Purpose
To report the visual and anatomic outcomes of anti–vascular endothelial growth factor (VEGF) monotherapy in the management of marked submacular hemorrhage secondary to neovascular age-related macular degeneration (AMD).
Design
Retrospective, interventional, consecutive case series.
Methods
Nineteen eyes of 18 patients with neovascular AMD and fovea involving submacular hemorrhage comprising greater than 50% of the lesion area were treated with anti-VEGF monotherapy. Main outcome measures included mean visual acuity change from baseline, mean central lesion thickness change from baseline, mean number of injections at 6 months, and adverse events. Snellen visual acuity was converted to approximate ETDRS letter score for the purpose of statistical analysis.
Results
The mean change in approximate ETDRS letter score from baseline was +12 letters at 3 months ( P = .003), +18 letters at 6 months ( P = .001), and +17 letters at 12 months follow-up ( P = .02). Seven eyes received ranibizumab, 6 eyes received bevacizumab, and 6 eyes received both at various time points. The mean number of injections at 6 months was 4.7. The mean OCT central lesion thickness decreased from 755 μm to 349 μm at 6 months follow-up ( P = .0008).
Conclusions
Management with anti-VEGF monotherapy may yield visual and anatomic improvements in eyes with marked submacular hemorrhage secondary to neovascular AMD.
Submacular hemorrhage, a well-recognized complication of neovascular age-related macular degeneration (AMD), may cause substantial visual impairment. Potential mechanisms include iron toxicity, shearing of photoreceptors by fibrin clots, and physical separation of photoreceptors from the retinal pigment epithelium. A more obvious cause of severe visual loss is subretinal fibrosis and disciform macular scar formation. Historically, management options have included surgical submacular clot evacuation or pneumatic displacement with or without the use of tissue plasminogen activator (tPA). These strategies have produced mixed visual results with potentially high complication rates. In practice, many of these patients are probably simply observed, usually with relatively poor visual results.
The Minimally Classic/Occult Trial of Anti-VEGF Antibody Ranibizumab in the Treatment of Neovascular AMD (MARINA) and Anti-VEGF Antibody for the Treatment of Predominantly Classic Choroidal Neovascularization in AMD (ANCHOR) study protocols excluded patients with predominantly hemorrhagic neovascular lesions. However, the effectiveness of anti–vascular endothelial growth factor (VEGF) therapy for neovascular AMD has prompted its application in cases of submacular hemorrhage secondary to choroidal neovascularization (CNV). To date, there are few clinical series validating the efficacy of anti-VEGF therapy in treating these types of lesions. The present study therefore aims to evaluate the visual and anatomic outcomes of anti-VEGF monotherapy in the management of marked submacular hemorrhage secondary to neovascular AMD.
Methods
Institutional review board approval from the University of Miami Miller School of Medicine was obtained to review patient data for this retrospective, interventional, consecutive case series from 3 treating physicians (H.W.F., W.E.S., P.J.R.) at Bascom Palmer Eye Institute. Informed consent was not required for this de-identified review.
Medical records were reviewed for all patients presenting between 2008 and 2011 with a diagnosis of neovascular AMD and fovea involving submacular hemorrhage comprising greater than 50% of the lesion area for which they were receiving anti-VEGF monotherapy. Eyes with submacular hemorrhage secondary to causes other than neovascular AMD, prior vitrectomy surgery, and less than 3 months follow-up were excluded. Of note, no eyes seen by the abovementioned treating physicians underwent pneumatic displacement of submacular hemorrhage during the time interval examined by this retrospective review. The following data were collected: age, sex, date of submacular hemorrhage diagnosis, baseline area of submacular hemorrhage, best-corrected Snellen visual acuity using the most up-to-date distance correction at each visit, optical coherence tomography (OCT) analysis of central lesion thickness, and number and type of intravitreal injections administered.
The Cirrus spectral-domain OCT (SDOCT) instrument (Carl Zeiss Meditec Inc, Dublin, California, USA) was used for all study evaluations. The central macula of each eye was imaged using a 5-line raster scan pattern. This protocol resulted in the acquisition of a data set organized as 4096 A-scans in each B-scan and as 5 horizontal B-scans in each raster array. Central lesion thickness was manually measured to be the distance from the internal limiting membrane to Bruch membrane on SDOCT images centered on the fovea.
