was significantly negatively correlated with the patient age (central scleral thickness = 972.6 − 7.5 × age, R 2 = 0.33; P < .0001, Figure 3 ) and the axial length (AL) (central scleral thickness = 1626.8 − 39.4 × AL, R 2 = 0.31; P < .0001, Figure 4 ), while it tended to be positively correlated (borderline significance) with the central choroidal thickness (central scleral thickness = 427.9 + 0.75 × central choroidal thickness, R 2 = 0.08; P = .052, Figure 5 ).
In our database, 45 eyes with a dome-shaped macula and 336 eyes without dome-shaped macula that had both refractive data and axial length data and had no history of cataract surgery or refractive surgery were available for the analysis. In these eyes, a refraction of −6 diopters or more was noted in 43/45 eyes (95.6%) with a dome-shaped macula and 332/336 eyes (98.8%) without a dome-shaped macula, and among the eyes with an axial length of 26 mm or more, there were 42/45 eyes (93.3%) with a dome-shaped macula and 316/336 eyes (94.0%) without a dome-shaped macula. There were no major differences in the occurrence of a dome-shaped macula based on these parameters.
Based on the scans of the 6 × 6-mm region centered on the fovea, macular complications were observed in 14 eyes (macular complications group), and no complications were observed in 35 eyes (no complications group). The comparison of the 2 groups by Mann-Whitney U test indicated that the macular complications group had a lower mean logMAR BCVA (macular complications group 0.299 ± 0.37, no complications group −0.003 ± 0.17; P = .003), thinner central choroidal thickness (macular complications group 66.1 ± 36.9 μm, no complications group 111.9 ± 58.6 μm; P = .012), and greater bulge height (macular complications group 187.6 ± 114.8 μm, no complications group 97.5 ± 46.7 μm; P = .001). The central scleral thickness had a tendency to be higher in the macular complications group, but without any significant differences between the groups (macular complications group, 582.7 ± 200.4 μm; no complications group, 470.0 ± 117.8 μm; P = .059). No significant differences were observed between the 2 groups in terms of sex, age, intraocular pressure, refraction, axial length, and central retinal thickness ( Table 1 ).
| Characteristic | With Macular Complications | No Complications | P Values (Mann-Whitney U Test) |
|---|---|---|---|
| Number of eyes | 14 | 35 | |
| Sex (male/female) | 1/13 | 6/29 | .656 |
| Age (y) | 63.2 ± 15.0 | 62.1 ± 10.2 | .542 |
| Visual acuity (logMAR) | 0.299 ± 0.37 | −0.003 ± 0.17 | .003 a |
| Intraocular pressure (mm Hg) | 13.8 ± 3.5 | 13.4 ± 2.5 | .489 |
| Refractive error (D) | −9.3 ± 4.6 | −11.9 ± 4.2 | .142 |
| Axial length (mm) | 28.9 ± 2.9 | 28.4 ± 1.8 | .358 |
| Central retinal thickness (μm) | 211.6 ± 57.3 | 208.0 ± 31.6 | .799 |
| Central choroidal thickness (μm) | 66.1 ± 36.9 | 111.9 ± 58.6 | .012 a |
| Central scleral thickness (μm) | 582.7 ± 200.4 | 470.0 ± 117.8 | .059 |
| Height of inward bulge (μm) | 187.6 ± 114.8 | 97.5 ± 46.7 | .001 a |
The macular complications group was further classified into 2 groups: 6 eyes with choroidal neovascularization (the CNV group, 4 of which showed chorioretinal atrophy, Figure 6 ) and 8 eyes with pigment epithelial detachment (the PED group, 5 of which showed serous retinal detachment, Figure 6 ). The comparison of the 3 groups (CNV group, PED group, and no complications group) by a Kruskal-Wallis 1-way analysis of variance revealed no significant differences among them in sex, intraocular pressure, refraction, and central retinal thickness. The CNV group was significantly older and had a significantly longer axial length compared to the other groups (age, in years: CNV group, 74.8 ± 5.5; PED group, 54.5 ± 13.9; no complications group, 62.1 ± 10.2; P = .008 [vs PED], P = .0035 [vs no complications group]; axial length, in mm: CNV group, 31.4 ± 1.0; PED group, 27.1 ± 2.4; no complications group, 28.4 ± 1.8; P = .0055 [vs PED], P = .0006 [vs no complications group]). The mean (logMAR) BCVA in the CNV group was significantly lower than that of the other 2 groups (CNV group, 0.66 ± 0.27; PED group, 0.027 ± 0.11; no complications group, −0.003 ± 0.17; P = .0026 [vs PED], P = .0002 [vs no complications group]).
The central choroidal thickness in the CNV group was significantly thinner compared to that of the no complications group (CNV group, 46.7 ± 11.3 μm; no complications group, 111.9 ± 58.6 μm; P = .022). The PED group showed a significantly larger value for the central scleral thickness compared to the other groups (CNV group, 434.5 ± 123.0 μm; PED group, 693.9 ± 175.6 μm; no complications group, 470.0 ± 117.8 μm; P = .012 [vs CNV], P = .0012 [vs no complications group]). The bulge height in the PED group was significantly higher than that in the no complications group, and although the difference was not significant, the bulge height in the PED group had a tendency to be higher than that in the CNV group (CNV group, 126.8 ± 74.5 μm; PED group, 233.1 ± 122.4 μm; no complications group, 97.5 ± 46.7 μm; P = .081 [vs CNV], P = .0002 [vs no complications group]) ( Table 2 ).
| Characteristic | Complication | P Value (Kruskal-Wallis Test) | ||||
|---|---|---|---|---|---|---|
| CNV | PED | No Complications | CNV vs PED | CNV vs No Complications | PED vs No Complications | |
| Number of eyes | 6 | 8 | 35 | |||
| Sex (male/female) | 0/6 | 1/7 | 6/29 | .4705 | .2918 | .7697 |
| Age (y) | 74.8 ± 5.5 | 54.5 ± 13.9 | 62.1 ± 10.2 | .0080 a | .0035 a | .1111 |
| Visual acuity (logMAR) | 0.66 ± 0.27 | 0.027 ± 0.11 | −0.003 ± 0.17 | .0026 a | .0002 a | .3058 |
| Intraocular pressure (mm Hg) | 14.5 ± 1.6 | 13.3 ± 4.4 | 13.4 ± 2.5 | .8442 | .3712 | .8378 |
| Refractive error (D) | −10.8 ± 4.4 | −8.0 ± 4.7 | −11.9 ± 4.2 | .4642 | .7433 | .0734 |
| Axial length (mm) | 31.4 ± 1.0 | 27.1 ± 2.4 | 28.4 ± 1.8 | .0055 a | .0006 a | .115 |
| Central retinal thickness (μm) | 200.0 ± 74.1 | 220.3 ± 44.4 | 208.0 ± 31.6 | .4777 | .3281 | .6507 |
| Central choroidal thickness (μm) | 46.7 ± 11.3 | 80.6 ± 43.3 | 111.9 ± 58.6 | .0929 | .0222 a | .115 |
| Central scleral thickness (μm) | 434.5 ± 123.0 | 693.9 ± 175.6 | 470.0 ± 117.8 | .0118 a | .5186 | .0012 a |
| Height of inward bulge (μm) | 126.8 ± 74.5 | 233.1 ± 122.4 | 97.5 ± 46.7 | .0814 | .3014 | .0002 a |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


