Fig. 11.1
Vestibular episodic syndrome includes several clinical entities which may overlap in the same patient and their relatives resulting in intermediate or complete phenotypes such as MD with migraine. SNHL sensorineural hearing loss, AIED autoimmune inner ear disease
This high prevalence of migraine in familial MD when it was compared to the general population, and the frequent occurrence of migraine symptoms during the vertigo spells suggests a common pathophysiological link between migraine and MD.
11.2 Why Is There a Link Between Ménière’s Disease and Migraine?
The clinical practice indicates that some common triggers (stress, menstruation) may facilitate a migraine attack or an episode of vertigo in patients with Ménière’s disease or migraine [20, 21]. These triggers could be epigenetic factors which will enhance the risk of suffering the disease. Moreover, overlapping clinical features among MD, migraine, and vestibular migraine suggest a shared pathogenic mechanism in a multifactorial disease. Of note, acetazolamide is effective in rare genetic disorders related to migraine-like episodic ataxia [22], several patients with VM, and also in some MD patients who do not respond to betahistine [23], suggesting that the membrane homeostasis and its threshold of excitability are crucial to control an attack of migraine or vertigo.
There are several neurobiological or molecular mechanisms to explain the reported association between MD and migraine. These mechanisms include (Fig. 11.2):
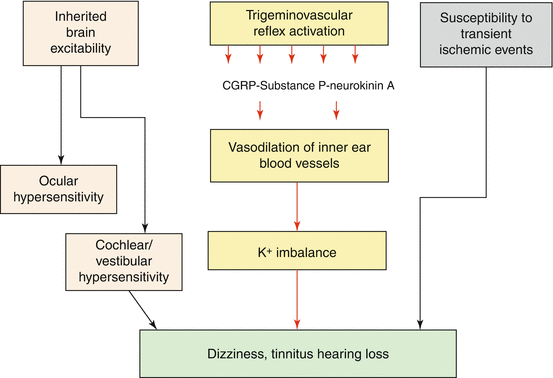
(a)
Inherited alteration of brain excitability which can be observed as hypersensitivity to visual, auditory, or vestibular stimulation
(b)
Sensitization of the trigemino-vascular pathway with release of neuropeptides
(c)
Neurogenic vasodilation of meningeal and inner ear blood vessels
(d)
Susceptibility to transient ischemic events

Fig. 11.2
Neurobiological mechanisms associated with migraine which could be associated with MD
Neurogenic vasodilation and extravasation from meningeal vessels is a well-studied mechanism in migraine [24]. The release of neuropeptides such as calcitonin gene-related peptide (CGRP), neurokinin A, and substance P from dural and cerebral blood vessels produces a local inflammatory response contributing to pain [25]. Furthermore, the electrical stimulation of the trigeminal nerve and the chemical stimulation of cochlear and vertebrobasilar arteries with substances such as capsaicin, histamine, or serotonin produce the extravasation of these neuropeptides in the inner ear [26, 27].
However, the activation of the trigemino-vascular pathway itself is not enough to explain the cochlear-vestibular dysfunction observed in MD. So, an alteration in the ion transport mediated by voltage-dependent channels in the inner ear and brain cortex maybe the common genetic susceptibility leading to variable phenotypes in migraine, MD, or overlapping migraine-Ménière’s disease complex.
Both MD and migraine are multifactorial disorders and the interaction of genetic, epigenetic, and environmental factors contribute to the clinical phenotype. This interaction has been observed in the hypersensitivity to vestibular stimulation found in some individuals with migraine where rotatory or caloric vestibular testing can specifically trigger a migraine attack [28].
Familial clustering has been described in patients with MD and migraine [17, 29]. Moreover, familial MD shows an autosomal dominant inheritance pattern with incomplete penetrance and most of these families have multiple members with migraine [30]. However, familial MD shows genetic heterogeneity [4] and migraine does not segregate with MD in all pedigrees [31]. Currently, it is not clear if there is a genetic basis to explain the association between migraine, episodic vertigo, and MD. Since both disorders seem to be heterogeneous and several genes may contribute to its development, it is likely that genetic or epigenetic factors confer susceptibility to this association, rather than be deterministic [29].