Snellen visual acuities were converted to approximate Early Treatment Diabetic Retinopathy Study (ETDRS) letter scores using a validated formula for the purpose of statistical analysis. Changes in visual acuity and OCT central lesion thickness measurements from baseline were assessed by a paired Student t test. A P value less than .05 was considered statistically significant. Clinical outcome measures included mean visual acuity change from baseline, mean central lesion thickness change from baseline, mean number of injections at 6 months, and adverse events during treatment or follow-up.
Results
Nineteen eyes of 18 patients met the inclusion and exclusion criteria. The mean age was 81 years. Seven eyes received ranibizumab, 6 eyes received bevacizumab, and 6 eyes received both. The range to last follow-up examination was 4-30 months. The mean number of injections over the first 6 months was 4.7 (range 2-7, n = 15). The mean baseline area of submacular hemorrhage was 39.0 mm 2 (range 4.3-170.2 mm 2 , n = 16).
Eighteen of 19 eyes (95%) were evaluated at the 3-month follow-up interval. Median Snellen visual acuity improved from 20/400 (mean 20/300; range 20/40 to hand motions) at baseline to 20/250+1 (mean 20/160−2; range 20/30 to count fingers [CF]) at 3 months following presentation. Fifteen of 19 eyes (79%) were evaluated at the 6-month follow-up interval. Median Snellen visual acuity improved from 20/400 (mean 20/300+2; range 20/40 to CF) at baseline to 20/200 (mean 20/125+1; range 20/20 to 20/800) at 6 months following presentation. Ten of 19 eyes (53%) were evaluated at the 12-month follow-up interval. Median Snellen visual acuity improved from 20/400−1 (mean 20/250−1; range 20/40 to CF) at baseline to 20/125−2 (mean 20/125+1; range 20/20 to 20/800) at 12 months following presentation. Table 1 lists the visual acuity outcomes converted to approximate ETDRS letter scores at 3, 6, and 12 months follow-up. The mean change in approximate ETDRS letter score from baseline was +12 letters at 3 months ( P = .003, n = 18), +18 letters at 6 months ( P = .001, n = 15), and +17 letters at 12 months ( P = .02, n = 10). Table 2 lists the distribution of visual acuity gains in each cohort.
| Follow-up Period | Eyes, No. | Mean Baseline Snellen VA | Mean Baseline Approximate ETDRS Letter Score (SD) | Mean Follow-up Snellen VA | Mean Follow-up Approximate ETDRS Letter Score (SD) | Mean Approximate ETDRS Letter Score Change (SD) | P Value |
|---|---|---|---|---|---|---|---|
| Month 3 | 18 | 20/300 | 26 (24) | 20/160−2 | 38 (27) | 12 (15) | .003 |
| Month 6 | 15 | 20/300+2 | 28 (23) | 20/125+1 | 46 (28) | 18 (18) | .001 |
| Month 12 | 10 | 20/250−1 | 29 (28) | 20/125+1 | 46 (29) | 17 (18) | .02 |
| Month 3 (n = 18) | Month 6 (n = 15) | Month 12 (n = 10) | |
|---|---|---|---|
| ≥3 lines gain | 8 (44%) | 9 (60%) | 6 (60%) |
| Stable | 9 (50%) | 6 (40%) | 4 (40%) |
| ≥3 lines loss | 1 (6%) | 0 | 0 |
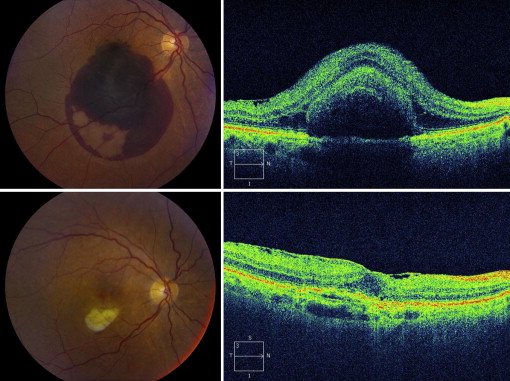
The hemorrhage was located in the subretinal space in 4 of the 19 eyes (21%), in the sub–retinal pigment epithelial (RPE) space in 5 eyes (26%), and in both the subretinal and sub-RPE spaces in 10 eyes (53%) ( Figures 1-3 ). The mean OCT central lesion thickness decreased from 755 μm (range 274-1496 μm) at baseline to 349 μm (range 156-692 μm) at 6 months follow-up ( P = .0008, n = 12). No adverse ocular (eg, retinal detachment, endophthalmitis, cataract progression) or systemic events were observed over the course of the study.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