Several cross-sectional and population-based studies using MRI have demonstrated an increased incidence of brain white matter hyperintensities, silent infarct-like lesions, and volumetric changes in gray and white matter regions in migraineurs. These findings suggest that migraine is related with posterior circulation territory ischemic stroke, probably as an independent cerebrovascular risk factor [32].
The risk of transient ischemic events is increased in patients with migraine and sudden sensorineural hearing loss (SSNHL) [33–35]. A population-based study indicates that migraine could be associated with SSNHL, but migraine is comorbid with several cardiovascular risk factors, i.e., diabetes and hypertension, and they probably can explain this comorbidity. Combined audio-vestibular symptoms are also a hallmark of transient ischemic attack and such symptoms typically results from ischemia in the anterior inferior cerebellar artery [36]. Vascular risk factors are probably the common link between migraine and transient episodic symptoms of vertigo, tinnitus, or hearing loss that can culminate in stroke with permanent hearing or vestibular loss, or both mimicking Ménière’s disease [37].
If patients with migraine have an increased risk of cerebral ischemic events, this ischemic risk could affect the cochlear blood flow [38].
11.3 How to Differentiate Vestibular Migraine from Ménière’s Disease?
The clinical picture of VM and MD often overlaps, especially in the early stages of MD; so, it is a real diagnostic challenge to distinguish between both diseases in a patient with recurrent spells of spontaneous vertigo who suffer from migraine. Until now, we do not rely on clinical criteria for VM thereby hindering the differential diagnosis. Undoubtedly, the new definition of VM facilitates the distinction. But anyway, there is no specific diagnostic test for MD or VM; in both conditions, it is a diagnosis of exclusion based on clinical history and a documented fluctuating progressive hearing loss in the case of MD.
The necessity to differentiate between both entities is particularly relevant in a patient with a known MD, who also has migraine and whose symptoms do not respond to conservative medical treatment. In this situation, it is imperative to rule out a VM before attempting a destructive and irreversible procedure.
We need to distinguish between VM and MD, first, because they have a different therapeutic approach and, second, because they also exhibit a different prognosis.
We can try to differentiate between MD and VM according to a combination of their clinical features, the results of functional testing, and magnetic resonance imaging (MRI).
11.3.1 Clinical Features
First of all, the diagnostic criteria for MD [5] and VM [6] are not enough and a follow-up of several months is usually required to distinguish both syndromes. So, fluctuating hearing loss, tinnitus, and aural pressure may also occur in VM, but hearing loss does not progress to profound levels. Susceptibility to motion sickness may be associated with VM; however, as it also occurs with various other vestibular disorders, it is not included as diagnostic criteria.
According to the 3rd edition of the International Criteria for Headache Disorders (ICHD-III beta), “when the criteria for MD are met, particularly hearing loss as documented by audiometry, MD should be diagnosed, even when migraine symptoms occur during the vestibular attacks. Only patients who have two different types of attacks, one fulfilling the criteria for VM and the other for MD, should be diagnosed with both disorders. A future revision of ICHD may include a VM/MD overlap syndrome” [39].
Phonophobia is the most common auditory symptom associated with migraine attacks [11, 14]. Sometimes it is difficult to distinguish phonophobia from hyperacusis.
Tinnitus and aural fullness, both unilateral and bilateral, have been described in migraine and VM patients [14, 40]. More recently, Brantberg and Baloh [2011] found that 68 % of the patients of a group with MD described two or more of the three characteristic auditory symptoms (unilateral hearing loss, unilateral tinnitus, and unilateral aural fullness) with at least half of the vertigo spells as compared with 18 % of a group of patients with benign recurrent vertigo. Furthermore, none of the three auditory symptoms were reported by 19 % of MD patients and 68 % by benign recurrent vertigo patients. A multivariate analysis revealed that the association of unilateral auditory symptoms is the most useful clinical differential characteristic to distinguish between MD and VM [12].
A longitudinal study in patients with VM has demonstrated that auditory symptoms during the vertigo spells increase from 16 % initially to 49 % after a median follow-up of 9 years, and cochlear symptoms in the interictal interval change from 26 to 77 % at the same time interval [41]. Specifically, hearing loss in the free interval varies from 15 to 38 % (Table 11.1).
Table 11.1
Comparison between Ménière’s disease and vestibular migraine
Ménière’s disease | Vestibular migraine | |
|---|---|---|
Age of onset | 35–60 years | Any age (20–60) |
Gender | Equal | Female preponderance |
Duration of attacks | 20 min to hours | Seconds to days |
Triggers | + | ++ |
Family history of vertigo or hearing loss | + | + |
Family history of migraine | + | ++ |
Present or previous history of migraine | +/++ | +++ |
Interictal hearing loss | +++ | + |
Symptoms during an attack | ||
Vertigo | ++ | +++ |
Photo-/phonophobia | ++ | + |
Headache | + | ++/+++ |
Auditory symptoms | +++ | +/++ |
Aura | −/+ | + |
Interictal vestibular function abnormalities | + (early stage)/+++ (advance stage) | ++ |
11.3.2 Functional Testing
11.3.2.1 Hearing
The exact determination of hearing thresholds is crucial to distinguish VM and MD. Typically, MD begins with a low-frequency fluctuating unilateral sensorineural hearing loss coincident with spells of vertigo. As the episodes of vertigo recur and the disease progresses, the hearing loss worsens, involving all frequencies, until it stabilizes, no longer fluctuates, and becomes permanent [42]. The initial presentation of MD is highly variable and may be apparent initially only with episodic vertigo or fluctuating hearing loss.
Sensorineural hearing loss is uncommon in patients with VM although several patterns of hearing loss have been described: unilateral/bilateral and fluctuating/permanent [40, 43]. Fluctuating sensorineural hearing loss is not specific of MD as it has also been observed in VM [41] and other disorders such as autoimmune inner ear disease, Cogan’s syndrome, otosyphilis, and enlarged vestibular aqueduct syndrome. When hearing impairment is present in VM patients, audiometry usually demonstrates a low-frequency, mild-moderate, bilateral sensorineural hearing loss; but permanent unilateral hearing loss and even SSNHL have also been associated with migraine and vestibular migraine [33–35]. Unlike MD, hearing loss is usually episodic and it progresses much more slowly.
11.3.2.2 Vestibular Bedside Examination
The presence of vestibular abnormalities in bedside examination and vestibular function testing is well recognized in MD. Head-shaking nystagmus and vibration-induced nystagmus are the more common vestibular signs in patients with MD [44, 45]. The head impulse test yields positive result in less than half of MD patients. Spontaneous nystagmus is often detected during an attack, but its direction varies, beating both toward the affected ear (irritative nystagmus) or the healthy ear (paralytic nystagmus), so it cannot be considered a localizing finding. In the intercrisis period, the frequency of spontaneous and positional nystagmus varies widely among different series.
Patients with migraine and VM also exhibit abnormal results in clinical examination and vestibular function testing both during and between the attacks: spontaneous nystagmus, positional nystagmus, gaze-evoked nystagmus, saccadic pursuit, and unilateral vestibular hypofunction [41, 46, 47]. However, these findings are not specific for VM and can indicate either a peripheral or central origin.
Recently, Shin et al. [18] have compared interictal vestibular function in both patients with MD and VM. They have found that patients with MD show significantly more abnormal results than patients with VM using head-shaking nystagmus, vibration-induced nystagmus, or bithermal caloric tests. Overall, abnormal result was found on at least one of these three tests in 84 % of MD patients and 66 % of patients with VM. It should be noted that in patients with VM, 50 % had abnormal head-shaking nystagmus, 32 % abnormal vibration-induced nystagmus, and 25 % abnormal canal paresis [48].
11.3.2.3 Instrumental Testing
Although instrumental vestibular examination is not usually recommended to distinguish MD and VM, it may yield relevant information and anticipate the diagnosis of MD. Recently, Neff et al. (2012) has found differences statistically significant in the following vestibular tests when comparing MD patients and VM group: head thrust test, head-shaking nystagmus, vibration-induced nystagmus, abnormal caloric asymmetry, rotatory chair gain, abnormal rotatory chair phase, symmetry and summary, and VEMPs [45].
Unilateral vestibular hypofunction in caloric testing is observed in up to 75 % of unilateral MD patients [48], although it is worth noting that a normal bithermal caloric response has been reported in up to 50 % of patients in some series. Unilateral canal paresis usually indicates the involved ear, but it has also been demonstrated in 19 % of patients on the normal side [49].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


